Author Affiliations
Abstract
Introduction: Guillain-Barré syndrome (GBS) is a sudden-onset autoimmune disease of the nerves, which leads to paralysis and muscle weakness, impaired sensory feeling, and even death. Despite the common knowledge about the racial disparities in access to healthcare and overall health outcomes, the exact role of these disparities in the prognosis of GBS remains unclear. This study examines one-year outcomes (paralysis, numbness, muscle weakness, and mortality) in Black/African American and White/Caucasian patients matched for comorbidities.
Methodology: A retrospective cohort study of propensity-matched Black/African American and White/Caucasian patients with GBS. The matching was done by comorbidities such as diabetes, obesity, pregnancy-associated disorders, respiratory disorders, and circulatory diseases. Outcome differences between the racial groups were evaluated using risk assessment, Kaplan-Meier survival analysis, and t-tests.
Results: The Black-White patient rates of paralysis (p = 0.163), numbness (p = 0.849), and muscle weakness (p = 0.333) did not reveal statistically significant differences between Black and White patients. Black and white patients did not differ significantly in terms of mortality rates (p = 0.694). Kaplan-Meier analysis also did not demonstrate any difference in time-to-event in any outcome.
Conclusion: When major comorbidities were adjusted, race did not seem to impair one-year GBS. These results affirm the importance of equal treatment and prompt intervention. Future research needs to focus on healthcare access, disparities in treatment, and recovery in the long term, with the aim of gaining a clearer picture of potential disparities.
Keywords
Guillain-Barré syndrome, Autoimmune, Racial disparities, African American, Caucasians.
Introduction
GBS is a group of clinical disorders that present as acute inflammatory polyradiculoneuropathy, which causes weakness and impaired reflexes.[1,2] Males are about 1.5 times more affected than females and have an overall incidence rate of 1–2 per 100,000 population per year worldwide.[3] GBS is a disease of peripheral neuropathy, where the body’s immune system erroneously targets and damages the peripheral nervous system.[3] The progression of the disease can be rapid, with deterioration occurring over hours to weeks. Although the symptoms of GBS might be minor, in some cases, the condition might deteriorate and result in the potentially fatal symptoms of circulatory instability, autonomic dysregulation, and respiratory dysfunction. These serious consequences have been found to make a good prognosis in a majority of the GBS patients, where the results vary between residual weakness and a complete neurological recovery.[4]
The pathophysiological mechanisms of GBS typically occur in two critical stages: the initiation phase, driven by an immunological trigger, followed by immune-mediated damage to axonal structures and/or myelin.[5] GBS is conventionally classified into two distinct subtypes: acute inflammatory demyelinating polyradiculoneuropathy (AIDP) and acute motor axonal neuropathy (AMAN). The most frequent precipitating event that leads to GBS is infection, with nearly 75 percent of them happening before GBS.[6] The prevalence varies across regions, influenced by differing exposures to infectious pathogens. Campylobacter jejuni and Mycoplasma pneumoniae are the most common bacterial causes of GBS.[7] The most common viruses that cause GBS are cytomegalovirus (CMV), Zika virus, and dengue virus.[8]
GBS is typically a treatable condition, with most patients ultimately fully recovering. Approximately 80 percent of patients regain independent ambulation, and over half of them return to their pre-illness baseline within one year.[9] However, up to 10% of patients may require prolonged mechanical ventilation for several months, and more than 10% are left with significant disability. Previous reports also claim that diabetes mellitus (DM) is an independent poor prognostic factor for the ability to walk unaided 3 months after symptom onset.[10]
The Erasmus GBS outcome score (EGOS) is a prognostic model based on age, diarrhea, and GBS disability score at two weeks post-hospital admission. This measurement scale offers an effective method for predicting a patient’s likelihood of walking independently within six months after contracting the infection.[11] Mortality rates can range from 20% in patients who become ventilator-dependent to 3-7% across all GBS cases, with death typically caused by sepsis, acute respiratory distress syndrome (ARDS), pulmonary embolism, or cardiac arrest. Prognostic factors such as age above 60 years, rapid course of the illness (the onset of weakness occurs within 7 days of the onset of the symptoms to admission into the hospital), significant weakness at the time of admission, need to undergo mechanical ventilation, history of diarrheal disease, and extreme neuropathy during electrophysiologic evaluations are unfavorable.
The ability to predict which patients with GBS will develop respiratory insufficiency or have a poor prognosis has been a long-held desire worldwide, as it would enable physicians to take the necessary precautions and provide additional treatment for the patients most at risk.[12] Inadequate access to rehabilitation services further contributes to disparities in recovery outcomes.
Methodology
Data source and study design: We conducted a retrospective cohort study using the TriNetX Research Network, which aggregates de-identified electronic health records (EHRs) from participating healthcare organizations. The platform was used for cohort identification and all embedded analyses; patient-level data were not exportable.
Cohort identification and index date: Patients with GBS were identified by ICD-10-CM G61.0. Race was categorized as Black/African American or White/Caucasian from structured EHR fields. The index date was the first recorded GBS diagnosis.
Missing data handling: We conducted complete-case analyses for matching variables. Records with missing age, sex, or race were excluded before propensity score estimation. Comorbidities were defined based on diagnosis codes within the 365 days preceding the index date; in this network, the absence of a code is considered equivalent to the absence of the corresponding condition. Multiple imputation was not feasible within the platform.
Propensity score matching (PSM): Propensity scores were estimated using logistic regression, which included age, sex, and comorbidity categories (diabetes, obesity, pregnancy-related conditions, respiratory disease, circulatory disease, and neurologic disorders). One-to-one nearest-neighbor matching without replacement used a caliper of 0.2 SD of the logit of the propensity score. Covariate balance was assessed using standardized mean differences (SMD), with SMD <0.10 prespecified as acceptable; post-match SMDs were <0.02 for all variables. After matching, each racial cohort consisted of 4,153 patients (pre-match counts: White, 29,999; Black, 4,379).
Outcome definitions and windows: Outcomes were identified by ICD-10-CM codes during follow-up: paralysis (G83), numbness/paresthesia (R20, R20.2), and muscle weakness (M62.81). Mortality was defined as a death status recorded in the EHR. The time-to-event observation window began 180 days after the index date and extended for 3,650 days (approximately 10 years). The primary analysis summarized one-year outcomes after the index date.
Statistical analysis: In the matched cohorts, we estimated absolute risk, risk difference, and risk ratio for each outcome. Time-to-event was evaluated with Kaplan–Meier methods and log-rank tests, and hazard ratios with 95% CIs were obtained from Cox proportional hazards models. For continuous measures, independent sample t-tests were used. A two-sided p<0.05 was considered statistically significant.
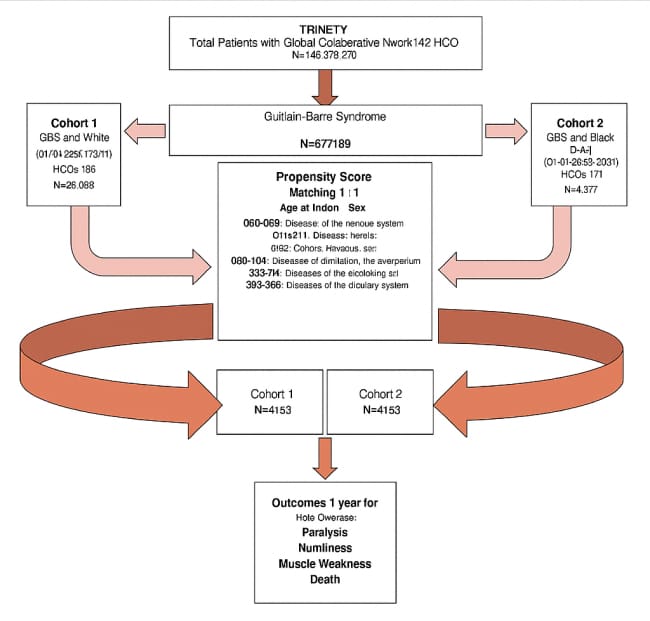
Figure 1: Cohort selection flow diagram
Results
Paralysis: There was no significant difference in the risk based on risk analysis (Risk difference (RD) = -0.004, p = 0.163). Relative risk (RR) = 0.787, Odds ratio (OR) = 0.784, and a very minimal change towards a decrease in risk by being White. There was no significant difference in survival according to Survival Analysis (Log-rank p = 0.105). The point estimate of hazard ratio (HR) is 0.755 (p = 0.907), indicating that survival to paralysis is not significantly different. The analysis of Mean differences revealed no significance in the mean time to paralysis (p = 1.000). These findings imply that the racial background has no significant impact on the propensity to paralysis development following GBS when accounting for comorbid conditions.
Numbness: The risk analysis produced no significant difference in the risk (RD = 0.001, p = 0.849). RR = 1.010 with OR = 1.012, which implies almost no difference in risk between groups. Log-rank testing of Survival Analysis, p = 0.603, indicated no significant difference in terms of survival. HR = 0.970 (p = 0.067), indicating that the time to numbness was comparable between the groups. The analysis of Mean Differences revealed there is no significant difference in Mean time to numbness (p = 0.560). Its implications are (i) there might be no difference in the burden of sensory impairments experienced by the two racially different groups after GBS.
Muscle weakness: The risk analysis did not indicate any difference in risk (RD = -0.006, p = 0.333). RR = 0.931, OR = 0.925, indicating a slight, non-significant decrease in risk among White patients. There was no significant difference in survival between the two groups, as determined by Survival Analysis (Log-rank p = 0.134). HR = 0.890 (p = 0.896), indicating it is not relevant to report on whether there is any difference in the progression to muscle weakness. The comparison of the mean difference indicated that there was no significant difference between the mean time to muscle weakness (p = 0.952). These outcomes indicate that muscle weakness following GBS in both racial groups persists and occurs at a specific percentage.
Mortality: The risk analysis revealed no significant impact on risk (RD = 0.002, p = 0.694). RR = 1.033, OR = 1.035, indicating no difference in mortality risk between the groups. The difference in survival was not statistically significant, as indicated by a p-value of 0.791 according to the survival analysis. HR = 0.978 (p = 0.367), so time to death suggests no significant difference. The results suggest that racial background does not affect survival in the case of GBS after matching the patients on major comorbid disorders.
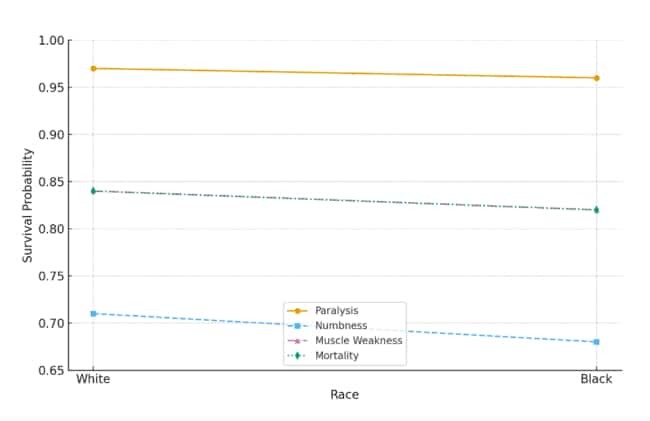
Figure 2: Kaplan-Meier survival probability by race and condition
Survival rates for paralysis, numbness, muscle weakness, and mortality by race
GBS Patients:
| Outcome | RD | RR | OR | Log rank test (LRT, p-value) | HR | Mean time to event (MTE, p-value) | Number of instances (NI, mean ± SD) | Median instances | Survival probability at the end |
| Paralysis | -0.004 (p = 0.163) | 0.787 | 0.784 | 0.105 | 0.755 (p = 0.907) | 1.000 | 4.25 ± 9.28 | 1 | White: 97.22%, Black: 96.12% |
| Numbness | 0.001 (p = 0.849) | 1.010 | 1.012 | 0.603 | 0.970 (p = 0.067) | 0.560 | White: 3.40 ± 5.31, Black: 3.63 ± 8.11 | 2 | White: 71.38%, Black: 68.92% |
| Muscle Weakness | -0.006 (p = 0.333) | 0.931 | 0.925 | 0.134 | 0.890 (p = 0.896) | 0.952 | White: 5.43 ± 11.14, Black: 5.39 ± 9.31 | 2 | White: 84.42%, Black: 82.51% |
| Mortality | 0.002 (p = 0.694) | 1.033 | 1.035 | 0.791 | 0.978 (p = 0.367) | — | — | — | White: 83.67%, Black: 82.47% |
Table 1: Outcome comparison between Black/African American and White/Caucasian
Discussion
This study examined one-year outcomes of GBS in propensity-matched Black/African American and White/Caucasian patients, focusing on paralysis, numbness, muscle weakness, and mortality. The results demonstrated no statistically significant differences between the two groups across all outcomes. While there were minor variations in risk estimates, survival probabilities, and hazard ratios, none reached statistical significance, suggesting that racial differences did not significantly impact clinical outcomes after accounting for comorbidities such as diabetes, obesity, respiratory disease, circulatory disease, and pregnancy-related conditions.
Paralysis: It is one of the most severe consequences of GBS and a key determinant of long-term disability.[13] In this study, the risk of paralysis was slightly higher in Black/African American patients (1.8%) compared to White patients (1.4%), but this difference was not statistically significant (p = 0.163). The risk ratio (RR = 0.787) and odds ratio (OR = 0.784) suggested a trend toward a lower risk in White patients; however, the confidence intervals crossed 1, indicating that this finding could be due to chance. Likewise, the Kaplan-Meier survival curve showed no significant difference in survival probability (97.22 percent in White patients versus 96.12 percent in Black patients, p = 0.105). The hazard ratio (HR = 0.755, p = 0.907) also helped confirm the absence of a significant difference between groups in terms of time to paralysis. The outcome implies that racial background should not influence the risk of paralysis following GBS when comorbid conditions are adjusted. This correlates with other studies that have established that clinical subtype and immediate infections have greater impacts on GBS severity and progression than racial effects.
Numbness: It is a common and often persistent symptom of GBS, affecting quality of life even when major motor symptoms improve.[14] The risk of numbness was nearly identical between Black/African American (13.9%) and White (14.1%) patients, with a risk difference (RD) of only 0.001 and a non-significant p-value of 0.849. The risk ratio (RR = 1.010) and odds ratio (OR = 1.012) showed no meaningful difference in the probability of developing numbness. Kaplan-Meier analysis revealed a slightly lower one-year survival probability in Black patients (68.92% vs. 71.38%). Still, the log-rank test (p = 0.603) and hazard ratio (HR = 0.970, p = 0.067) indicated that this difference was not statistically significant. The mean time to develop numbness also did not differ significantly between groups (p = 0.560). These findings suggest that both racial groups experience a similar burden of sensory impairment following GBS. Prior studies also indicate that sensory symptoms in GBS are not racially patterned but correlate more with axonal versus demyelinating variants and with disease severity.[15]
Muscle weakness: Muscle weakness is another critical outcome in GBS, as persistent weakness can lead to long-term disability and functional limitations.[16] The risk of developing muscle weakness was slightly higher in Black/African American patients (8.3%) compared to White patients (7.8%), but this difference was not statistically significant (p = 0.333). The risk ratio (RR = 0.931) and odds ratio (OR = 0.925) indicated a small, non-significant trend toward a lower risk in White patients. Kaplan-Meier survival analysis revealed that Black patients had a slightly lower one-year survival probability (82.51% vs. 84.42%), although this difference was not statistically significant (p = 0.134). The hazard ratio (HR = 0.890, p = 0.896) indicated no significant difference in time to event between groups. The mean time to muscle weakness was also nearly identical (p = 0.952). These results indicate that muscle weakness following GBS occurs at similar rates and progression in both racial groups, suggesting that disparities in treatment or disease severity are not likely driving factors. Muscle weakness in GBS has been closely associated with antecedent infections like Campylobacter jejuni and the presence of axonal damage, which are independent of racial background.[17]
Mortality: The most serious consequence of GBS is mortality; severe cases often result in respiratory failure, autonomic dysfunction, or complications of prolonged hospital stay. There was no significant difference in the one-year mortality between White (6.9%) and Black/African American (6.6%) patients. The difference in risk (RD = 0.002, p = 0.694) and the risk ratio (RR = 1.033) indicated no significant risk in mortality between the groups. The Kaplan-Meier survival analysis method indicated comparable one-year survival rates (83.67 percent of patients of White races vs. 82.47 percent of patients of Black races) and a non-significant log-rank test (p = 0.791). The HR (0.978, p = 0.367) also indicated that no significant difference existed in the time-to-mortality earlier seen between races. The results suggest that racial background does not affect survival in the case of GBS after matching the patients on major comorbid disorders.[18,19]
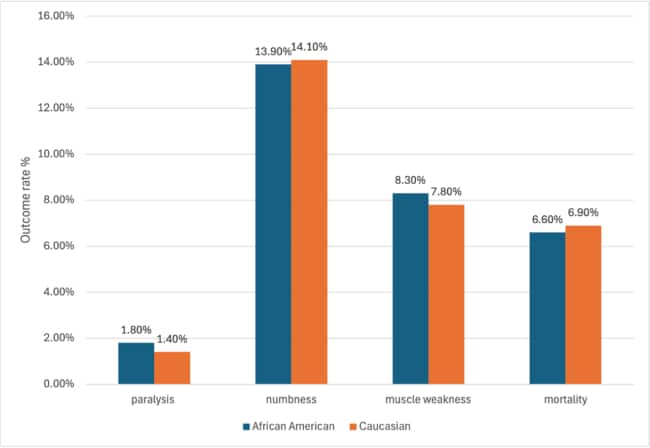
Figure 3: Outcome comparison rate by racial group in GBS patients
Implications: This study found no significant racial differences in one-year outcomes of GBS after controlling for key comorbidities. The similar results between Black and White patients of paralysis, numbness, muscle weakness, and death indicate that under the normalization of clinical care, race alone is not a predictor of short-term outcomes. These results highlight the importance of distributing early diagnosis and treatment to reduce disparities. New studies must examine whether there is long-term functional recovery and the availability of rehabilitation, and whether all people may have varying utilization of treatment to identify hidden factors contributing to outcome variations. Identification of these variables can guide specific measures that can be implemented to minimize inequalities further and equalize their health outcomes, including neuroimmune diseases such as GBS.
Beyond comorbidity balance, system-level factors likely shape GBS outcomes and could mask or magnify racial differences. Timeliness and consistency of acute care are key examples, including the time from presentation to neurology evaluation, IVIG or plasma-exchange initiation, and respiratory monitoring intensity (e.g., early NIF/VC trending, ICU vs. step-down admission), as well as standardized dysautonomia protocols. Care organization, including weekend or after-hours presentation, interfacility transfer delays, and hospital GBS volume, may also influence trajectories. Post-acute resources also matter, including access to inpatient rehabilitation, home health services, equipment, and timely outpatient neuromuscular follow-up. Insurance type, language services, neighborhood deprivation, and transportation can affect each step, from acute therapy to rehabilitation adherence. None of these constructions is fully captured by comorbidity adjustment, yet they plausibly affect paralysis, weakness, and survival.
Future work should explicitly model these domains using hierarchical (patient-within-hospital) analyses with hospital random effects; include year and hospital-level covariates to address secular trends and center practices; and incorporate proxies for access and care coordination (e.g., documented receipt and timing of IVIG/plasma exchange, ICU admission within 24 hours, discharge destination, outpatient neurology follow-up within 90 days). Where feasible, linkage to community indices (area deprivation) and facility characteristics (teaching status, volume) would clarify whether equivalent outcomes persist after accounting for care environment. Approaches such as instrumental variables (e.g., distance to tertiary centers) or mediation analysis for treatment timing could further distinguish between patient-level and system-level contributions, while preserving the core finding of similar one-year outcomes in matched Black and White patients.[18,19]
Conclusion
In the present study of propensity-matched Black/African American and white Guillain-Barr Syndrome patients, no significant racial disparities in one-year outcomes were identified. There was no statistically significant difference among the four key results or outcomes, which consisted of mortality, muscle weakness, numbness, and paralysis. These findings suggest that racial background is not essential in creating disparities in GBS, with consideration of other diseases.
This highlights the importance of early intervention and equitable access to healthcare, ensuring that all patients receive a timely diagnosis and an appropriate course of action. The possible differences that could not be realized during this study may be further explained by future research that requires a larger population and more comprehensive assessments of healthcare accessibility, treatment courses, and social factors related to health.
References
- Ropper AH, Wijdicks EFM. Guillain-Barré Syndrome. Medscape. Updated July 29, 2024.
Guillain-Barre Syndrome - Burns TM. Guillain-Barré syndrome. Semin Neurol. 2008;28(2):152-167. doi:10.1055/s-2008-1062261
PubMed | Crossref | Google Scholar - Kalita J, Misra UK, Goyal G, Das M. Guillain-Barré syndrome: subtypes and predictors of outcome from India. J Peripher Nerv Syst. 2014;19(1):36-43. doi:10.1111/jns5.12050
PubMed | Crossref | Google Scholar - van Doorn PA, Ruts L, Jacobs BC. Clinical features, pathogenesis, and treatment of Guillain-Barré syndrome. Lancet Neurol. 2008;7(10):939-950. doi:10.1016/S1474-4422(08)70215-1
PubMed | Crossref | Google Scholar - Sejvar JJ, Baughman AL, Wise M, et al. Population incidence of Guillain-Barré syndrome: a systematic review and meta-analysis. Neuroepidemiology. 2011;36(2):123-133. doi:10.1159/000324710
PubMed | Crossref | Google Scholar - Shahrizaila N, Lehmann HC, Kuwabara S. Guillain-Barré syndrome. Lancet. 2021;397(10280):1214-1228. doi:10.1016/S0140-6736(21)00517-1
PubMed | Crossref | Google Scholar - Leonhard SE, van der Eijk AA, Andersen H, et al. An International Perspective on Preceding Infections in Guillain-Barré Syndrome: The IGOS-1000 Cohort. Neurology. 2022;99(12):e1299-1313. doi:10.1212/WNL.0000000000200885
PubMed | Crossref | Google Scholar - Langerak T, van Rooij I, Doornekamp L, et al. Guillain-Barré syndrome in Suriname: clinical presentation and identification of preceding infections. Front Neurol. 2021;12:635753. doi:10.3389/fneur.2021.635753
PubMed | Crossref | Google Scholar - Rajabally YA, Uncini A. Outcome and its predictors in Guillain-Barré syndrome. J Neurol Neurosurg Psychiatry. 2012;83(7):711-718. doi:10.1136/jnnp-2011-301882
PubMed | Crossref | Google Scholar - Bae JS, Kim YJ, Kim JK. Diabetes mellitus exacerbates the clinical and electrophysiological features of Guillain-Barré syndrome. Eur J Neurol. 2016;23(3):439-446. doi:10.1111/ene.12885
PubMed | Crossref | Google Scholar - van Koningsveld R, Steyerberg EW, Hughes RA, Swan AV, van Doorn PA, Jacobs BC. A clinical prognostic scoring system for Guillain-Barré syndrome. Lancet Neurol. 2007;6(7):589-594. doi:10.1016/S1474-4422(07)70130-8
PubMed | Crossref | Google Scholar - Walgaard C, Lingsma HF, Ruts L, et al. Early recognition of poor prognosis in Guillain-Barre syndrome. Neurology. 2011;76(11):968-975. doi:10.1212/WNL.0b013e3182104407
PubMed | Crossref | Google Scholar - Esposito S, Longo MR. Guillain-Barré syndrome. Autoimmun Rev. 2017;16(1):96-101. doi:10.1016/j.autrev.2016.09.022.
PubMed | Crossref | Google Scholar - van den Berg B, Walgaard C, Drenthen J, et al. Guillain-Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol. 2014;10(8):469-482. doi:10.1038/nrneurol.2014.121.
PubMed | Crossref | Google Scholar - Leonhard SE, Mandarakas MR, Gondim FAA, et al. Diagnosis and management of Guillain-Barré syndrome in ten steps. Nat Rev Neurol. 2019;15(11):671-683. doi:10.1038/s41582-019-0250-9
PubMed | Crossref | Google Scholar - Yuki N, Hartung HP. Guillain-Barré syndrome. N Engl J Med. 2012;366(24):2294-2304. doi:10.1056/NEJMra1114525
PubMed | Crossref | Google Scholar - Kuwabara S, Yuki N. Axonal Guillain-Barré syndrome: concepts and controversies. Lancet Neurol. 2013;12(12):1180-1188. doi:10.1016/S1474-4422(13)70215-1
PubMed | Crossref | Google Scholar - Tewedaj ZD, Huluka DK, Kebede YT, et al. A retrospective analysis of the clinical profile and factors associated with mortality and poor hospital outcomes in adult Guillain-Barré syndrome patients. Sci Rep. 2024;14(1):15520. doi:10.1038/s41598-024-65265-0
PubMed | Crossref | Google Scholar - Lawn ND, Fletcher DD, Henderson RD, Wolter TD, Wijdicks EF. Anticipating mechanical ventilation in Guillain-Barré syndrome. Arch Neurol. 2001;58(6):893-898. doi:10.1001/archneur.58.6.893
PubMed | Crossref | Google Scholar
Acknowledgments
Not applicable
Funding
None
Author Information
Corresponding Author:
Rawan Elkomi
Department of Internal Medicine
Howard University College of Medicine, Washington, USA
Email: rawan.elkomi@gmail.com
Co-Authors:
Somtochi Edeh, Sair Ahmad Tabraiz, Amara Chike, Emmanuel Kerolle, Samrawit Zinabu, Mekdem Bisrat, Elizabeth Beyene
Department of Internal Medicine
Howard University, Washington, DC, USA
Jeff Palmer, Quinton Johnson, Shivani Waghmare
Department of Medicine
Howard University College of Medicine, Washington, DC, USA
Miriam Michael
Department of Internal Medicine
Howard University College of Medicine, Washington, USA
University of Maryland School of Medicine, Baltimore, USA
Authors Contributions
Jeffrey Palmer II and Rawan Elkomi conceptualized and designed the study, conducted analyses, contributed to drafting, and revised the manuscript. Somtochi Edeh, Amara Chike, Emmanuel Kerolle, and Mekdem Bisrat contributed to data extraction, processing, validation, and analysis, and reviewed the manuscript. Quinton Johnson and Samrawit Zinabu conducted a literature review and synthesis, contributed to the drafting of the manuscript, and revised it. Sair Ahmad Tabraiz and Shivani Waghmare assisted with data curation, analyses, and results drafting, and reviewed the manuscript. Elizabeth Beyene provided clinical interpretation, contributed to the Discussion, and revised the manuscript. Miriam Michael provided senior supervision, guided study design and interpretation, and critically reviewed the manuscript. All authors approved the final version and are accountable for the content of the work.
Ethical Approval
This study analyzed exclusively de-identified electronic health records, as mandated by the Code of Federal Regulations (CFR) 45 and the Health Insurance Portability and Accountability Act (HIPAA) de-identification standard. According to these regulations, it does not constitute human subjects research and was exempt from institutional review board (IRB) review. Informed consent was not required.
Conflict of Interest Statement
The authors declare that they have no conflicts of interest.
Guarantor
Miriam Michael served as the guarantor of the work.
DOI
Cite this Article
Palmer J, Elkomi R, Edeh S, et al. Racial Differences in Guillain-Barré Syndrome Outcomes: One-Year Retrospective Analysis. medtigo J Neurol Psychiatr. 2025;2(3):e3084235. doi:10.63096/medtigo3084235 Crossref









