Author Affiliations
Abstract
Background: Wegener granulomatosis (WG), a type of antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, is an autoimmune disease that damages blood vessels. Treatment with rituximab differs from cyclophosphamide in that it affects the cluster of differentiation 20 (CD20) on B cells and alters deoxyribonucleic acid (DNA) due to replication, which divides cells such as neutrophils and lymphocytes.
Methods: We systematically searched electronic databases, including PubMed, Scopus, Elsevier, and Cochrane Library, spanning from each database’s inception to August 10, 2024. We will calculate pooled effect estimates with 95% confidence intervals using the random-effects model, adhering to the guidelines specified in the preferred reporting items for systematic reviews and meta-analyses (PRISMA).
Results: This meta-analysis includes four studies, totaling 504 patients, evaluating the safety and effectiveness of rituximab compared to cyclophosphamide in patients with ANCA-associated vasculitis, with two studies focusing on patients with renal diseases.
Conclusion: This meta-analysis indicates that rituximab has a higher mortality rate. On the other hand, cyclophosphamide shows a higher vasculitis damage score. According to the Birmingham vasculitis activity score (BVAS) for WG, there is no significant difference in preference between the two drugs. We recommend that patients with ANCA use a different investigation to confirm a positive result, balancing efficacy and minimizing harmful consequences.
Keywords
Wegener granulomatosis, Antineutrophil cytoplasmic antibody associated vasculitis, Birmingham vasculitis activity score, Deoxyribonucleic acid, Rituximab, Cyclophosphamide.
Introduction
ANCA-associated vasculitis is a disease that can cause damage to various organs in patients, potentially leading to life-threatening complications. WG, an autoimmune disorder, causes inflammation of blood vessels due to vasculitis, which in turn damages tissue in various organs. Symptoms of WG include joint pain, skin rashes, and nosebleeds. We can decrease side effects and complications by two primary treatments for this condition: rituximab and cyclophosphamide. Rituximab, a monoclonal antibody medication, has demonstrated promising results in inducing remission, especially in patients with relapsing diseases. Additionally, it can target the CD20 protein on the surface of B cells. On the other hand, cyclophosphamide is a cytotoxic drug, a potent immunosuppressive drug that has long been used to treat autoimmune diseases such as ANCA-associated vasculitis. It works by alkylating DNA due to replication, dividing cells like neutrophils and lymphocytes.
Methodology
Search strategy: Following guidelines specified in the PRISMA, we systematically searched electronic databases, including PubMed, Scopus, Cochrane Library, Elsevier, spanning from each database’s inception to August 8, 2024. Our search strategy involved a combination of relevant keywords and standardized index terms tailored to the comparison between the effectiveness of rituximab and cyclophosphamide in patients with ANCA-associated vasculitis.
Type of studies: This review and meta-analysis will look at studies that followed patients over time or looked back at records. These studies need to have included 10 or more people with ANCA. The research must compare what happened to patients who took rituximab versus those who took cyclophosphamide. All patients in the studies should have ANCA-associated vasculitis.
Types of intervention: The focus of our study is the use of a placebo in patients with a condition called ANCA-associated vasculitis. We are looking at how rituximab and cyclophosphamide work in patients with a specific type of condition known as WG, in a study.[1,2] we have divided patients into two groups, each receiving a different drug along with a placebo.
Eligible studies must focus on clinically relevant outcomes regarding the efficacy and safety of rituximab and cyclophosphamide. These results can include complications such as infections, leukopenia, and hypersensitivity reactions.
Exclusion criteria: Studies will be excluded if they do not involve human subjects, are not published in English, have a sample size smaller than ten in either rituximab or cyclophosphamide, present overlapping data from the same institution, or have a median follow-up duration of less than 6 months. Studies with inadequate reporting of outcomes, such as failing to specify the exact number of events and the total patient-years for comparisons between the drugs approaches, will be excluding. Furthermore, studies that do not report outcomes specifically for the effect of rituximab and cyclophosphamide will be excluded. Reviews, case reports, conference abstracts, letters to the editor, and editorials will also be excluded.
Quality assessment: Before conducting the statistical analysis, we assessed the risk of bias and the quality of the included studies. For the two eligible randomized trials (RCTs), we used Version 2 of the Cochrane risk-of-bias tool for randomized trials (RoB 2) and conducted the assessment using Cochrane review manager web.
Results
The PRISMA flowchart provides a detailed description of the selection process for the systematic review and meta-analysis on the use of rituximab and cyclophosphamide in Patient with ANCA associated vasculitis. Initial research generated a total of 48 records from different databases:11 records from PubMed, 24 records from Scopus, 2 records from Cochrane, and 11 additional records identified from Elsevier. After removing duplicates, 41 records remained for further evaluation. The selection process started with these 41 records, of which 18 were excluded to focus on other interventions or to be irrelevant to the research question. This exception left 23 records for title projection; after that, there were other files that were excluded due to a lack of characteristics and had no results. Five full-text articles were evaluated for their eligibility, and 4 studies met the inclusion criteria. These 4 studies were later included in the systematic review and quantitative synthesis (meta-analysis).
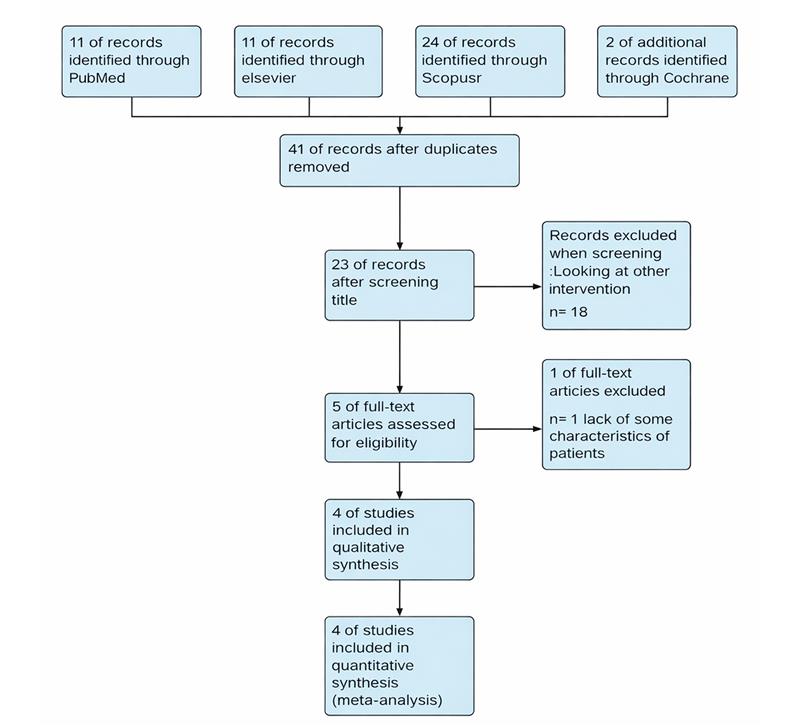
Figure 1: The flowsheet of search results according to PRISMA guidelines
| Sample size | Study design | Population | Data collection | Outcome | |
| Stone JH et al. (USA and
Netherland, 2010) |
197 patients | Randomized controlled trial (RCT) | Patient with severe antineutrophil
cytoplasmic antibody (ANCA)-associated Vasculitis |
Rituximab (375mg per square meter of body- surface area per week for 4 weeks)
compared to cyclophosphamide (2mg per kilogram of body weight per day) |
Treatment with rituximab was not inferior. To daily treatment cyclophosphamide for the induction of remission in severe vasculitis is associated with ANCA and may be superior in recurrent diseases. |
| Jones RB et al. (Multiple centers of
Europe, 2014) |
44 patients | RCT | Patient with renal
problem and severe antineutrophil cytoplasmic antibody (ANCA) |
Rituximab compared to cyclophosphamide | At 24 month, the result rates are compounded death, terminal renal failure and relapse did not differ between groups, the return of B lymphocytes was regarding the return. |
| Geetha D et al.
(North America, Europe, and Asia 2014) |
102 patients | RCT | Patient with renal
disease and severe antineutrophil cytoplasmic antibody (ANCA) |
Rituximab follow up placebo compared to cyclophosphamide and azathioprine | The primary endpoint of this post-hoc analysis achieved full remission. Secondary endpoints where remission extends to 12 and 18 months, the slope of growth in 18-month eGFR, rates of disease recurrence and rates of serious adverse events. |
| Miloslavsky EM et al. (USA, Europe, 2013) | 107 patients | RCT | Patient with Severe Antineutrophil Cytoplasmic Antibody- Associated Vasculitis | Rituximab compared to cyclophosphamide and azathioprine | Current treatment regimens are effective in controlling AAV, but in about a quarter of patients, active disease persists or recurs within the first six months despite treatment.PR3-ANCA positivity is a risk factor for serious disease recurrence or progression. ANCA titers and B-cell detectability are poor predictors of disease relapse and remission during the first six months. |
Table 1: Summary of the four studies compared with rituximab vs cyclophosphamide
Table 1 presents data from four randomized controlled trials conducted in different countries, focusing on patients with severe ANCA-associated vasculitis. Additions, two studies were focusing on patients with renal diseases.[1,2] The study involved 197 patients divided into two groups: 99 patients received rituximab 375 mg per square meter of body area per 4 weeks, and 98 patients received cyclophosphamide 2 mg per kilogram of body weight per day.[3] The secondary study was focusing on patients with renal diseases, divided into two groups: 33 patients receiving rituximab and 11 patients receiving cyclophosphamide.[1]
This study had nine dead patients, ninety-six patients’ deaths from the group that received rituximab, vs 3 patients’ deaths from the group that received cyclophosphamide. The third study found that patients with renal disease were investigated for renal estimated glomerular filtration rate (eGFR) at 12 and 18 months. This study included patients who had received rituximab before the placebo versus patients who had received cyclophosphamide before the placebo, azathioprine, and showed adverse effects for each study. The last study included 197 patients with 3 deaths, 24 remissions, and 170 completed the research; this study investigated ANCA and risk factors for serious diseases.
Separately in superscripts. Place the actual footnote at the bottom of the column in which it was cited. Do not put in the rituximab group, and the study was aging of patients 54 in the cyclophosphamide group, 56 in the rituximab group. ANCA was the most important in 98% of the rituximab group vs. 96% in cyclophosphamide. We can determine type of ANCA by enzyme-linked immunosorbent assay (ELISA), the types of ANCA are proteinase 3 (PR3), myeloperoxidase (MPO), 3 studies from 4 studies show are in patient taken rituximab have percent of ANCA more than patient taken cyclophosphamide.[2,3]
| Group | Age Cyclophosphamide | Age Rituximab | Gender Rituximab | Gender Cyclophosphamide | BVAS/WG Rituximab | BVAS/WG Cyclophosphamide | ANCA (%) Rituximab | ANCA (%) Cyclophosphamide |
| Stone JH, 2010 | 51.5 ± 14.1 | 54.0 ± 16.8 | Male 46, Female 54 | Male 54, Female 46 | 8.5 ± 3.2 | 8.2 ± 3.2 | 98% | 96% |
| Jones RB, 2014 | NA | NA | 33 | 11 | NA | NA | 63% | 54% |
| Geetha D, 2014 | 54 | 56 | Male 47, Female 53 | Male 57, Female 43 | 8.7 (2.74) | 8.7 (3.51) | 59% | 55% |
| Miloslavsky EM, 2013 | NA | NA | 99 | 98 | 8 (1.6) | 8 (1.4) | 86% | 86% |
Table 2: Patient’s characteristics
Table 2 compares patient characteristics from four studies. The study included 99 patients in the rituximab group (male 46 patients and female 54 patients), 98 patients in the cyclophosphamide group (male 54 patients and female 46 patients). In Geetha D, patients receiving rituximab males 47 and females 53 vs males 57 and females 43 in the cyclophosphamide group and this study had a BVAS for WG. The age group was in both studies, 51.5±14.1 in the cyclophosphamide group, 54.0±16.8.[2,3]
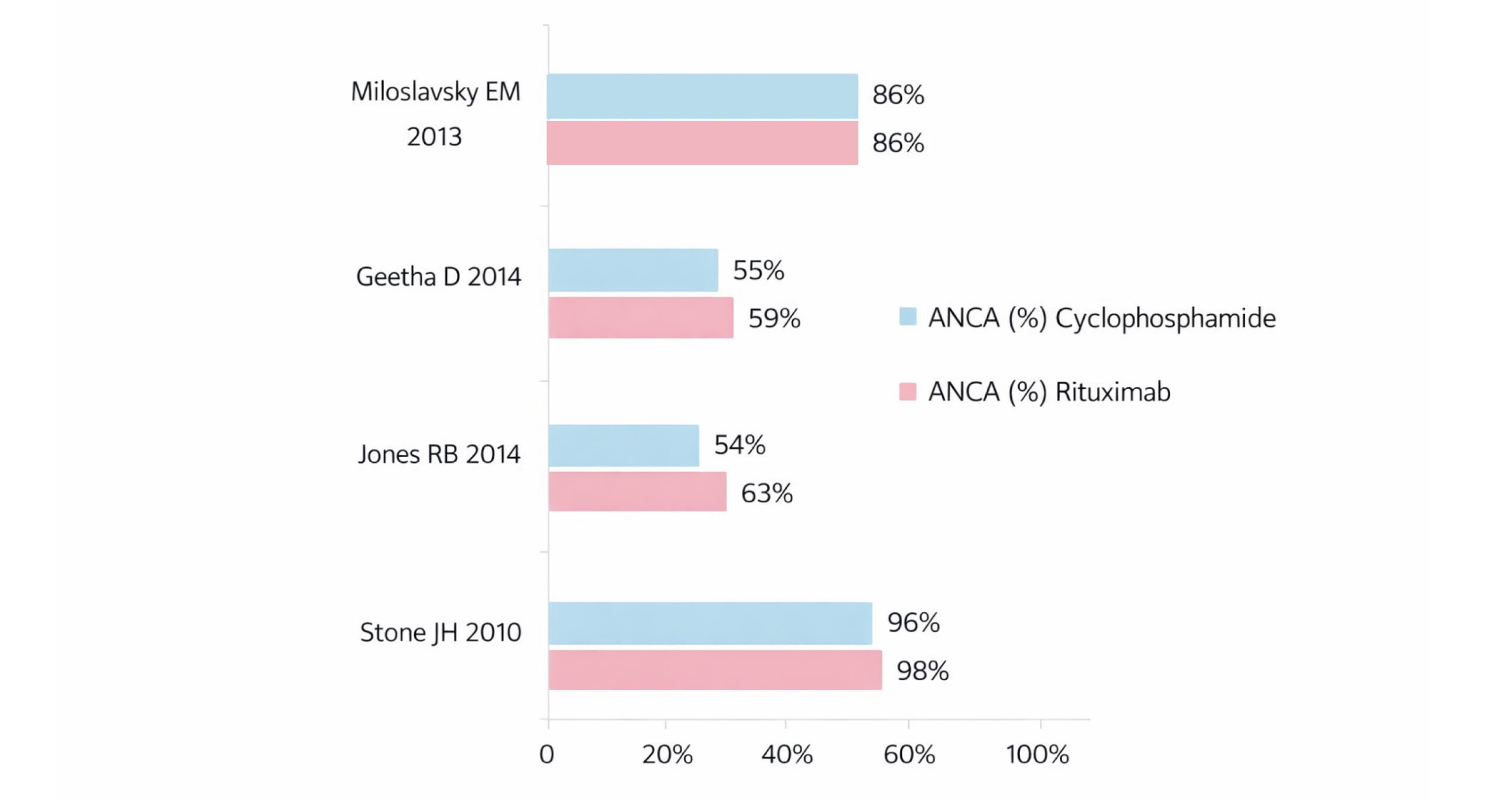
Figure 2: Percent of ANCA in patients in studies
This image shows the percentages of ANCA for each study and for the two drugs; ANCA is different from one study to another. In this study, 98% of patients in the rituximab group and 96% in the cyclophosphamide group had positive outcomes, which is higher than in other studies. Notably, this study had lower ANCA levels when compared to previous studies (rituximab 59% vs. cyclophosphamide 55%). Both drugs showed an equal efficacy rate of 86%, 3 out of 4 studies demonstrated that patients receiving rituximab had a high percentage of ANCA positivity when compared to those receiving cyclophosphamide.[2-4]
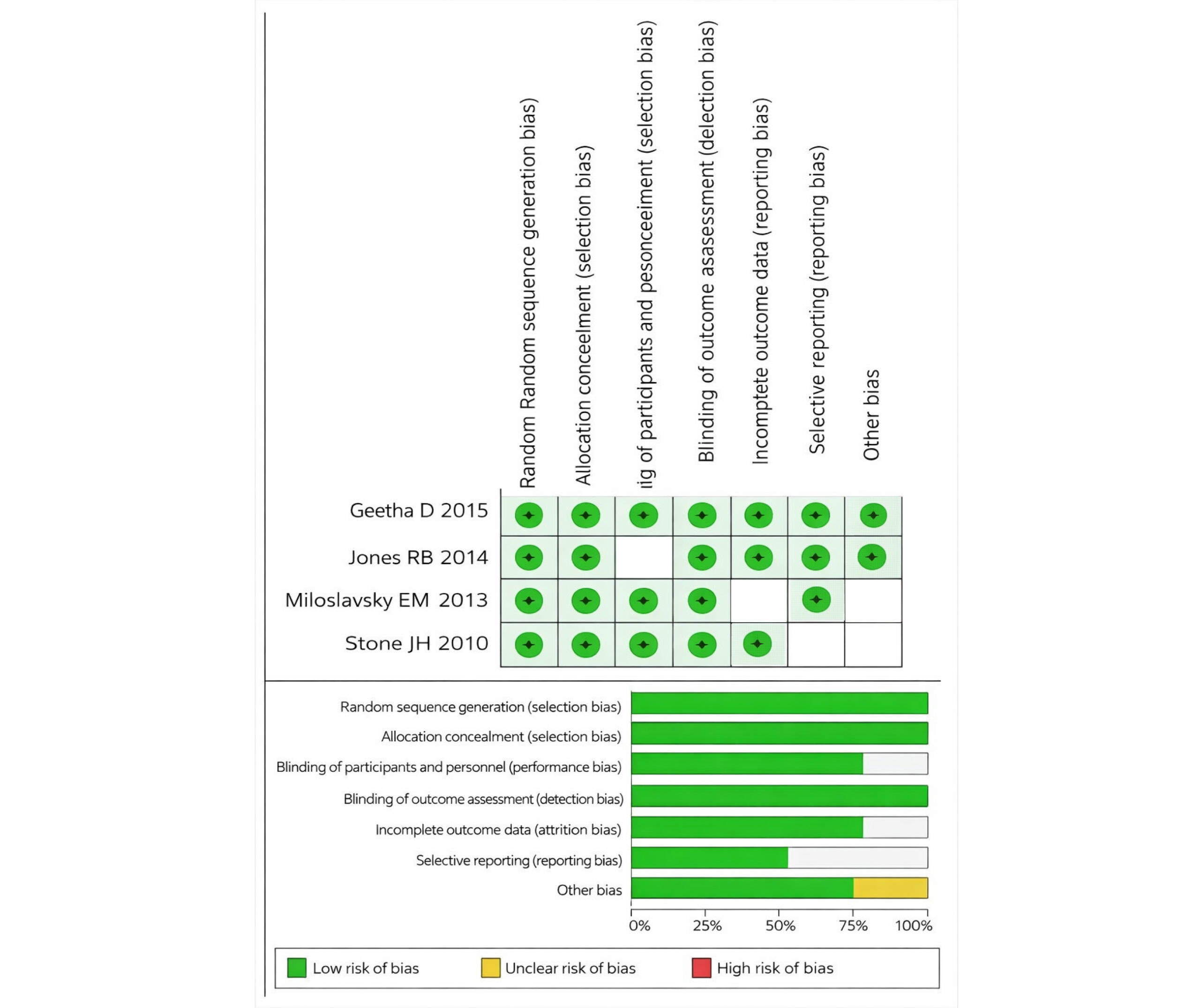
Figure 3: Risk of bias assessment using RoB-2 tool
The risk of bias assessment for the systematic review and meta-analysis on the use of rituximab compared to cyclophosphamide in patients with ANCA-associated vasculitis includes evaluations of four studies.[1-4] The assessment indicates that all studies have a minimal risk of bias in most categories, such as selection, performance, detection, attrition, and reporting biases. Key areas like random sequence generation, allocation concealment, blinding, and data reporting were consistently rated as insignificant risk, reflecting robust methodologies.
This consistent rating of minimal risk across these key domains suggests that the individual studies conducted with robust methodologies minimize the potential for biased outcomes. In the overall assessment across the studies, more of the categories continue to reflect an insignificant risk of bias. Random sequence generation, allocation concealment, incomplete outcome data, selective reporting, and other biases were all reported with 100% minimal risk.
However, the concerns were noted in the categories of performance bias and detection bias, where approximately 33% of the assessments indicated an unclear risk of bias. This ambiguity arises from the blinding of participants and personnel and the blinding of outcome assessment, which are crucial for preventing performance and detection biases.
Overall, the studies demonstrate a strong methodological quality with minimal risk of bias, lending credibility to the findings of the systematic review and meta-analysis. Despite the minor concerns in performance and detection biases, the predominance of low-risk ratings supports the validity and reliability of the conclusions drawn regarding the effectiveness of the rituximab compared cyclophosphamide in patient with ANCA associated vasculitis.
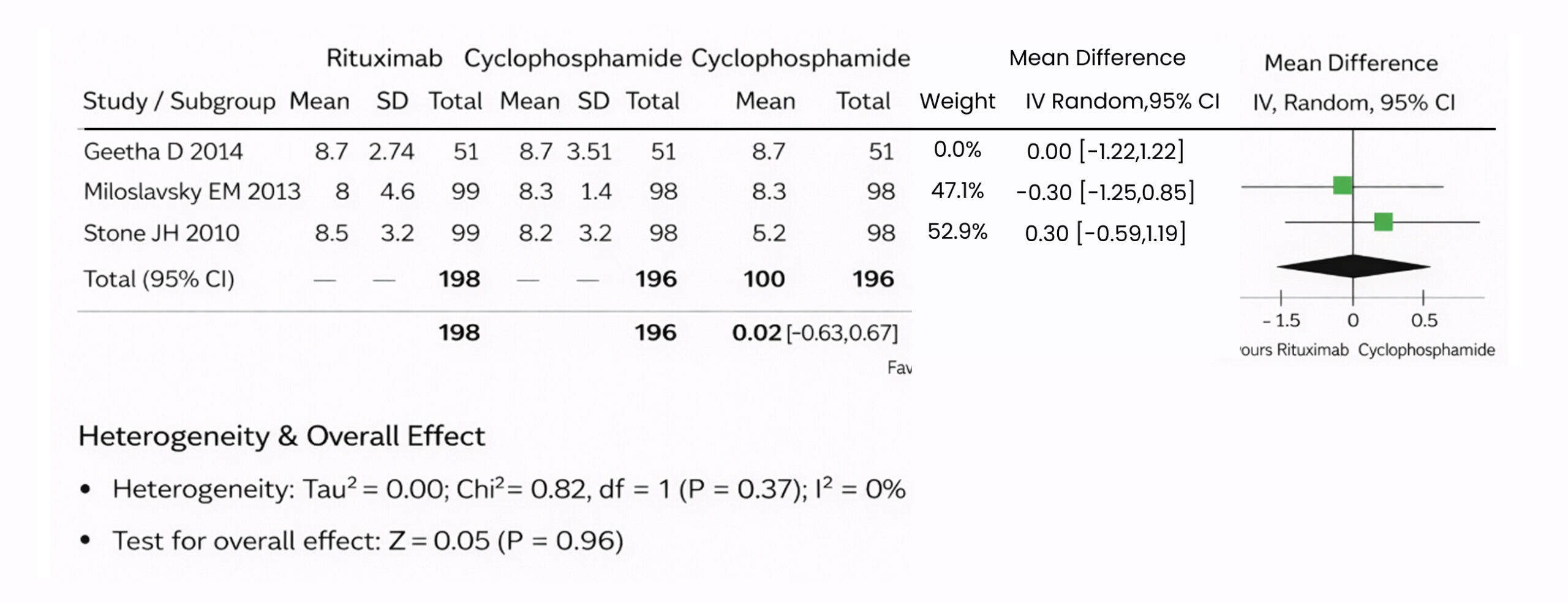
Figure 4: The forest plot of the BVAS for WG of the three included studies using a random effect model
This image is a forest plot from a meta-analysis comparing the effectiveness between two drugs, rituximab vs cyclophosphamide, in patients with ANCA-associated vasculitis. It includes data from three studies.[2-4] For each study, the mean, standard deviation (SD), and total number of participants in both rituximab and cyclophosphamide groups are listed. The mean difference between these groups, along with the 95% confidence interval (CI), is provided for each study.
The inverse variance method intravenous (IV) and a random-effect model were used to calculate these differences. The weight assigned to each study in the meta-analysis is based on the sample size and variance.
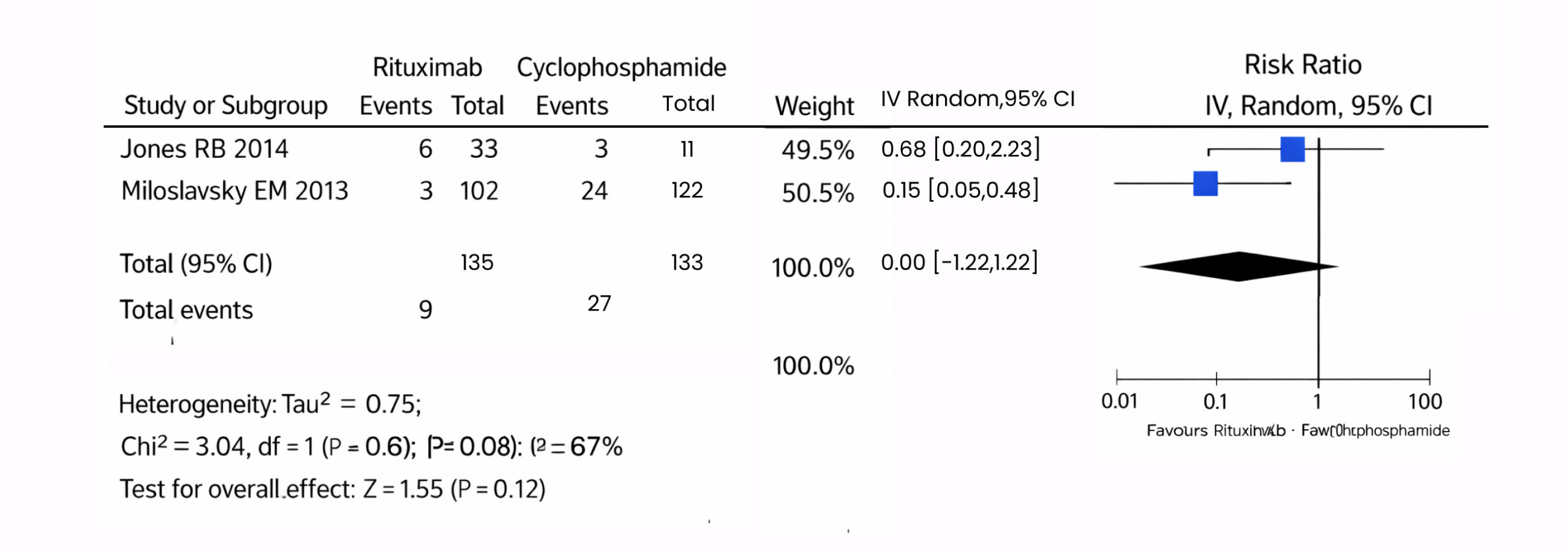
Figure 5: The forest plot of uncontrollable and death patients in the 2 included studies using a random-effects model
This image is a forest plot of a meta-analysis comparing uncontrolled and each drug because of the higher risk between the two drugs, rituximab vs cyclophosphamide, in a patient with ANCA-associated vasculitis, including data from two studies.[1,4] For each study, the events of death or uncontrollable and the total number of Participants in the rituximab and cyclophosphamide groups are listed.
Whether the differences in results across studies are due to chance, with a p value of 0.96 and I² of 0%, indicating no heterogeneity. Hence, to ensure that the overall evidence was not dependent on a single study, we conducted a sensitivity analysis in multiple scenarios, excluding a study in each scenario, to make sure the effect size was not dependent on any individual event studies. The overall mean difference between the rituximab and the cyclophosphamide don’t favour a pooled effect size 0.02, 95% Cl (-0.63 to 0.67), p = 0.96. Pooled studies were not homogenous (Chi² = 0.82, p = 0.37, I2 = 0%).
The hazard ratio between these groups, as well as the 95% CI, is provided for each study. The inverse variance method IV and the random effects model were used.to account for these differences. The weight assigned to each study in the meta-analysis is based on sample size and variance. The Chi-square (Chi²) test for heterogeneity assesses whether differences in results between studies are due to chance, with a p-value of 0.12 and an I² of 67% indicating a lack of heterogeneity. The overall risk ratio between rituximab and cyclophosphamide is favourable, rituximab RR 0.31, 95% CI (0.07 to 1.36), p = 0.07, p = 0.12. Pooled studies were homogeneous (Chi2=3.04, p =0.08, I2 =67%).
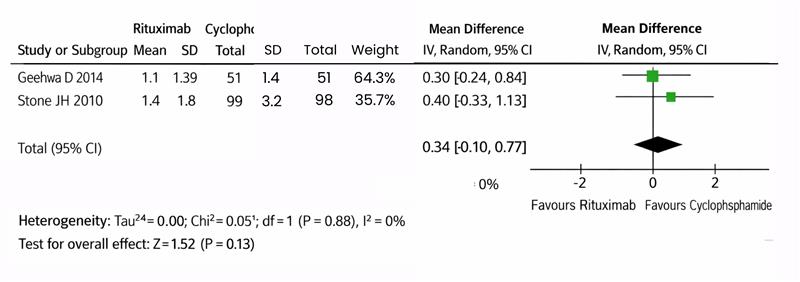
Figure 6: The forest plot of the vasculitis damage index of the 2 included studies using a random-effects model
This image is a forest plot from a meta-analysis comparing the effectiveness of the vasculitis damage index between two drugs (rituximab vs cyclophosphamide) in patients with ANCA-associated vasculitis, which includes data from two studies.[5-8] For each study, the mean, SD, and total number of participants in both the rituximab and cyclophosphamide groups are listed. The mean difference between these groups, along with the 95% CI is provided for each study. The inverse variance method IV and a random effect model were used to calculate these differences. The weight assigned to each study in the meta-analysis is based on the sample size and variance. The Chi² test for heterogeneity assesses whether the differences in results across studies are due to chance, with a p-value of 0.13 and I² of 0% indicating no heterogeneity. The overall mean difference between the rituximab and the cyclophosphamide favoured the cyclophosphamide pooled effect size 0.34, 95% Cl (-0.1 to 0.77), p = 0.13. Pooled studies were homogenous (Chi2 = 0.05, p = 0.83, I2 = 0%).
Discussion
In our meta-analysis, we included all studies that compared two drugs (rituximab vs cyclophosphamide) in patients with ANCA-associated vasculitis and renal disease. We focus on patients with WG, and we evaluated the two drugs by the vasculitis damage index and uncontrollable deaths. In addition, side effect for each drug. Also, we considered studies that reported patient remission and relapse. Our analysis included critical investigations in patients with renal diseases such as ANCA and leukocytes, number B cell and sensitivity surface for B cell because given rituximab is mechanism on B cell surface. Therefore, the doctors must be careful to ensure investigations before giving any drugs to patients with ANCA-associated vasculitis. Our study found that cyclophosphamide has a more significant effect on blood vessels with vasculitis damage compared to rituximab, as supported by two studies. These studies indicated that cyclophosphamide scores higher on the vasculitis damage index. Conversely, rituximab was associated with a higher risk of uncontrolled disease. Using the BVAS for WG, two studies reported effect sizes of 0.02, with one favouring cyclophosphamide and the other favouring rituximab. This highlights the complexity of choosing the most appropriate treatment and suggests that the decision should be tailored to individual patient profiles regarding Safety and effectiveness. In 2 studies that focused on patients with renal studies, we definitively identified ANCA. In addition, two types are PR3, MPO, rituximab has higher ANCA in some studies, but it was associated with a risk of mortality with 9 patients reported dead in two studies. On the other hand, cyclophosphamide was found to be uncontrolled in 24 patients after using it and has side effects like fatigue, SOB.
The heterogeneity of treatment in patients with Wegener’s granulomatosis included in assorted studies makes it challenging to draw definitive conclusions about the efficacy of rituximab and cyclophosphamide across the spectrum of these complex cases. Future research focusing on comparing the effectiveness of rituximab and cyclophosphamide would provide more targeted insights into the benefits and limitations of the drugs in these distinct scenarios. It is important to note that this meta-analysis only included two studies specifically investigating renal diseases, end-stage renal diseases (ESRD), and serum albumin. This limited sample size restricts the statistical power of the analysis and underscores the urgent need for additional research in this area to confirm these preliminary findings and establish more definitive conclusions.
Conclusion
Our systematic review and meta-analysis found a clinically significant reduction in side effects and efficacy of rituximab use and cyclophosphamide in patients with ANCA-associated vasculitis. These results are particularly important. During the process of ANCA-associated vasculitis with rituximab and cyclophosphamide, we looked at our meta-analysis to determine which medication is most effective and safe for patients with ANCA and kidney disease. Our research has shown that rituximab is associated with higher mortality rates and poorer disease control compared to cyclophosphamide. On the other hand, cyclophosphamide has significant side effects, especially for patients with kidney disease, and cyclophosphamide can lead to more serious vasculitis. So, we will pay attention to choosing the right treatment for a patient suffering from kidney disease to balance effectiveness and minimize harmful consequences.
References
- Jones RB, Furuta S, Cohen Tervaert JW, et al. Rituximab versus cyclophosphamide in ANCA-associated renal vasculitis: 2-year results of a randomised trial. Ann Rheum Dis. 2015;74(6):1178-1182. doi:10.1136/annrheumdis-2014-206404 PubMed | Crossref | Google Scholar
- Geetha D, Specks U, Stone JH, et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis with renal involvement. J Am Soc Nephrol. 2015;26(4):976-985. doi:10.1681/ASN.2014010046 PubMed | Crossref | Google Scholar
- Stone JH, Merkel PA, Spiera R, et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N Engl J Med. 2010;363(3):221-232. doi:10.1056/NEJMoa0909905 PubMed | Crossref | Google Scholar
- Miloslavsky EM, Specks U, Merkel PA, et al. Clinical outcomes of remission induction therapy for severe antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum. 2013;65(9):2441-2449. doi:10.1002/art.38044 PubMed | Crossref | Google Scholar
- Fauci AS. Wegener’s granulomatosis: prospective clinical and therapeutic experience with 85 patients for 21 years. Ann Intern Med. 1983;98(1):76-85. doi:10.7326/0003-4819-98-1-76 PubMed | Crossref | Google Scholar
- Finkielman JD, Lee AS, Hummel AM, et al. ANCA are detectable in nearly all patients with active severe Wegener’s granulomatosis. Am J Med. 2007;120(7):643.e9-14. doi:10.1016/j.amjmed.2006.08.016 PubMed | Crossref | Google Scholar
- Nachman PH, Hogan SL, Jennette JC, Falk RJ. Treatment response and relapse in antineutrophil cytoplasmic autoantibody-associated microscopic polyangiitis and glomerulonephritis. J Am Soc Nephrol. 1996;7(1):33-39. doi:10.1681/ASN.V7133 PubMed | Crossref | Google Scholar
- Walton EW. Giant-cell granuloma of the respiratory tract (Wegener’s granulomatosis). BMJ. 1958;2(5091):265-270. doi:10.1136/bmj.2.5091.265 PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
Not reported
Author Information
Corresponding Author:
Momen Shabib
Department of Medicine
Yarmouk University, Jordan
Email: moumenaldabobe@yahoo.com
Co-Authors:
Osamah Al-Ramahi, Mohammad Al-Sharab, Mohammad Al-Oran, Mohammad Al-khrissat
Department of Medicine
Yarmouk University, Jordan
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
This meta-analysis is based on previously published studies. Hence, ethical approval or patient consent is not required.
Conflict of Interest Statement
Not reported
Guarantor
None
DOI
Cite this Article
Momen S, Osamah AR, Mohammad AS, Mohammad AO, Khrissat MA. Rituximab Versus Cyclophosphamide in the Management of Wegener’s Granulomatosis: A Systematic Review and Meta-Analysis. medtigo J Pharmacol. 2024;1(1):e3061116. doi:10.63096/medtigo3061116 Crossref









