Author Affiliations
Abstract
Pellucid marginal degeneration (PMD) is a rare corneal ectatic disorder that can lead to progressive visual impairment. Spontaneous corneal perforation is an uncommon but severe complication. This case highlights the need for early recognition and management of PMD to prevent vision-threatening outcomes. A 28-year-old female presented with acute right eye pain, rapid visual deterioration over a few hours, and progressive bilateral visual decline over three years. Examination and corneal topographic studies confirmed bilateral PMD with spontaneous corneal perforation in the right eye and classic signs of peripheral thinning and ectasia in the left eye. Conservative management with topical corticosteroids, prophylactic antibiotics, and patching led to visual improvement. The patient’s condition improved without the need for surgical intervention, demonstrating the potential effectiveness of timely conservative therapy in selected cases. This case emphasizes the need for prompt recognition and intervention in PMD to prevent severe complications. Increased awareness of corneal ectatic disorders can aid in timely diagnosis and management, preserving vision in affected individuals.
Keywords
Pellucid marginal degeneration, Spontaneous corneal perforation, Corneal ectasia, Peripheral corneal thinning, Corneal perforation management, Early diagnosis.
Introduction
PMD is a bilateral progressive non-inflammatory peripheral corneal thinning disorder causing irregular astigmatism and progressive visual reduction of unknown etiology.[1] Though the pathophysiology is still unclear, collagen abnormalities are thought to be a contributing factor to PMD, given its similarity to keratoconus. As the cornea thins, it can become weak and be pushed outwards by the intraocular pressure. On electron microscopy, there is increased spacing between fibrous collagen compared to the normal cornea collagen structure. Histologically, Bowman’s membrane is absent or broken, although a normal epithelium, endothelium, and Descemet’s membrane are seen.[2] Patients with this disease classically present with ongoing distance vision reduction, without correction by glasses. However, pinhole visual acuity testing is usually normal. The disease is seen more in patients within the second to fifth decades of life and those with family histories of other corneal ectatic disorders like keratoconus and keratoglobus.[3] PMD is considered a rare disease, and corneal perforation has been reported infrequently. However, it is hypothesized that the prevalence of PMD may be underestimated due to some cases being misdiagnosed as keratoconus.[2] In more advanced cases, acute corneal hydrops may result from a rupture in Descemet’s membrane, although this is rarely seen.[4]
PMD is a clinical diagnosis based on slit lamp exam and reported symptoms of the patient. On slit-lamp examination, a characteristic peripheral band of corneal thinning in the inferior cornea from 4 to 8 o’clock can be seen. The area of thinning is always epithelialized, clear, and avascular without any lipid deposition. Corneal topography or tomography can also support the diagnosis. In addition, pachymetry can be used to measure inferior corneal thinning and will show corneal thickening from the center to the periphery, which is the opposite of the normal pattern. Treatment of PMD is difficult due to the degree of corneal topography abnormality, but it can include wearing glasses or contact lenses. If medical treatment fails, surgical options may include penetrating keratoplasty.[2]
Case Presentation
A 28-year-old female patient from Addis Ababa, Ethiopia, presented to Menellik II Hospital, a tertiary eye care center, with sudden right eye pain associated with rapid vision reduction over a few hours. The patient reported accompanying symptoms of tearing, redness, and photosensitivity. She disclosed a history of progressive distance visual decline over the past three years, unresponsive to multiple spectacle correction attempts. The patient recalled experiencing similar ocular pain and visual reduction in her left eye eight months prior, for which she received multiple eye drops, including hypertonic saline and topical corticosteroids. Although no diagnosis was made at the time, the clinical features and course suggest the possibility of a prior episode of spontaneous perforation or corneal hydrops in the left eye. This indicates a likely sequential, rather than simultaneous, bilateral presentation.
The patient denied any history of trauma, ocular itching, allergies, contact lens use, or current systemic or topical ocular medication. She reported no systemic illnesses. Ocular examination at presentation, using the Snellen chart, revealed best-corrected visual acuity of count fingers at 1 meter in the right eye and 20/40 in the left eye. Intraocular pressure was normal in both eyes, and external examination showed no signs of ocular trauma.
Slit lamp biomicroscopy of the right eye revealed mild bulbar conjunctival injection. Corneal findings included inferior corneal edema and perforation from 3 to 8 o’clock, with a crescent pattern of involvement approximately 1.5mm from the inferior limbus (Figure 1-3). The corneal sensation was intact and comparable. Fluorescein staining showed leakage but no ulcer. No corneal vascularization or deposits, such as lipid or hemosiderin, were observed. The anterior chamber was shallow with a van Herick grading of 1. No anterior chamber reaction, hyphema, or hypopyon was noted (Figure 4). The pupil was oval, mid-dilated, and sluggishly reactive. Further examination of the lens, anterior vitreous, and fundus was unremarkable.
Slit lamp examination of the left eye revealed significant diagnostic clues, including an inferior subepithelial opacity approximately 2mm from the limbus, extending from 4 to 9 o’clock position with a linear length of 8mm. This opacity was associated with a crescent-shaped stromal thinning and an area of ectasia and bulging just superior to it (Figure 5,6). No vascularization, Fleischer’s iron rings, or Vogt’s striae were observed. The anterior chamber depth was graded as +4, with no reaction noted. Examination of the lens, vitreous, and fundus of the left eye yielded normal findings.
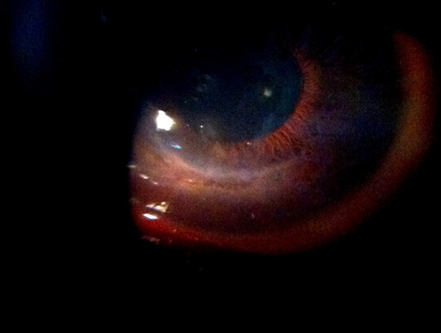
Figure 1: Inferior corneal perforation – Right eye (OD)
A slit lamp image showing an inferior corneal perforation extending from the 3 to 8 o’clock position, consistent with advanced PMD. The affected area is crescent-shaped and located approximately 1.5 mm from the limbus
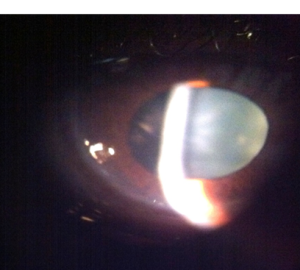
Figure 2: Magnified view demonstrating the characteristic crescentic zone of corneal thinning and edema inferiorly, further supporting the diagnosis of spontaneous perforation due to PMD
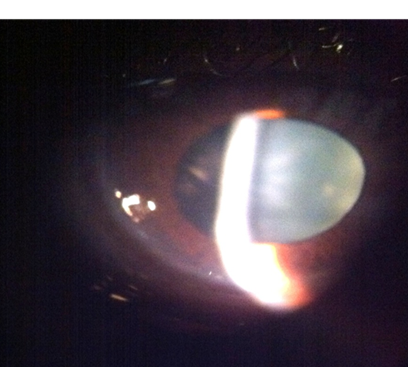
Figure 3: This image highlights the clear, avascular epithelium overlying the perforated zone, a hallmark feature of PMD-related corneal thinning without active ulceration or infection

Figure 4: flat anterior chamber (AC) – OD
Slit lamp photograph showing a shallow anterior chamber with a Herick grade of 1, consistent with corneal perforation and aqueous leakage.

Figure 5: Left eye (OS) Subepithelial opacity, peripheral thinning, and characteristic corneal protrusion
Slit lamp image of the left eye reveals a crescent-shaped subepithelial opacity 2 mm from the limbus, extending from 4 to 9 o’clock, indicative of early PMD with stromal thinning.
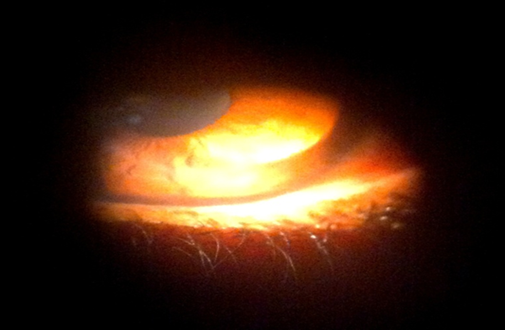
Figure 6: Ectasia and bulging above the area of thinning in the left eye (OS)
Figure 6 shows corneal protrusion just superior to the thinned region, a classic sign of PMD, with no evidence of vascularization or iron deposition. To assist in diagnosing the patient’s condition, several investigations were conducted. Retinoscopy revealed astigmatism in the left eye, while autorefraction indicated inferior corneal steepening in the same eye. Specific refractive measurements were obtained: the right eye showed a sphere of 0.87, cylinder of -5.75, and an axis of 67°, whereas the left eye demonstrated a sphere of -3.5, cylinder of -1, and an axis of 90°. Corneal pachymetry readings were 496 μm for the right eye (OD) and 502 μm for the left eye (OS). These findings, particularly the inferior steepening on autorefraction and the presence of peripheral thinning on pachymetry, are consistent with the typical presentation of pellucid marginal degeneration. The significantly increased cylindrical power in the right eye further supports the diagnosis of irregular astigmatism due to ectatic corneal changes.
| Parameter | Right eye (OD) | Left eye (OS) |
| Symptoms | Sudden pain, tearing, photophobia, rapid visual loss | Similar episode 8 months prior, gradual decline over 3 years |
| Visual acuity | Counting fingers at 1 meter | 20/40 |
| Slit lamp findings | Inferior corneal edema and perforation (3–8 o’clock), clear epithelium, shallow AC | Inferior stromal thinning (4–9 o’clock), ectasia above thinning, deep AC |
| AC | Shallow (Van Herick Grade 1) | Deep (+4), no reaction |
| Corneal vascularization | None | None |
| Corneal sensation | Intact | Intact |
| Pupil | Oval, mid-dilated, sluggish | Normal |
| Fundus and lens exam | Unremarkable | Unremarkable |
| Refraction (Autorefraction) | Sphere: +0.87, Cylinder: –5.75, Axis: 67° | Sphere: –3.50, Cylinder: –1.00, Axis: 90° |
| Pachymetry | 496 µm | 502 µm |
Table 1: Comparative summary of clinical findings between right and left eyes
Additionally, a lensometer was used to assess the patient’s current spectacles. The right eye prescription was found to be sphere -3D, cylinder -1D, axis 90°, while the left eye had a sphere of -3D.
Case Management
For the right eye, a conservative management approach was initiated. This included the administration of prophylactic broad-spectrum antibiotics (ciprofloxacin 0.3%), cycloplegic (atropine), and topical steroids. The treatment plan also encompassed daily follow-ups, full-time patching, and bed rest.
The patient was followed daily for the first week, during which conservative management successfully maintained anterior chamber depth without leakage or development of secondary keratitis or cataract. Daily patching was discontinued after a stable chamber depth of +1 was achieved, and prophylactic topical antibiotics were continued. Subsequently, the patient was monitored closely every other day. Topical dexamethasone gradually increased to treat the corneal hydrops, resulting in visual improvement to 20/120 prior to refraction. These findings support the short-term efficacy of conservative treatment in preserving corneal integrity and promoting visual recovery.
Discussion
PMD is a progressive, non-inflammatory peripheral corneal thinning disorder characterized by peripheral thinning of the inferior cornea from 4 o’clock to the 8 o’clock position, accompanied by 1-2 mm of normal cornea between the limbus and the thinned cornea. This ecstatic condition of the cornea causes poor quality of vision due to the associated irregular astigmatism.[5,6] Progressive corneal ectasia in keratoconus and PMD can rarely lead to Descemet’s membrane rupture, causing acute hydrops, which have been shown to respond favorably to conservative management.[4,7]
Although uncommon, this presentation of spontaneous perforation has been reported in the literature, with corneal perforation occurring one year apart between the two eyes.[7] This clinical scenario appears to be similar to that of our patient, as she gave a history of a similar presentation in her left eye eight months before her current episode. The absence of trauma, eye rubbing, or use of contact lenses makes the presence of spontaneous perforation in our patient unique.
The treatment for PMD primarily involves contact lens wear. Initially, eyeglasses can be prescribed, but due to the progressive nature of the ectasia, irregular astigmatism typically necessitates contact lens wear, including soft contact lenses, toric hydrophilic contact lenses, hybrid contact lenses, and rigid gas-permeable contact lenses.[1] Surgery is reserved for 10-15% of cases.[8]
Regarding the treatment of corneal hydrops and spontaneous corneal perforation, Forooghian et al. reported conservative management of spontaneous perforation using a therapeutic bandage lens and prophylactic topical antibiotic therapy. This approach requires close observation for worsening of the perforation size and potential infectious keratitis.[8] In our patient, we initiated prophylactic therapy as soon as we settled on the diagnosis, using topical ciprofloxacin 0.3%, atropine 1%, and dexamethasone 0.1% eye drops with full-time patching (a therapeutic bandage contact lens was not available).
As for the surgical management of advanced PMD, Rashed and Rabinowitz suggested a two-step approach, starting with an inferior lamellar keratoplasty to eliminate the ectatic thin cornea, followed by standard-sized penetrating keratoplasty.[1,9] They followed five patients using this approach, showing marked improvement in corneal topography and visual acuity. However, the full applicability is yet to be studied, as long-term follow-up is required to assess the efficacy of these newer procedures. Others have suggested the use of C-shaped lamellar keratoplasty, wedge resection, and replacement by a freehand corneo-scleral graft, diathermy, and large epikeratophakia grafts, which have also been used in the management of keratoconus.
Kymionis et al.[3] suggested using an intracorneal ring segment in the early stages of the disease. Their study showed improvement in the inferior corneal steepening and, ultimately, in visual acuity. Corneal biomechanical changes may benefit from cross-linking using vitamin B12 and ultraviolet A. This was attempted in a patient with an early PMD after a failed intracorneal ring segment.
Although advanced surgical techniques have shown promise, they may not be readily accessible in all clinical settings. In many resource-limited settings, diagnosing PMD can be particularly challenging due to limited access to both corneal imaging modalities, such as topography or tomography, and specialist expertise. In such contexts, clinical features of PMD may be mistaken for keratoconus or other ectatic disorders, especially when diagnosis is based solely on slit-lamp findings. Greater clinician awareness and training in recognizing the clinical signs of PMD are therefore essential for timely diagnosis.
Conclusion
This case highlights a rare presentation of PMD in a young adult who developed spontaneous corneal perforation without any history of trauma or contact lens use. The diagnosis was established through clinical examination and corneal imaging, with notable findings in both eyes. Conservative treatment with topical antibiotics, cycloplegics, corticosteroids, and patching resulted in symptom improvement without the need for surgical intervention. This case emphasizes the importance of considering PMD in patients with ongoing visual deterioration despite spectacle correction. Early diagnosis of the condition helps prevent the vision-threatening complication of perforation or misdiagnosis, especially in settings with limited resources and trained staff.
References
- Rasheed K, Rabinowitz YS. Surgical treatment of advanced pellucid marginal degeneration. Ophthalmology. 2000;107(10):1836-1840. doi:10.1016/s0161-6420(00)00346-8 PubMed | Crossref | Google Scholar
- Paulus YM, Bunya VY, Woodward MA, Weissbart SB, Vannadil H, Paulus YM. Pellucid marginal corneal degeneration. EyeWiki. 2024. Pellucid marginal corneal degeneration
- Kymionis GD, Karavitaki AE, Kounis GA, Portaliou DM, Yoo SH, Pallikaris IG. Management of pellucid marginal corneal degeneration with simultaneous customized photorefractive keratectomy and collagen crosslinking. J Cataract Refract Surg. 2009;35(7):1298-1301. doi:10.1016/j.jcrs.2009.03.025 PubMed | Crossref | Google Scholar
- Gharebaghi D, Fallahi B, Javadzadeh A, Amiraslanzadeh G. Spontaneous corneal hydrops and perforation in pellucid marginal degeneration; a case report. J Ophthalmic Vis Res. 2009;4(3):174-176. Spontaneous corneal hydrops and perforation in pellucid marginal degeneration; a case report
- Kaushik S, Jain AK, Saini JS. Unilateral pellucid marginal degeneration. Eye (Lond). 2003;17(2):246-248. doi:10.1038/sj.eye.6700301 PubMed | Crossref | Google Scholar
- Maguire LJ, Klyce SD, McDonald MB, Kaufman HE. Corneal topography of pellucid marginal degeneration. Ophthalmology. 1987;94(5):519-524. doi:10.1016/s0161-6420(87)33416-5 PubMed | Crossref | Google Scholar
- Forooghian F, Assaad D, Dixon WS. Successful conservative management of hydrops with perforation in pellucid marginal degeneration. Can J Ophthalmol. 2006;41(1):74-77. doi:10.1016/S0008-4182(06)80071-2 PubMed | Crossref | Google Scholar
- Lee WB, O’Halloran HS, Grossniklaus HE. Pellucid marginal degeneration and bilateral corneal perforation: case report and review of the literature. Eye Contact Lens. 2008;34(4):229-233. doi:10.1097/ICL.0b013e318164771b PubMed | Crossref | Google Scholar
- Tuffaha BT, Alio JL, Piñero DP. Crosslinking for the Management of Pellucid Marginal Degeneration in a Case of an Unsuccessful Implantation of Intracorneal Ring Segments. Int J Kerat Ect Cor Dis 2012;1(2):120-124. doi:10.5005/jp-journals-10025-1022 Crossref | Google Scholar
Acknowledgments
None
Funding
None
Author Information
Corresponding Author:
Miriam Michael
Department of Internal Medicine,
Howard University, Washington, DC, USA
Email: michaelclarkefamily@gmail.com
Co-Authors:
Mahlet Abrie, Yewbnesh Somega, Samrawit Zinabu, Mekdem Bisrat, Elizabeth Beyene, Huda Gasmelseed
Department of Internal Medicine
Howard University, Washington, DC, USA
Khalid Mohamed, Kira Yates, Luke Nelson, Francisco Kelley, Ahmad Mohammed
College of Medicine
Howard University, Washington, DC, USA
Authors Contributions
Mahlet Abrie and Yewbnesh Somega contributed to the conceptualization of the study and were responsible for writing the original document. Khalid Mohamod edited the review and wrote the final version. Kira Yates, Luke Nelson, Francisco Kelley, and Ahmad Mohammed contributed to writing the final document, editing references, and formatting. Samrawit Zinabu, Elizabeth Beyene, Mekdem Bisrat, and Huda Gasmelseed assisted in editing the final document. Miriam Michael contributed to the conceptualization, edited the final document, and provided supervision throughout the project.
Conflict of Interest Statement
None
Guarantor
None
DOI
Cite this Article
Abrie M, Somega Y, Mohamed K, et al. Spontaneous Corneal Perforation in Pellucid Marginal Degeneration: A Case Report from Menelik II Referral Hospital, Ethiopia. medtigo J Emerg Med. 2025;2(2):e3092225. doi:10.63096/medtigo3092225 Crossref









