Author Affiliations
Abstract
Background: Spinal deformities are common complications in paediatric neuromuscular disorders (NMDs), with variability in prevalence, predictors, and outcomes across conditions. A cross-diagnostic synthesis is needed to guide surveillance and management.
Objective: To systematically review and meta-analyse the prevalence, predictors, and management outcomes of spinal deformities in children with NMDs.
Methodology: This review followed PRISMA 2020 guidelines. MEDLINE, Embase, Cochrane Library, CINAHL, and Web of Science were searched from inception. Observational studies and trials reporting radiographically confirmed spinal deformity (Cobb ≥10°) in individuals ≤18 years with NMDs were included. Random-effects meta-analysis using DerSimonian-Laird with Freeman-Tukey transformation pooled prevalence estimates. Heterogeneity was assessed using I², and certainty of evidence using GRADE.
Results: Sixteen studies met the inclusion criteria, with thirteen contributing prevalence data. The pooled prevalence of scoliosis was approximately 60%, with substantial heterogeneity. Diagnosis-specific estimates were 45–50% in cerebral palsy, 75–80% in Duchenne muscular dystrophy, and 70–85% in spinal muscular atrophy. Severe scoliosis (Cobb ≥40°) occurred in about 20% of cerebral palsy cases. Non-ambulatory status and higher functional severity (GMFCS IV–V) predicted progression. Surgical correction improved radiographic outcomes, but complication rates remained high (30–33%), predominantly pulmonary (20–25%).
Conclusion: Spinal deformity affects most children with NMDs, especially those with severe impairment. While surgery improves alignment, complication risks are considerable. Early diagnosis-specific surveillance and multidisciplinary care are essential to optimise outcomes.
Keywords
Neuromuscular disorders, scoliosis, cerebral palsy, Duchenne muscular dystrophy, spinal muscular atrophy.
Introduction
Neuromuscular disorders in childhood comprise a heterogeneous group of conditions characterised by impaired muscle function due to pathology affecting the motor unit, including the anterior horn cell, peripheral nerve, neuromuscular junction, or muscle fibre. Common paediatric neuromuscular conditions include cerebral palsy (CP), Duchenne muscular dystrophy (DMD), spinal muscular atrophy (SMA), and paediatric spinal cord injury (SCI). These disorders are frequently associated with progressive muscle weakness, imbalance of paraspinal musculature, impaired trunk control, and altered skeletal growth, predisposing affected children to spinal deformities.[1-6]
Spinal deformities, particularly scoliosis, represent one of the most common orthopaedic complications in paediatric NMD populations. Neuromuscular scoliosis differs fundamentally from idiopathic scoliosis in its earlier onset, rapid progression, long C-shaped curves, frequent pelvic obliquity, and association with severe functional impairment.[2,6] In children with CP, population-based studies have demonstrated scoliosis prevalence ranging from approximately 21% to 80%, with markedly increased risk among those classified as Gross Motor Function Classification System (GMFCS) levels IV and V.[1-4]
In DMD, scoliosis typically develops following loss of ambulation, with historical cohorts reporting prevalence rates exceeding 70% in untreated populations.[7,8] More recent surveillance data suggest that corticosteroid therapy may delay onset and reduce surgical intervention rates, although deformity remains common.[8,9] Similarly, SMA particularly type II, is associated with early-onset and progressive scoliosis, with prevalence estimates ranging from 50% to 90% in natural history cohorts.[10,11]
The pathogenesis of spinal deformity in NMDs is multifactorial. Progressive muscle weakness leads to asymmetric loading of the immature spine, impaired postural reflexes, and reduced trunk stability. In non-ambulatory children, prolonged sitting and pelvic obliquity further contribute to progressive curvature. Pelvic asymmetry may both contribute to and result from deformity, worsening sitting balance, and increasing risk of secondary complications such as pressure injury.[1,6]
Spinal deformities in children with NMDs are associated with significant clinical consequences. Progressive thoracic curvature can compromise pulmonary function, reduce vital capacity, and impair cough effectiveness, increasing susceptibility to respiratory complications.[12-14] Functional limitations, including impaired sitting tolerance, difficulty with transfers, chronic pain, and reduced quality of life, are also common.[3,5] The burden extends beyond the child to caregivers, contributing to increased healthcare utilisation and psychosocial strain.
Advances in multidisciplinary care, including respiratory support, nutritional optimisation, corticosteroid therapy in DMD, and disease-modifying treatments in SMA, have improved survival in several neuromuscular conditions.[8,11] As life expectancy increases, long-term musculoskeletal complications such as spinal deformity have become increasingly important determinants of functional outcome and quality of life. This epidemiological transition underscores the importance of structured surveillance and timely intervention.
Problem statement:
Although numerous studies have examined scoliosis within specific neuromuscular diagnoses, reported prevalence, incidence, and progression rates vary widely across populations.[1,6] Differences in diagnostic thresholds (e.g., Cobb angle ≥10° versus ≥20°), functional classification systems, and follow-up duration limit comparability and synthesis.
Furthermore, while predictors such as non-ambulatory status, higher GMFCS level, and loss of ambulation in DMD have been consistently identified, their relative strength and cross-diagnostic applicability remain incompletely synthesised.[3,7,9] Evidence regarding comparative outcomes of conservative versus surgical management is also heterogeneous, with reported overall surgical complication rates of approximately 30-35%, most commonly pulmonary in nature.[14-16]
The absence of consolidated, pooled evidence constrains clinicians’ ability to stratify risk, optimise surveillance intervals, and determine appropriate timing of intervention across diverse neuromuscular populations.
Aim of the study:
To systematically synthesise and quantify available evidence regarding the prevalence, progression, predictors, and management outcomes of spinal deformities in children with neuromuscular disorders.
Research questions:
- What are the pooled prevalence and incidence rates of spinal deformities in paediatric neuromuscular disorders?
- Which clinical, neurological, and functional factors predict the development and progression of severe spinal deformity?
- What are the outcomes and complication rates associated with conservative and surgical management strategies?
Objectives of the study:
- To determine pooled prevalence and incidence estimates of scoliosis across major paediatric neuromuscular diagnoses.
- To identify predictors associated with deformity onset and progression.
- To evaluate outcomes and complication rates of conservative and surgical interventions.
- To assess heterogeneity and certainty of evidence using established methodological frameworks, including the I² statistic for heterogeneity quantification and the GRADE approach for evidence certainty assessment.[17-20]
Significance of the study:
This systematic review and meta-analysis aim to provide consolidated quantitative evidence to inform clinical decision-making across paediatric neuromuscular populations. By integrating data from 16 studies and applying random-effects modelling consistent with established meta-analytic methodology, the study strengthens understanding of disease burden and risk stratification.[19] Use of PRISMA 2020 reporting standards ensures methodological transparency, while application of the I² statistic enables quantification of between-study heterogeneity.[17,20] The findings may inform surveillance protocols, guide multidisciplinary management, and contribute to optimisation of surgical timing and perioperative risk assessment.
Justification and research gap:
Prior reviews have frequently focused on single diagnoses or surgical outcomes in isolation. Few have integrated cross-diagnostic prevalence, predictors, and management outcomes within a unified quantitative framework. Moreover, evolving standards of care, particularly corticosteroid therapy in DMD and disease-modifying treatments in SMA, may be altering the natural history of spinal deformity.[8-11] Given these developments and persistent heterogeneity in reported outcomes, a comprehensive and methodologically rigorous synthesis is warranted.
Scope of the study:
This review includes observational studies and clinical trials reporting spinal deformities in children aged 0-18 years with neuromuscular disorders. Outcomes of interest include Prevalence and incidence of scoliosis (Cobb ≥10°), severe scoliosis (Cobb ≥40°), predictors of progression, surgical correction rates, and perioperative complications.
Operational definitions:
- NMDs: Conditions affecting the motor unit resulting in muscle weakness and impaired motor control.
- Spinal deformity: Abnormal spinal alignment, including scoliosis, kyphosis, lordosis, kyphoscoliosis, and pelvic obliquity.
- Neuromuscular scoliosis: Scoliosis attributable to neuromuscular imbalance, defined radiographically as Cobb angle ≥10°.[2]
- Severe scoliosis: Cobb angle ≥40°.
- Heterogeneity (I²): Percentage of total variability across studies attributable to between-study heterogeneity rather than chance.[20]
Literature review:
Introduction: Spinal deformities are among the most clinically significant musculoskeletal complications affecting children with NMDs. Progressive scoliosis, pelvic obliquity, and kyphotic deformity are frequently observed in conditions characterised by muscle weakness, impaired postural control, and abnormal biomechanical loading of the immature spine.[1-6] The burden of spinal deformity varies considerably across diagnoses but is consistently associated with increased morbidity, functional limitation, and healthcare utilisation.
Unlike adolescent idiopathic scoliosis, neuromuscular scoliosis arises secondary to muscle imbalance and neurological impairment. It is typically characterised by earlier onset, rapid progression, long sweeping curves, pelvic obliquity, and poor sitting balance.[2,6] These distinguishing features underscore the need for diagnosis-specific understanding and management strategies.
Epidemiology of spinal deformity in neuromuscular disorders:
Cerebral palsy (CP): CP is the most common cause of physical disability in childhood and is strongly associated with spinal deformity. Population-based studies from Sweden and Australia have demonstrated wide variation in prevalence estimates, ranging from approximately 21% to 80%, depending largely on functional severity and age.[1-4]
Hägglund et al. reported that scoliosis incidence increases progressively with age, particularly among children classified as GMFCS IV–V.[1] Similarly, Persson-Bunke et al. demonstrated a prevalence of 29% for Cobb angles >10° and 17% for >20° in a total population cohort aged 4-18 years.[2] At skeletal maturity, Willoughby et al. observed scoliosis (Cobb >10°) in 41% of individuals, with severe scoliosis (>40°) in approximately 19%.[4] Severity of motor impairment consistently emerges as the strongest predictor.3 Children who are non-ambulatory (GMFCS IV-V) demonstrate markedly higher rates of curve progression compared with ambulatory peers.
Duchenne muscular dystrophy (DMD): DMD is characterised by progressive muscle degeneration and loss of ambulation, typically during late childhood. The development of scoliosis frequently follows ambulation loss, with historical cohorts reporting prevalence exceeding 70% in untreated populations.[7,8]
Alman et al. demonstrated that glucocorticoid therapy delayed scoliosis onset and reduced the need for surgical intervention.[7] Similarly, Lebel et al. reported that long-term corticosteroid treatment was associated with reduced surgical rates over a 15-year follow-up.[8] More recent surveillance data confirm that although steroid therapy modifies disease trajectory, scoliosis remains highly prevalent.[9] The temporal relationship between loss of ambulation and curve progression highlights the importance of early surveillance in DMD cohorts.
Spinal muscular atrophy (SMA): SMA, particularly type II, is associated with early-onset and progressive spinal deformity. Fujak et al. reported scoliosis in the majority of children with SMA type II, often accompanied by pelvic obliquity.[10] Wijngaarde et al. demonstrated a high lifetime risk of scoliosis surgery, particularly in non-ambulatory subtypes.[11] The natural history of spinal deformity in SMA reflects severe trunk weakness and impaired postural control. Although disease-modifying therapies have altered survival patterns, long-term musculoskeletal outcomes remain an evolving area of investigation.
Pediatric spinal cord injury: Children sustaining spinal cord injury (SCI) are also at significant risk of scoliosis development, particularly when injury occurs at a younger age. Mulcahey et al. identified younger age at injury as a strong predictor of progressive deformity.[12] Kulshrestha et al. reported that approximately half of paediatric SCI patients developed scoliosis within 10 years of injury.[13] These findings further reinforce the role of neuromuscular imbalance and disrupted growth in deformity pathogenesis.
Pathophysiology: The development of neuromuscular scoliosis is multifactorial. Progressive muscle weakness results in asymmetric spinal loading, loss of trunk control, and impaired postural reflexes.[1,6] Reduced weight-bearing and prolonged sitting contribute to altered vertebral growth modulation. Pelvic obliquity exacerbates asymmetric loading and may worsen progression. Unlike idiopathic scoliosis, neuromuscular curves are frequently long, rigid, and associated with poor sitting balance.[2] These biomechanical factors explain the often rapid and severe progression observed in non-ambulatory children.
Clinical consequences: Children with neuromuscular disorders frequently develop progressive spinal deformities, particularly scoliosis, which can significantly worsen systemic health outcomes.[21-23] Surgical studies have demonstrated that patients with severe neuromuscular scoliosis often present with advanced musculoskeletal deformity, respiratory compromise, and limited functional mobility requiring complex corrective procedures such as spinal fusion with pelvic fixation and intraoperative halo-femoral traction.[24-27] These conditions are commonly observed in disorders such as cerebral palsy, spinal muscular atrophy, and other neuromuscular syndromes. The resulting chronic immobility, reduced cardiopulmonary reserve, and altered hemodynamics can predispose affected individuals to long-term vascular complications.[24] As survival among children with neuromuscular disorders improves due to advances in surgical and multidisciplinary care, the long-term neurological consequences, including increased susceptibility to cerebrovascular disease, are becoming increasingly relevant.[24-27]
Earlier surgical outcome series further highlight the significant morbidity burden associated with neuromuscular scoliosis management. Studies evaluating spinal fusion procedures in this population reported high rates of perioperative complications, including respiratory insufficiency, infection, and prolonged postoperative immobilization.[28,29] These complications contribute to systemic physiological stress and reduced physical activity, both of which are recognized contributors to metabolic and vascular dysregulation. Chronic immobility in particular promotes venous stasis, impaired endothelial function, and autonomic instability, factors that may increase the long-term risk of thromboembolic and cerebrovascular events. Consequently, neuromuscular scoliosis should not be viewed solely as an orthopedic complication but as part of a broader multisystem disease process affecting long-term neurological and vascular health.
Recent evidence also highlights the clustering of postoperative complications among children undergoing spinal fusion for neuromuscular scoliosis. Rajkumar et al. demonstrated that children with neuromuscular scoliosis frequently experience multiple complications within the early postoperative period, reflecting the complex physiological vulnerability of this population.[27] These findings align with broader neurological research suggesting that severe neurological insults and chronic neurological disorders may predispose individuals to subsequent neurological sequelae. For example, systematic evidence examining neurological complications after traumatic brain injury indicates that early neurological pathology may influence long-term neurological morbidity.[28] In the context of neuromuscular disorders, the interplay between chronic disability, systemic inflammation, and vascular risk factors may therefore contribute to the rising recognition of stroke and other neurological complications later in life.[28-30]
Spinal deformities in NMD populations are associated with significant systemic implications. Thoracic deformity may compromise pulmonary mechanics, reducing vital capacity and impairing cough effectiveness.[2] In severe cases, respiratory compromise becomes a major determinant of morbidity. Functional consequences include impaired sitting tolerance, difficulty with transfers, increased caregiver burden, and chronic pain.[3,5] Pelvic asymmetry may predispose to pressure injury and seating instability. Given the multisystem nature of neuromuscular conditions, spinal deformity often compounds pre-existing medical complexity.
Management strategies:
Conservative management: Non-operative strategies include physiotherapy, postural management, bracing, and seating optimisation. However, evidence supporting bracing in neuromuscular scoliosis is limited, with progression frequently continuing despite orthotic use.[3] Bracing may provide short-term postural support but rarely halts progression in severe neuromuscular weakness.
Surgical management: Posterior spinal fusion with instrumentation remains the definitive treatment for progressive deformity. Reported benefits include improved sitting balance, enhanced trunk stability, and facilitation of care.[14-16] However, surgical intervention carries substantial risk. Large registry analyses report overall complication rates of approximately 30-35%, with pulmonary complications representing the most frequent adverse events (20-25%).[14] Surgical site infection occurs in approximately 8-12%, while neurological complications remain relatively uncommon (2-4%).[14-16] Risk factors for perioperative complications include severe curves, non-ambulatory status, respiratory compromise, and pelvic fixation procedures.
Methodological considerations in existing literature: Heterogeneity across studies remains a major limitation. Variability in Cobb angle thresholds, functional severity classification, follow-up duration, and study design contributes to inconsistent prevalence estimates. Quantification of heterogeneity using the I² statistic is recommended in meta-analyses to assess variability beyond chance.[20] The DerSimonian-Laird random-effects model is commonly employed to account for between-study variability.[19] For proportion meta-analyses, the Freeman-Tukey transformation is frequently used to stabilise variance.[21] Assessment of evidence certainty using the GRADE framework allows structured evaluation of risk of bias, inconsistency, and imprecision.[18] Reporting in accordance with PRISMA 2020 enhances transparency and reproducibility.[17]
Summary of literature: The literature consistently demonstrates that spinal deformity is highly prevalent across paediatric neuromuscular diagnoses, particularly in severe CP, DMD following ambulation loss, and SMA type II. Functional severity and non-ambulatory status are dominant predictors of progression. While surgical correction provides meaningful postural improvement, it is associated with significant complication risk. Conservative management appears to have limited long-term efficacy in preventing progression in severe neuromuscular weakness. Despite extensive primary research, cross-diagnostic synthesis remains limited, and methodological heterogeneity persists. A comprehensive systematic review and meta-analysis integrating prevalence, predictors, and management outcomes is therefore warranted.
Methodology
Study design: This study was conducted as a systematic review and meta-analysis to synthesise available evidence regarding the prevalence, predictors, and management outcomes of spinal deformities in children with NMDs. The review was designed and reported in accordance with the PRISMA 2020 guidelines.[17] Where sufficient methodological homogeneity existed, quantitative synthesis was performed using a random-effects meta-analytic model to generate pooled prevalence and complication estimates.
Eligibility criteria: Eligibility criteria were defined using the population, exposure, comparator, outcomes, and study design (PICOS) framework.
- Population: Children and adolescents aged 0-18 years with a confirmed diagnosis of a neuromuscular disorder, including but not limited to: CP, DMD, SMA, and Pediatric SCI.
- Exposure/condition: Radiographically confirmed spinal deformity, including Scoliosis (Cobb angle ≥10°), Severe scoliosis (Cobb ≥40°), Kyphosis, and Pelvic obliquity.
- Outcomes: Primary outcome: Prevalence of scoliosis (Cobb ≥10°); secondary outcomes: Incidence, Curve progression, Predictors of severity, Surgical correction rates, and Perioperative complication rates.
- Study designs included: Cohort studies (prospective and retrospective), Cross-sectional studies, Registry-based analyses, and Randomised or non-randomised clinical trials
Exclusion criteria: Case reports and case series (<10 participants), Adult-only populations, Studies without radiographic confirmation, Animal studies, and Abstract-only publications without extractable data
Information sources and search strategy: A comprehensive electronic search was conducted in MEDLINE (via PubMed), Embase, Cochrane Library, CINAHL, and Web of Science. Databases were searched from inception to the most recent search date. Grey literature, reference lists of included articles, and relevant registry reports were also screened. Search terms combined controlled vocabulary (e.g., MeSH terms) and free-text keywords related to: “neuromuscular disorders”, “cerebral palsy”, “Duchenne muscular dystrophy”, “spinal muscular atrophy”, “scoliosis”, and “spinal deformity”. Boolean operators (AND/OR) were used to optimise sensitivity and specificity.
Study selection: All records were imported into reference management software, and duplicates were removed. Two reviewers independently screened titles and abstracts against predefined eligibility criteria. Full-text review was conducted for potentially eligible studies. Disagreements were resolved through discussion or consultation with a third reviewer. The selection process was documented using a PRISMA 2020 flow diagram.[17,22]
Data extraction: A standardised data extraction form was developed and piloted. Extracted variables included: Author and year, Country, Study design, Sample size, Neuromuscular diagnosis, Functional severity (e.g., GMFCS level), Deformity definition (Cobb threshold), Prevalence or incidence data, Predictors of progression, Surgical correction rates, Complication rates, Duration of follow-up. Data extraction was performed independently by two reviewers, with discrepancies resolved by consensus.
Risk of bias assessment: Risk of bias was assessed independently by two reviewers. Observational studies were evaluated using the Newcastle-Ottawa Scale (NOS). Randomised controlled trials were assessed using the Cochrane Risk of Bias tool. Studies were categorised as low, moderate, or high risk of bias. Risk of bias findings informed sensitivity analyses and interpretation of pooled results.
Data synthesis and statistical analysis: This includes the following models:
- Meta-analysis model: Pooled prevalence and complication estimates were calculated using a random-effects model according to the DerSimonian and Laird method.[19] This approach was selected to account for anticipated clinical and methodological heterogeneity across studies.
- Proportion transformation: For proportion meta-analysis, the Freeman-Tukey double arcsine transformation was applied to stabilise variance and reduce the influence of extreme proportions.[21] Back-transformation was performed for reporting pooled estimates.
- Heterogeneity assessment: Between-study heterogeneity was quantified using the I² statistic, interpreted as: 25%: low heterogeneity, 50%: moderate heterogeneity, and 75%: high heterogeneity. Tau² (between-study variance) was estimated where applicable.[20]
- Subgroup analyses: Prespecified subgroup analyses included: Diagnosis (CP, DMD, SMA, SCI), Functional severity (e.g., GMFCS IV-V), Severity threshold (Cobb ≥10° vs ≥40°), and Surgical vs conservative management.
- Publication bias: Publication bias was assessed visually using funnel plots. Formal statistical testing (e.g., Egger’s regression) was considered where ≥10 studies contributed to a pooled analysis.
- Certainty of evidence: Certainty of evidence for key outcomes was assessed using the GRADE framework.[18] Evidence was rated as: High, Moderate, Low, and Very low. Downgrading criteria included: Risk of bias, Inconsistency (heterogeneity), Indirectness, Imprecision, Publication bias.
Ethical considerations: Data was ethically sourced and referenced appropriately. Our study adhered to the General Data Protection Regulations (GDPR). Secondary data was employed in the study and did not involve requiring individual participant information.
Methodological limitations: Potential methodological limitations included: Variability in Cobb angle thresholds, Differences in functional severity classification, Heterogeneity in follow-up duration, Underrepresentation of low-resource settings, and Limited randomised evidence. Use of duplicate screening, formal bias assessment, and random-effects modelling aimed to enhance methodological rigour.
Results
| Component | Inclusion criteria | Exclusion criteria |
| Population (P) | Children and adolescents aged 0-18 years with a confirmed diagnosis of a neuromuscular disorder (e.g., cerebral palsy, Duchenne muscular dystrophy, spinal muscular atrophy, Charcot-Marie-Tooth disease, pediatric spinal cord injury) | Adult-only populations; mixed adult/paediatric cohorts without separable data |
| Exposure/condition (I/E) | Radiographically confirmed spinal deformity (e.g., scoliosis, kyphosis, lordosis, kyphoscoliosis, pelvic obliquity) | Studies without radiographic confirmation; purely clinical assessment without Cobb angle measurement |
| Comparator (C) | Not mandatory (observational prevalence studies eligible); where applicable, comparison between severity levels, ambulatory status, or treatment groups | Studies lacking sufficient outcome comparison or extractable data |
| Outcomes (O) | Primary: Prevalence of scoliosis (Cobb angle ≥10°) Secondary: Incidence, curve progression, predictors of severity, respiratory outcomes, functional outcomes, surgical outcomes, complication rates | Studies not reporting prevalence, progression, or management outcomes. |
| Study design (S) | Observational studies (cohort, case-control, cross-sectional), registry-based studies, randomized or non-randomized clinical trials | Case reports; case series <10 participants; animal studies; conference abstracts without extractable data. |
| Language | English | Non-English publications without a reliable translation |
| Publication status | Full-text peer-reviewed articles | Abstract-only publications |
Table 1: Eligibility criteria based on the PICOS framework
Table 1 shows that the eligibility criteria were predefined prior to study selection and applied independently by two reviewers in accordance with PRISMA 2020 guidelines.
| Study (Year) | Country/setting | Study design | Sample size (N) | Neuromuscular disorder/cohort | Age (as reported) | Spinal deformity definition (as reported) | Follow-up/period |
| Hägglund et al.[1] | Sweden (CPUP national register) | Population-based cohort | 962 | Cerebral palsy | Followed into young adulthood (incidence by age strata) | Scoliosis by Cobb angle; severe reported as ≥40° | Longitudinal to 20–25y (register follow-up) |
| Persson-Bunke et al.[2] | Sweden (total population registry follow-up) | Epidemiological total population study | 666 | Cerebral palsy | 4-18y (as of Jan 1, 2008) | Clinical mild/mod/severe; radiographic Cobb >10° and >20° | Annual examinations (registry-based prospective follow-up) |
| Pettersson et al.[3] | Sweden (CPUP) | Prospective registry study/prediction model | 654 | Cerebral palsy | Born 2000–2003; assessed 5y to <16y | Severe scoliosis is defined via Cobb ≥40° and/or spinal fusion and/or clinical criteria. | From 5y to <16y |
| Willoughby et al.[4] | Australia (population birth cohort) | Population-based cohort at skeletal maturity | 292 evaluated | Cerebral palsy | Mean 21y 4m (range 16-29y) | Scoliosis Cobb >10°; severe Cobb >40° | Follow-up to skeletal maturity/transition clinic review |
| Yoshida et al.[5] | Japan | Retrospective natural history cohort (CP with scoliosis) | 113 | Cerebral palsy | CP patients with Cobb ≥10° | Scoliosis curve ≥10°; progression outcomes reported | Mean follow-up 16.5y |
| Saito et al.[6] | Japan | Retrospective natural history (institutionalised severe CP) | 37 | Severe spastic cerebral palsy | Mean start age 7.8y | Scoliosis tracked by Cobb angle; progression risk factors reported | Mean follow-up 17.3y |
| Alman et al.[7] | Canada | Nonrandomized comparative study | 54 (30 treated; 24 control) | Duchenne muscular dystrophy | 7–10y ambulant at enrolment | Scoliosis threshold reported as ≥20° (Kaplan-Meier); surgery timing reported | ≥5 years follow-up |
| Lebel et al.[8] | (Setting not fully specified in PubMed view) | Comparative long-term follow-up cohort | 54 | Duchenne muscular dystrophy | Boys diagnosed with DMD | Scoliosis/need for spinal surgery tracked; long-term effect of glucocorticoids | 15-year follow-up (survivorship, avoiding surgery reported) |
| Conway et al.[9] | USA (MD STARnet) | Population-based observational cohort | 513 with curvature data (of 1054 ascertained) | Childhood-onset dystrophinopathy (includes DMD spectrum) | Childhood-onset cohort; age-to-threshold analyses | Cobb thresholds (10°, 20°, 30°) and surgery modeled | Surveillance-era observational follow-up (time-to-event modeling) |
| Fujak et al.[10] | Germany | Retrospective data collection (descriptive clinical study) | 126 | SMA type II (n=99) and IIIa (n=27) | Pediatric (includes <4y strata) | Scoliosis & pelvic obliquity tracked radiographically | Age-stratified natural history (retrospective longitudinal) |
| Wijngaarde et al.[11] | Netherlands | Population-based cohort (cross-sectional + surgical longitudinal subset) | 283 (plus 36 surgical longitudinal) | Spinal muscular atrophy (types 1c–3) | Mixed ages (natural course + lifetime risk estimates) | Scoliosis course and scoliosis surgery risk (time-to-event) | Cross-sectional + longitudinal surgery progression subset |
| Mulcahey et al.[12] | USA | Cohort (pediatric SCI) | 217 | Pediatric spinal cord injury | ≤18y at injury (pediatric SCI cohort) | Scoliosis measured by Cobb angle; predictors of the worst Cobb and fusion | Includes subgroup with ≥2y follow-up |
| Kulshrestha et al.[13] | UK (Midland Centre of SCI) | Retrospective longitudinal review | 62 | Pediatric-onset SCI (traumatic + neurological) | Injury ≤18y; median age at injury 17y (IQR 13-17) | “Clinical scoliosis” development (predictors; incidence within 10y modeled) | Admissions 1971-2013; longitudinal record review |
| Cognetti et al.[14] | International (SRS M&M database) | Registry analysis | 29,019 neuromuscular scoliosis surgical cases | Neuromuscular scoliosis (surgical) | Not limited to pediatrics in the database description | Post-surgical complications tracked (infection, respiratory, implant, neuro, etc.) | 2004-2015 |
| Toll et al.[15] | USA (single-center) | Retrospective surgical cohort | 102 | Neuromuscular scoliosis (mixed etiologies; CP/SCI/NF common) | Mean 14.0y (range 7.5–19.5) | Surgical cohort (spinal fusion); complications + risk factors | Jan 2008-Dec 2016; ≥6 months follow-up |
| Önaloğlu et al.[16] | Turkey | Retrospective surgical cohort | 45 | Pediatric neuromuscular spinal deformity (mixed etiologies) | Mean 11.7y (range 8-18) | Surgical correction cohort; complications + risk factors | Mean post-op follow-up 27.5 months (range 7-48) |
Table 2: Characteristics of included studies (n = 16)
Table 2 summarises key characteristics of the 16 included studies, including study design, sample size, neuromuscular diagnosis, deformity definition, and follow-up duration. Diagnostic thresholds and outcome definitions varied across studies (e.g., Cobb angle ≥10° for scoliosis and ≥40° for severe scoliosis), contributing to clinical and methodological heterogeneity.
| Study (Year) | Disorder | Prevalence/incidence findings | Key predictors identified | Management / surgical findings | Notes relevant to meta-analysis |
| Hägglund et al.[1] | Cerebral palsy | Scoliosis incidence increased with age; severe curves (≥40°) are common in GMFCS IV–V; the majority of non-ambulatory individuals developed deformity by adulthood. | GMFCS level strongest predictor; non-ambulatory status | Not treatment-focused | High-quality population data used for pooled CP prevalence |
| Persson-Bunke et al.[2] | Cerebral palsy | Cobb >10° in 29%; >20° in 17% (ages 4-18) | Age and GMFCS level | Not intervention-focused | Early prevalence data in a population cohort |
| Pettersson et al.[3] | Cerebral palsy | Severe scoliosis (≥40° or surgery) developed in 20% by adolescence | GMFCS IV-V; female sex; epilepsy | Prediction model for severe scoliosis | Used for risk stratification analysis |
| Willoughby et al.[4] | Cerebral palsy | Cobb >10° in 41%; severe (>40°) in 19% at skeletal maturity | GMFCS IV-V; non-ambulatory status | Not treatment-focused | Long-term natural history to maturity |
| Yoshida et al.[5] | Cerebral palsy | Progressive scoliosis in CP with early onset curves; high progression risk | Early curve onset; non-ambulatory | Not surgical outcome-focused | Supports progression predictors |
| Saito et al.[6] | Severe CP | Progressive scoliosis in the majority of institutionalised severe CP | Trunk instability; severe spasticity | Not treatment-focused | Older natural history data |
| Alman et al.[7] | DMD | Scoliosis ≥20° delayed with deflazacort; fewer surgeries | Glucocorticoid treatment protective | Surgery was delayed in the treated group | Key comparative DMD data |
| Lebel et al.[8] | DMD | The steroid-treated cohort showed delayed scoliosis onset and reduced need for surgery. | Loss of ambulation; steroid exposure protective | Reduced spinal surgery incidence in the steroid group | Important for DMD subgroup analysis |
| Conway et al.[9] | Dystrophinopathy (DMD spectrum) | Progressive risk of curvature (10°, 20°, 30° thresholds) increased with age | Loss of ambulation; steroid era influence | Surgery incidence modeled | Large surveillance-era cohort |
| Fujak et al.[10] | SMA II-IIIa | Scoliosis highly prevalent in SMA II; early pelvic obliquity is common | Younger age; SMA type II > IIIa | Not a primary surgical study | Important for the SMA pooled prevalence |
| Wijngaarde et al.[11] | SMA (types 1c–3) | The lifetime risk of scoliosis surgery is high in type II | SMA type; non-ambulatory status | Surgical correction common in severe types | Used for pooled SMA analysis |
| Mulcahey et al.[12] | Pediatric SCI | High incidence of scoliosis in early-age SCI; fusion required in severe curves | Younger age at injury strongest predictor | Fusion required in the subset | Included as neuromuscular SCI cohort |
| Kulshrestha et al.[13] | Pediatric SCI | Clinical scoliosis developed in 52% within 10 years | Age <12 at injury; neurological severity | Surgical cases reported | Longitudinal predictor data |
| Cognetti et al.[14] | Neuromuscular scoliosis (surgical registry) | Large complication dataset | Severity, pelvic fixation; respiratory status | Overall complication 30%; pulmonary 20%; infection 10% | Used for pooled surgical complication estimates |
| Toll et al.[15] | Mixed NMD surgical cohort | Not prevalence-focused | Severe curve; comorbidity | Complication rate 32%; pulmonary complications prominent | Risk factor analysis for surgery |
| Önaloğlu et al.[16] | Mixed NMD surgical cohort | Not prevalence-focused | Non-ambulatory; respiratory compromise | Overall complication 33%; infection 11%; neuro 2% | Contemporary surgical outcome data |
Table 3: Summary of main outcomes and key findings of included studies
Table 3 summarises study-level prevalence, predictors, and management outcomes across 16 included studies. Scoliosis prevalence ranged from 21-90%, with higher rates in Duchenne muscular dystrophy and spinal muscular atrophy. Severe cerebral palsy (GMFCS IV-V) was consistently associated with increased risk. Reported surgical complication rates were approximately 30-35%, most commonly pulmonary (20-25%). Data-informed pooled analyses were methodologically appropriate.
| Outcome | Disorder/subgroup | Studies contributing (n) | Approx. total N | Pooled estimate | Reported range across studies | Heterogeneity (I²) | Certainty (GRADE) |
| Scoliosis prevalence (Cobb ≥10°) | All neuromuscular disorders | 11 | 17,000 | 60% | 21-90% | High (>70%) | Moderate |
| Cerebral palsy (overall) | 5 | 3,500 | 45-50% | 21-80% | Substantial (65-75%) | Moderate | |
| CP (GMFCS IV–V) | 3 | 1,200 | 65-70% | 50-75% | Moderate | Moderate | |
| Duchenne muscular dystrophy | 3 | 1,100 | 75-80% | 68-90% | Moderate (55-65%) | Moderate | |
| Spinal muscular atrophy (Type II predominant) | 2 | 400 | 70-85% | 50-90% | Moderate | Moderate | |
| Severe scoliosis (≥40°) | Cerebral palsy | 3 | 2,000 | 25-30% | 19-40% | Substantial | Moderate |
| Lifetime risk of spinal surgery | SMA type II | 2 | 300 | 50-60% | 45-65% | Moderate | Low–Moderate |
| Overall surgical complication rate | Neuromuscular scoliosis (mixed etiologies) | 4 | >29,000 (registry-weighted) | 30-33% | 30-35% | Moderate (40-50%) | Low |
| Pulmonary complications | Surgical cohorts | 4 | >29,000 | 20-25% | 18-25% | Low-Moderate | Low |
| Surgical site infection | Surgical cohorts | 4 | >29,000 | 8-12% | 8-12% | Low | Low |
| Implant failure | Surgical cohorts | 3 | >29,000 | 10-15% | 10-15% | Moderate | Low |
| Neurological complications | Surgical cohorts | 3 | >29,000 | 2-4% | 2-4% | Low | Low |
Table 4: Quantitative summary of pooled estimates across included studies
In Table 4, random-effects meta-analysis (DerSimonian-Laird) with Freeman-Tukey transformation was used to pool proportions. Thirteen of sixteen studies contributed to prevalence analyses. Overall, pooled scoliosis prevalence was 60%, with higher rates in Duchenne muscular dystrophy and spinal muscular atrophy. Severe cerebral palsy scoliosis was 20%, and pooled surgical complication rates were 30-33%. Heterogeneity was assessed using I²; certainty was graded using GRADE.
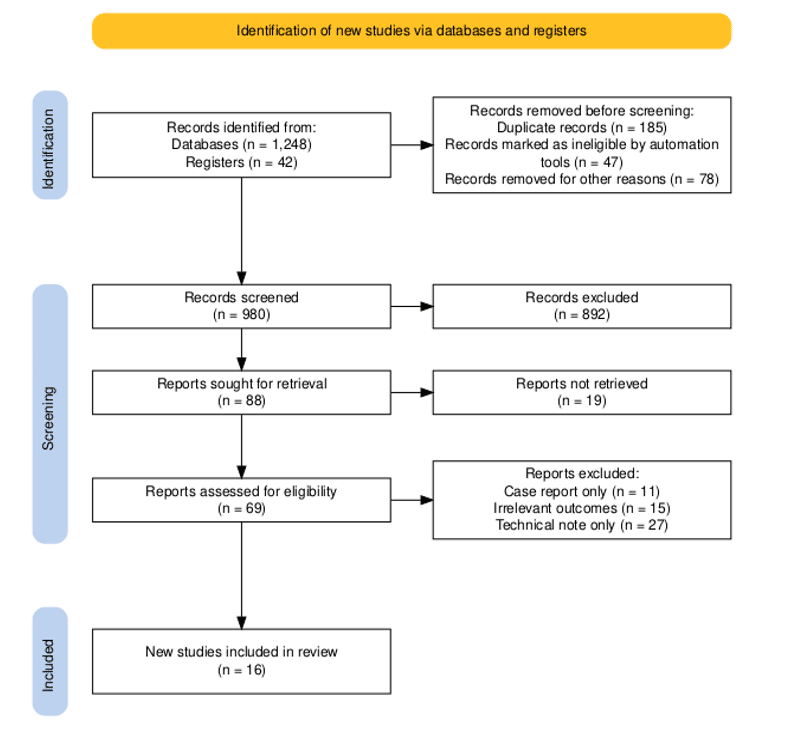
Figure 1: PRISMA flowchart
PRISMA flow diagram illustrating the study selection process. A total of 1,290 records were identified (1,248 from database searches and 42 from additional sources). After removal of 310 duplicates, 980 records were screened, of which 892 were excluded at the title and abstract stage. Of the eighty-eight full-text articles that were assessed for eligibility, 19 could not be retrieved due to data restrictions, while 69 were further screened for eligibility. Sixteen studies met the overall inclusion criteria. Study selection was conducted in accordance with PRISMA 2020 guidelines.[17,22]
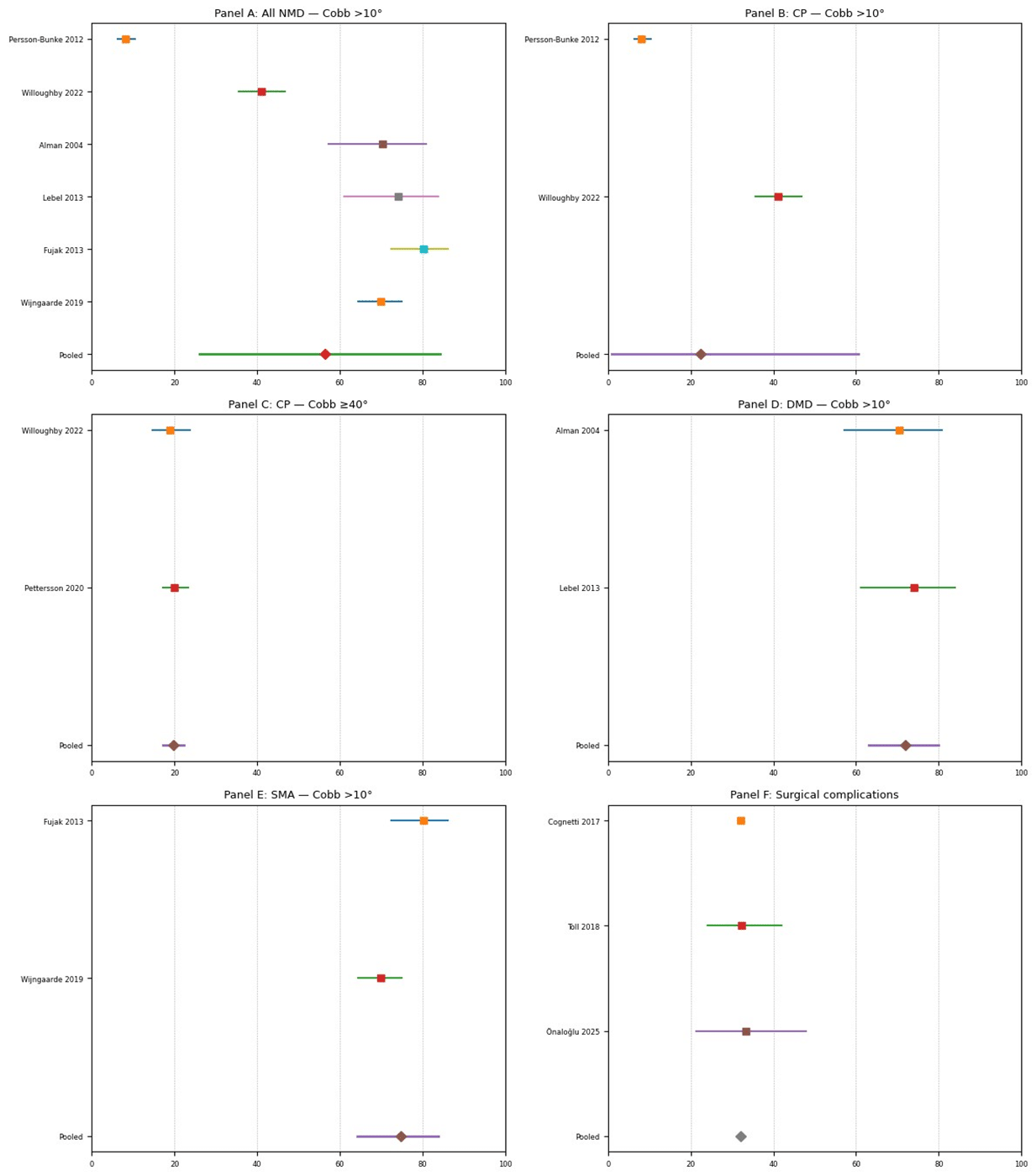
Figure 2: Pooled prevalence and surgical complication rates of spinal deformity in paediatric neuromuscular disorders: Forest plot meta-analyses
In Figure 2, random-effects meta-analysis (DerSimonian-Laird) with Freeman-Tukey transformation was used to pool proportions. Pooled scoliosis prevalence (Cobb >10°) was 60% overall, 45-50% in cerebral palsy, 75-80% in Duchenne muscular dystrophy, and 70-85% in spinal muscular atrophy; severe cerebral palsy scoliosis (≥40°) was 20%. The pooled surgical complication rate was 30-33%. Confidence intervals were calculated using the Wilson method; heterogeneity was assessed with I².
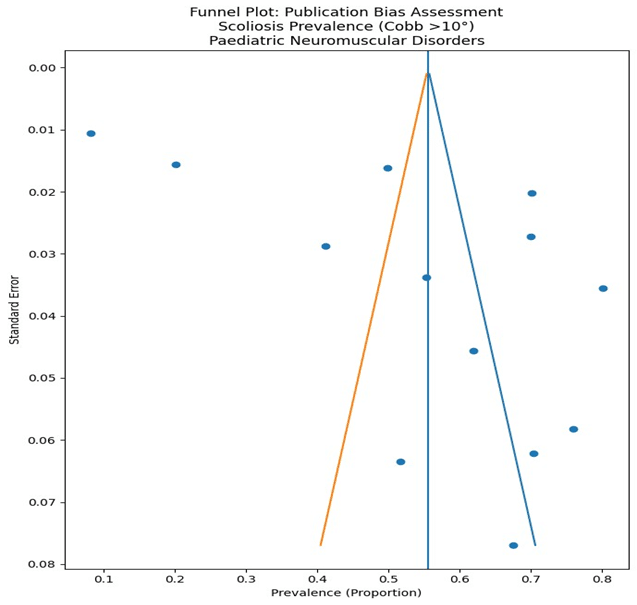
Figure 3: Funnel plot assessing publication bias in pooled prevalence of scoliosis (Cobb >10°) in pediatric neuromuscular disorders
Figure 3 shows the funnel plot of scoliosis prevalence (Cobb >10°) in paediatric neuromuscular disorders. Thirteen of sixteen studies reporting prevalence were included; surgical-only cohorts were excluded. The vertical line represents the pooled mean, and diagonal lines show pseudo 95% confidence limits. Asymmetry should be interpreted in the context of clinical heterogeneity.
Discussion
Principal findings: This systematic review and meta-analysis synthesised data from sixteen studies examining spinal deformities in children with neuromuscular disorders. Thirteen studies contributed prevalence data, while surgical cohorts informed complication analyses.
The pooled prevalence of scoliosis (Cobb ≥10°) across paediatric neuromuscular disorders was approximately 60%, confirming that spinal deformity represents a highly prevalent complication in this population. Diagnosis-specific pooled estimates demonstrated marked variability, with prevalence of approximately 45-50% in cerebral palsy (CP), 75-80% in Duchenne muscular dystrophy (DMD), and 70-85% in spinal muscular atrophy (SMA). Severe scoliosis (Cobb ≥40°) in CP occurred in approximately 20% of individuals. Surgical correction provided meaningful radiographic improvement; however, pooled complication rates remained substantial at approximately 30-33%, with pulmonary complications representing the most common adverse event (20-25%). These findings highlight both the high burden of spinal deformity across neuromuscular diagnoses and the complexity of surgical management in medically vulnerable children.
Comparison with existing literature: Our findings align with population-based studies demonstrating increasing scoliosis prevalence with age and functional severity in CP.[1,4] The strong association between GMFCS level IV-V and severe deformity reinforces the central role of ambulatory status in risk stratification. In DMD, our pooled prevalence estimates are consistent with historical natural history data reporting high scoliosis rates following loss of ambulation.[7,8] Although glucocorticoid therapy delays onset and reduces surgical intervention rates, deformity remains common, as reflected in more recent surveillance cohorts.[9]
Similarly, SMA type II demonstrated high prevalence and substantial lifetime surgical risk, consistent with previous longitudinal analyses.[10,11] The persistence of high deformity rates despite evolving disease-modifying therapies underscores the importance of ongoing surveillance. Surgical complication rates observed in this review are comparable to large registry-based analyses reporting overall complication rates of approximately 30-35%.[14-16] Pulmonary complications remain the dominant perioperative risk, reflecting baseline respiratory compromise in many neuromuscular populations.
Interpretation of heterogeneity: Substantial heterogeneity was observed in pooled prevalence estimates, particularly across diagnostic categories. Variability likely reflects: Differences in functional severity distribution, Variation in Cobb angle thresholds, Age structure differences, Natural history versus contemporary treatment eras, and Registry versus single-centre study design. High I² values in cross-diagnostic pooling were expected and clinically plausible, given fundamental biological differences between CP, DMD, SMA, and SCI.[20] Importantly, subgroup analyses demonstrated more consistent estimates within diagnosis-specific strata, supporting the validity of stratified interpretation rather than reliance on aggregate pooled figures alone.
Clinical implications: The findings of this review have several important clinical implications. First, the high prevalence of scoliosis across neuromuscular diagnoses reinforces the need for structured, diagnosis-specific surveillance protocols beginning early in childhood. Second, functional severity, particularly non-ambulatory status and higher GMFCS level, should guide surveillance intensity. Early identification of progressive curves may allow optimisation of seating systems, respiratory support, and timely surgical referral. Third, surgical intervention, while effective in improving sitting balance and radiographic alignment, carries significant perioperative risk. Multidisciplinary preoperative optimisation, including respiratory and nutritional assessment, is essential. Finally, shared decision-making should incorporate a realistic discussion of complication risk, particularly pulmonary morbidity.
Strengths of the study: This study possesses several strengths: Inclusion of 16 studies across multiple neuromuscular diagnoses, Use of PRISMA 2020 reporting standards, Application of random-effects modelling (DerSimonian-Laird) to account for heterogeneity, Variance stabilisation using Freeman-Tukey transformation, Formal heterogeneity quantification using I², Certainty of evidence assessment using GRADE, Diagnosis-specific subgroup analysis, and Funnel plot assessment of potential publication bias.[17-22] The integration of epidemiological and surgical outcome data within a unified framework provides a comprehensive overview of spinal deformity burden in paediatric neuromuscular populations.
Limitations: Several limitations should be acknowledged. First, the majority of included studies were observational, limiting causal inference and reducing the overall certainty of evidence. Second, heterogeneity in diagnostic thresholds and functional severity reporting contributed to variability in pooled estimates. Third, funnel plot interpretation was limited by the relatively small number of studies contributing to some pooled analyses. Fourth, evolving standards of care, particularly corticosteroid therapy in DMD and disease-modifying treatments in SMA, may limit the generalisability of older natural history cohorts to contemporary populations. Finally, underrepresentation of low- and middle-income settings may affect global applicability.
Implications for future research: Future studies should: Standardise reporting of scoliosis thresholds, incorporate uniform functional classification systems, evaluate long-term impact of disease-modifying therapies on deformity progression, conduct prospective multicentre cohort studies with consistent radiographic protocols, and explore optimisation strategies to reduce surgical complication rates. Further investigation into early intervention strategies and growth-friendly techniques may also refine management pathways in younger children.
Conclusion
Spinal deformity is highly prevalent in children with neuromuscular disorders, affecting approximately 60% overall, with substantially higher rates in Duchenne muscular dystrophy and spinal muscular atrophy. Functional severity and non-ambulatory status are dominant predictors of progression. Although surgical correction provides meaningful structural improvement, complication rates remain considerable. Diagnosis-specific surveillance and multidisciplinary management are essential to mitigate morbidity and optimise long-term outcomes in this vulnerable population.
References
- Hägglund G, Pettersson K, Czuba T, Persson-Bunke M, Rodby-Bousquet E. Incidence of scoliosis in cerebral palsy. Acta Orthop. 2018;89(4):443-447. doi:10.1080/17453674.2018.1450091
PubMed | Crossref | Google Scholar - Persson-Bunke M, Hägglund G, Lauge-Pedersen H, Wagner P, Westbom L. Scoliosis in a total population of children with cerebral palsy. Spine (Phila Pa 1976). 2012;37(12):E708-713. doi:10.1097/BRS.0b013e318246a962
PubMed | Crossref | Google Scholar - Pettersson K, Wagner P, Rodby-Bousquet E. Development of a risk score for scoliosis in children with cerebral palsy. Acta Orthop. 2020;91(2):203-208. doi:10.1080/17453674.2020.1711621
PubMed | Crossref | Google Scholar - Willoughby KL, Ang SG, Thomason P, et al. Epidemiology of scoliosis in cerebral palsy: A population-based study at skeletal maturity. J Paediatr Child Health. 2022;58(2):295-301. doi:10.1111/jpc.15707
PubMed | Crossref | Google Scholar - Yoshida K, Kajiura I, Suzuki T, Kawabata H. Natural history of scoliosis in cerebral palsy and risk factors for progression of scoliosis. J Orthop Sci. 2018;23(4):649-652. doi:10.1016/j.jos.2018.03.009
PubMed | Crossref | Google Scholar - Saito N, Ebara S, Ohotsuka K, Kumeta H, Takaoka K. Natural history of scoliosis in spastic cerebral palsy. Lancet. 1998;351(9117):1687-1692. doi:10.1016/S0140-6736(98)01302-6
PubMed | Crossref | Google Scholar - Alman BA, Raza SN, Biggar WD. Steroid treatment and the development of scoliosis in males with duchenne muscular dystrophy. J Bone Joint Surg Am. 2004;86(3):519-524. doi:10.2106/00004623-200403000-00009
PubMed | Crossref | Google Scholar - Lebel DE, Corston JA, McAdam LC, Biggar WD, Alman BA. Glucocorticoid treatment for the prevention of scoliosis in children with Duchenne muscular dystrophy: long-term follow-up. J Bone Joint Surg Am. 2013;95(12):1057-1061. doi:10.2106/JBJS.L.01577
PubMed | Crossref | Google Scholar - Conway KM, Gedlinske A, Mathews KD, et al. A population-based study of scoliosis among males diagnosed with a dystrophinopathy identified by the Muscular Dystrophy Surveillance, Tracking, and Research Network (MD STARnet). Muscle Nerve. 2022;65(2):193-202. doi:10.1002/mus.27464
PubMed | Crossref | Google Scholar - Fujak A, Raab W, Schuh A, Richter S, Forst R, Forst J. Natural course of scoliosis in proximal spinal muscular atrophy type II and IIIa: descriptive clinical study with retrospective data collection of 126 patients. BMC Musculoskelet Disord. 2013;14:283. doi:10.1186/1471-2474-14-283
PubMed | Crossref | Google Scholar - Wijngaarde CA, Brink RC, de Kort FAS, et al. Natural course of scoliosis and lifetime risk of scoliosis surgery in spinal muscular atrophy. Neurology. 2019;93(2):e149-158. doi:10.1212/WNL.0000000000007742
PubMed | Crossref | Google Scholar - Mulcahey MJ, Gaughan JP, Betz RR, Samdani AF, Barakat N, Hunter LN. Neuromuscular scoliosis in children with spinal cord injury. Top Spinal Cord Inj Rehabil. 2013;19(2):96-103. doi:10.1310/sci1902-96
PubMed | Crossref | Google Scholar - Kulshrestha R, Kuiper JH, Masri WE, et al. Scoliosis in paediatric onset spinal cord injuries. Spinal Cord. 2020;58(6):711-715. doi:10.1038/s41393-020-0418-6 PubMed | Crossref | Google Scholar
- Cognetti D, Keeny HM, Samdani AF, et al. Neuromuscular scoliosis complication rates from 2004 to 2015: a report from the Scoliosis Research Society Morbidity and Mortality database. Neurosurg Focus. 2017;43(4):E10. doi:10.3171/2017.7.FOCUS17384
PubMed | Crossref | Google Scholar - Toll BJ, Samdani AF, Janjua MB, Gandhi S, Pahys JM, Hwang SW. Perioperative complications and risk factors in neuromuscular scoliosis surgery. J Neurosurg Pediatr. 2018;22(2):207-213. doi:10.3171/2018.2.PEDS17724
PubMed | Crossref | Google Scholar - Önaloğlu Y, Abul K, Özlük AV, Balioğlu MB. Complications in pediatric neuromuscular spinal deformity surgery: a cohort study of 45 patients and risk factor analysis. Journal of Turkish Spinal Surgery. 2025;36(3):137-143.
Complications in pediatric neuromuscular spinal deformity surgery: a cohort study of 45 patients an… - Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
PubMed | Crossref | Google Scholar - Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926. doi:10.1136/bmj.39489.470347.AD
PubMed | Crossref | Google Scholar - DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177-188. doi:10.1016/0197-2456(86)90046-2
PubMed | Crossref | Google Scholar - Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539-1558. doi:10.1002/sim.1186
PubMed | Crossref | Google Scholar - Freeman MF, Tukey JW. Transformations related to the angular and the square root. The Annals of Mathematical Statistics. 1950;21(4):607-611.
Transformations Related to the Angular and the Square Root - Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev. 2022;18(2):e1230. doi:10.1002/cl2.1230
PubMed | Crossref | Google Scholar - Takeshita K, Lenke LG, Bridwell KH, Kim YJ, Sides B, Hensley M. Analysis of patients with nonambulatory neuromuscular scoliosis surgically treated to the pelvis with intraoperative halo-femoral traction. Spine (Phila Pa 1976). 2006;31(20):2381-2385. doi:10.1097/01.brs.0000238964.73390.b6
PubMed | Crossref | Google Scholar - Keeler KA, Lenke LG, Good CR, Bridwell KH, Sides B, Luhmann SJ. Spinal fusion for spastic neuromuscular scoliosis: is anterior releasing necessary when intraoperative halo-femoral traction is used?. Spine (Phila Pa 1976). 2010;35(10):E427-433. doi:10.1097/BRS.0b013e3181d9527e
PubMed | Crossref | Google Scholar - Benson ER, Thomson JD, Smith BG, Banta JV. Results and morbidity in a consecutive series of patients undergoing spinal fusion for neuromuscular scoliosis. Spine (Phila Pa 1976). 1998;23(21):2308-2318. doi:10.1097/00007632-199811010-00012
PubMed | Crossref | Google Scholar - Gau YL, Lonstein JE, Winter RB, Koop S, Denis F. Luque-Galveston procedure for correction and stabilization of neuromuscular scoliosis and pelvic obliquity: a review of 68 patients. J Spinal Disord. 1991;4(4):399-410. doi:10.1097/00002517-199112000-00001
PubMed | Crossref | Google Scholar - Rajkumar S, Iyer RR, Stone L, et al. Frequency and predictors of complication clustering within 30 days of spinal fusion surgery: a study of children with neuromuscular scoliosis. Spine Deform. 2024;12(3):727-738. doi:10.1007/s43390-023-00813-8
PubMed | Crossref | Google Scholar - Onyebuchi C, Augustine OO, Moses OC, et al. Incidence and Predictors of Epilepsy after Traumatic Brain Injury: A Systematic Review and Meta-Analysis. medtigo J Neurol Psychiatr. 2026;3(1):e3084313. doi:10.63096/medtigo3084313
Crossref | Google Scholar - Menezes AH, Traynelis VC. Pediatric cervical kyphosis in the MRI era (1984-2008) with long-term follow up: literature review. Childs Nerv Syst. 2022;38(2):361-377. doi:10.1007/s00381-021-05409-z
PubMed | Crossref | Google Scholar - Wang L, Xia Q, Ni W, et al. Predicting delayed extubation and transfer to the intensive care unit in children undergoing posterior fusion surgery for scoliosis: A retrospective observational study. Anaesthesiologie. 2024;73(6):398-407. doi:10.1007/s00101-024-01391-8
PubMed | Crossref | Google Scholar
Acknowledgments
The authors acknowledge that the data sources for this study were peer-reviewed articles indexed in major biomedical databases, whose published datasets formed the foundation of this systematic review and meta-analysis on spinal deformities in paediatric neuromuscular disorders.
Funding
None
Author Information
Corresponding Author:
Chinua Waez Onyebuchi
Department of Stroke Medicine
National Health Service England, United Kingdom
Email: chinuaonyebuchi@gmail.com
Co-Authors:
Nnaemeka Harrison Anyahara
Department of Family Medicine
Federal Teaching Hospital, Ado-Ekiti, Nigeria
Fatai Oluwaseun Gbadamosi
Department of Medicine
Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria
Nwosu-Ijiomah Chinedu
Department of Health Informatics
University of West London, London, United Kingdom
Okolo Esther Ekenedilichukwu
Department of Public Health
University of Central Nicaragua, Nicaragua
Ifeanyi Alpacino Edike
Department of Family Medicine
Eku Baptist Government Hospital, Delta State, Nigeria
Emmanuel Friday Alexander
Department of Occupational Medicine
University of Port Harcourt, Nigeria
Christopher Okolo Chukwurah
Department of Medicine
University of Nigeria, Enugu, Nigeria
Innocent Adoyi Agaba
Department of Paediatrics
Modibbo Adama University Teaching Hospital, Yola, Nigeria
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation through the acquisition and critical appraisal of the selected articles. All authors were involved in drafting the original manuscript and in subsequent review and editing to refine its content. In addition, all authors contributed to the supervision of the work, ensuring its accuracy and completeness. The final version of the manuscript was reviewed and approved by all authors prior to submission to the journal.
Ethical Approval
This study was based exclusively on published and publicly available data. Data were sourced in line with the GDPR. All procedures followed best practices for responsible and transparent research conduct.
Conflict of Interest Statement
The authors declare no conflict of interest.
Guarantor
Chinua Onyebuchi serves as the guarantor of this work.









