Author Affiliations
Abstract
Introduction: In the context of Iraq, encompassing the Kurdistan region, which is characterized as a developing nation with limited resources, the prevalence of Hepatitis B virus (HBV), Hepatitis C virus (HCV), and human immunodeficiency virus (HIV) is relatively low, as per World Health Organization (WHO) statistics. Despite this, preoperative virology screening is performed for all patients in the Kurdistan Region, regardless of the surgical procedure.
Objective: This study aims to ascertain the seroprevalence rates of HBV, HCV, and HIV among pre-operative patients, assessing their significance and exploring the potential for selective rather than routine screening.
Methodology: A retrospective cross-sectional descriptive study was undertaken, involving the collection of pre-operative virology screening test results from February and March 2023. The data were obtained from the laboratory departments at Maternity Teaching Hospital and Rozhhalat Emergency Hospital in Erbil, Kurdistan Region. A cluster sample comprising 3950 test results was compiled and analyzed using IBM Statistical Package for the Social Sciences (SPSS).
Results: Out of the total patient cohort, 0.53% (95% confidence interval [CI]: 0.33-0.81%) tested positive for HBsAg, 0.23% (95% CI: 0.10-0.43%) for anti-HCV antibody, and 0.076% (95% CI: 0.016-0.222%) for anti-HIV antibody/HIV antigen. Statistical analysis revealed no significant gender-based differences in the seropositivity for HBV, HCV, and HIV (p > 0.05).
Conclusion: The observed low seroprevalence among pre-operative patients in the Kurdistan Region, coupled with existing constraints on health resources, suggests that routine pre-operative virology screening may not be cost-effective. Instead, a strategy involving selective screening, heightened awareness among healthcare personnel of these viruses, and strict adherence to standard precautions is imperative and potentially adequate.
Keywords
Hepatitis B virus, Hepatitis C virus, Human immunodeficiency virus, Preoperative tests, Cross-sectional study.
Introduction
According to the WHO, an estimated 354 million people worldwide live with HBV and HCV infection. It is important to mention that these infections together are the most common cause of viral hepatitis related deaths, liver cirrhosis, and liver cancer.[1] While Iraq was among the countries with a low prevalence of hepatitis B (<2%), it showed a moderately high prevalence of hepatitis C (2%- <5%) in 2019.[2] HIV continues to be a major global public health problem, with approximately 38.4 million people living with the infection by the end of 2021 and 40.1 million deaths that have occurred so far.[3] Iraq has been considered a country with low HIV prevalence (<2%) and low epidemic level of HIV/acquired immunodeficiency syndrome (AIDS).[4]
Patients in the Kurdistan Region, Iraq, who are undergoing any surgery are routinely screened for HBV, HCV, and HIV preoperatively. However, this is not a universal preoperative screening process. For instance, high-income countries such as the United Kingdom, Germany, and France do not routinely conduct preoperative virology screening.[5-7] However, in developing countries other than Iraq, such as India and Turkey, preoperative virology screening is routinely used.[8-10] This raises the question of why this is a routine preoperative practice in the Kurdistan Region and whether it is justifiable from an epidemiological perspective. Furthermore, if it proves effective, it raises the question of whether this type of preoperative screening should be implemented universally as routine practice.
HBV, HCV, and HIV are responsible for the majority of blood-borne infections that are occupationally acquired. Furthermore, the greatest proportion of occupational transmission occurs through percutaneous injury with hollow-bore needles used for vascular access.[11] Surgeons are at high risk of acquiring these infections since they have higher rates of percutaneous injury than other healthcare workers. In developing countries, occupational blood exposure is of great concern, as exposure to bloodborne pathogens is higher in these settings.[12] Consequently, routine preoperative virology screening to identify positive patients and the implementation of appropriate precautions for their care could be considered standard practice, especially in developing countries. However, it raises the question whether adherence to standard precautions, including but not limited to the use of personal protective equipment and adequate sterilization with every patient, regardless of their infection status, is sufficient for transmission prevention.[13] Furthermore, this raises the question of whether universal triple screening (HBV, HCV, and HIV) prior to any invasive intervention is cost-effective from the perspective of the national health budget. Although patients in public hospitals do not directly pay for the screening, individuals in private institutions usually bear a cost of approximately 27,000 Iraqi dinars. Therefore, the unit cost of the screening test was assumed to be 27,000 IQD. In 2022, health expenditure in Iraq amounted to approximately $279.6 per capita.[14] Consequently, the cost of a single triple-screening test accounted for about 6.7% of the per-capita health resources allocated in that year. This cost-burden for a single test is substantial, especially in a resource limited-health system.
The HBV causes the potentially life-threatening liver condition hepatitis B, which can be acute or chronic. The virus is transmitted through vertical transmission and through contact with blood and other body fluids, including saliva, menstrual, vaginal, and seminal fluids. The latter can occur through needle stick injuries, piercings, tattooing, sexual transmission, as well as reuse of contaminated syringes or sharp objects in health care settings or among drug users in the community. While mortality in acute hepatitis B mainly results from acute liver failure, the high mortality in the chronic illness is due to the development of liver cirrhosis and hepatocellular carcinoma.[15]
Infection with the hepatitis C virus causes acute and chronic liver diseases as well. The blood-borne virus is most transmitted via medical equipment that is being reused or inadequately sterilized, particularly needles and syringes in health care settings, the transfusion of blood and blood products without prior screening, as well as sharing injection equipment among drug users. Vertical and sexual transmission via practices leading to the exposure of blood are less common.[16] The chronic hepatitis C infection is associated with an increased risk of liver cirrhosis and hepatocellular carcinoma.[17]
HIV weakens the host defence against many infections and types of malignancies. Regarding transmission, HIV is acquired through the exchange of various body fluids, including blood, breast milk, vaginal secretions, and semen. The risk of transmission is increased by certain behaviors in the community, including condomless anal or vaginal sex, having other sexually transmitted infections, and sharing contaminated injection equipment among drug users. Also, receiving unscreened blood transfusions, tissue transplantations, unsafe injections, unsterile medical procedures, and accidental needle stick injuries are routes of transmission in health care settings.[18] This study aims to determine the seroprevalence of HBV, HCV, and HIV among pre-operative patients to evaluate the importance of routine pre-operative virology screening in the Kurdistan Region. Thus, it can be determined whether routine preoperative virology screening is necessary in the Kurdistan Region, as it does not appear to be a universal practice.
Methodology
Study design and study setting: A retrospective cross-sectional study was conducted at two tertiary hospitals (Maternity Teaching Hospital and Rozhhalat Emergency Hospital) in Erbil City, the Kurdistan Region of Iraq.
Time of the study: This study was conducted from the 26th of March 2023 until the 14th of May 2023. The data collection period was from 2nd of April 2023 until 26th of April 14th, 2023. This Study was approved by the research committee at the University of Kurdistan- Hewlêr, School of Medicine, and by the General Directorate of Health – Erbil. A total of 3950 virology test results from February and March 2023 were collected from both hospitals.
Inclusion criteria: The only inclusion criterion for this study is that virology tests were ordered for patients prior to surgery. Other inclusion criteria, determined prior to data collection, included being at least 18 years old and residing in the Kurdistan Region. However, due to the limitations of the retrospective registry, demographic variables were restricted to gender, and all patients recorded in the surgical virology log during the study period were included.
Data collection: The official letter of the General Directorate of Health – Erbil was given to the director of each hospital to obtain their written and verbal consent to access the corresponding laboratory departments. There, virology screening test results, each providing information on HBV, HCV, and HIV for a single patient, were available. The study population comprises preoperative patients in the Kurdistan Region, Iraq, and the study sample was obtained via cluster sampling, with each hospital representing a cluster. The choice of these clusters was based on the observation that virology screening test results are not recorded with the corresponding indications, preventing the identification of preoperative patients. For that matter, hospitals in which most invasive procedures are surgeries were selected.
Regarding the virology screening test procedures within the laboratories, in Maternity Teaching Hospital, the blood samples were centrifuged to obtain serum, which was then assessed manually by using enzyme-linked immunosorbent assay (ELISA) in order to detect the hepatitis B surface antigen (HBsAg), the hepatitis C antibody (anti-HCV antibody), and the human immunodeficiency virus antibody (anti-HIV antibody). The results of these tests were later documented on a computer. At Rozhhalat Emergency Hospital, blood samples were centrifuged to obtain serum, which was then analyzed by the LiasonXL machine for the detection of HBsAg, anti-HCV antibody, anti-HIV antibody, and HIV antigen. The test results were later documented in an A4-sized notebook, all handwritten.
Statistical analysis: All test results were entered into a single table in Microsoft Excel 2010, and the proportions of positive test results among the total number of tests for HBsAg, anti-HCV antibody, and anti-HIV antibody/HIV antigen were calculated using IBM SPSS Statistics on Windows 10 Pro. These proportions represent the seroprevalence. Additionally, the proportion of positive test results for each virus was calculated for male and female patients. The 95% confidence intervals (CI) for the seroprevalence of each virus and the statistical significance of differences in positive test results between male and female patients were calculated. The latter was determined by analyzing the dependency between sex and positive test results for HBV, HCV, and HIV by the Pearson-Chi-Square Test using the mentioned program.
Ethical consideration: This study was approved by the research ethics committee of the University of Kurdistan- Hewlêr. Due to the retrospective nature of the study, the requirement for informed consent was waived by the ethical committee. Nevertheless, patient confidentiality was strictly maintained in this study through de-identification of the data prior to analysis.
Results
A total of 3950 patients were tested, among whom 2748 were female, and 1202 were male patients. The total number of patients that tested positive for HBsAG was 21 (0.53%) (95% CI: 0. 33-0.81%), for anti-HCV antibody was 9 (0.23%) (95% CI: 0.10-0.43%) and for anti-HIV antibody/ HIV antigen was 3 (0.076%) (95% CI: 0.016-0.222%). Among the total number of female patients, 13 were positive for HBsAg (0.47%), 4 were positive for anti-HCV antibody (0.15%), and 1 was positive for anti-HIV antibody/HIV antigen (0.036%). Regarding the total number of male patients, 8 were positive for HBsAg (0.67%), 5 for anti-HCV (0.42%), and 2 for anti-HIV antibody/HIV antigen (0.17%). The difference in the seropositivity for HBV between males and females was not statistically significant, as p>0.05 (Pearson-Chi-Square test, p=0.74). Additionally, there was no statistically significant difference in HCV seropositivity between males and females (Pearson chi-square test, p=0.26). Lastly, the difference in seropositivity for HIV also did not prove to be statistically significant, as p>0.05 (Pearson-Chi-Square test, p=0.39).
|
Valid |
HbsAg | Anti-HCV antibody | Anti-HCV antibody/HCV antigen | |||
| Number
(n) |
Valid
percentage (%) |
Number
(n) |
Valid
percentage (%) |
Number
(n) |
Valid
percentage (%) |
|
| Negative | 3929 | 99.5 | 3941 | 99.8 | 3947 | 99.9 |
| Positive | 21 | 0.5 | 9 | 0.2 | 3 | 0.1 |
| Total | 3950 | 100 | 3950 | 100 | 3950 | 100 |
Table 1: The number (n) of positive and negative patients and valid percentages (%) for HbsAg, anti-HCV antibody, and anti-HCV antibody/HCV antigen
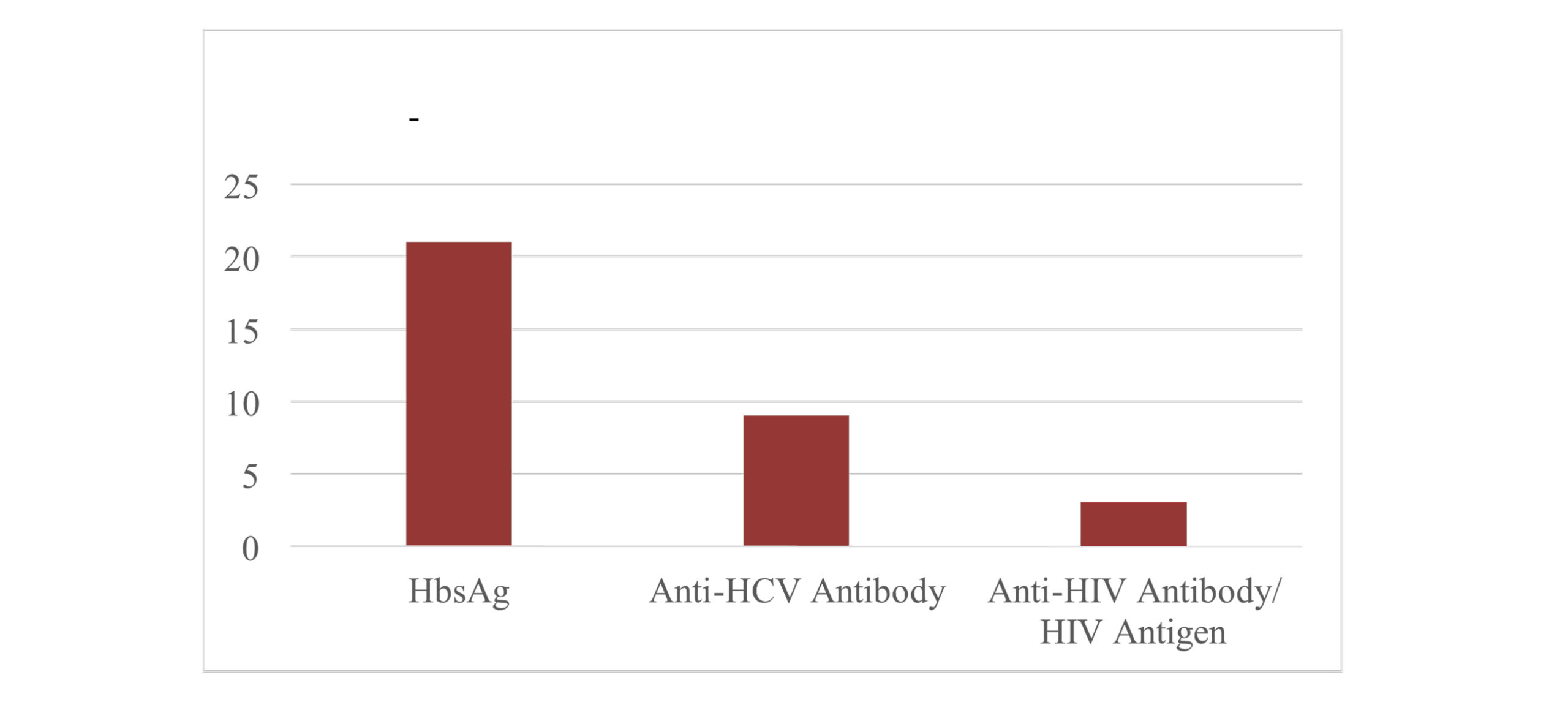
Figure 1: The number (n) of positive patients for HbsAg, anti-HCV antibody, and anti-HIV antibody/HIV antigen
|
Gender |
HbsAg | Anti-HCV antibody | Anti-HIV antibody/ HIV antigen | ||||
| Number
(n) |
Valid
percentage (%) |
Number
(n) |
Valid
percentage (%) |
Number
(n) |
Valid
percentage (%) |
||
|
Male |
Negative | 1194 | 99.3 | 1197 | 99.6 | 1200 | 99.8 |
| Positive | 8 | 0.7 | 5 | 0.4 | 2 | 0.2 | |
| Total | 1202 | 100 | 1202 | 100 | 1202 | 100 | |
|
Female |
Negative | 2735 | 99.5 | 2744 | 99.9 | 2747 | 100 |
| Positive | 13 | 0.5 | 4 | 0.1 | 1 | 0 | |
| Total | 2748 | 100 | 2748 | 100 | 2748 | 100 | |
Table 2: The number (n) and valid percentages (%) of positive and negative male and female patients for HbsAg, anti-HCV antibody, anti-HIV antibody/HIV antigen
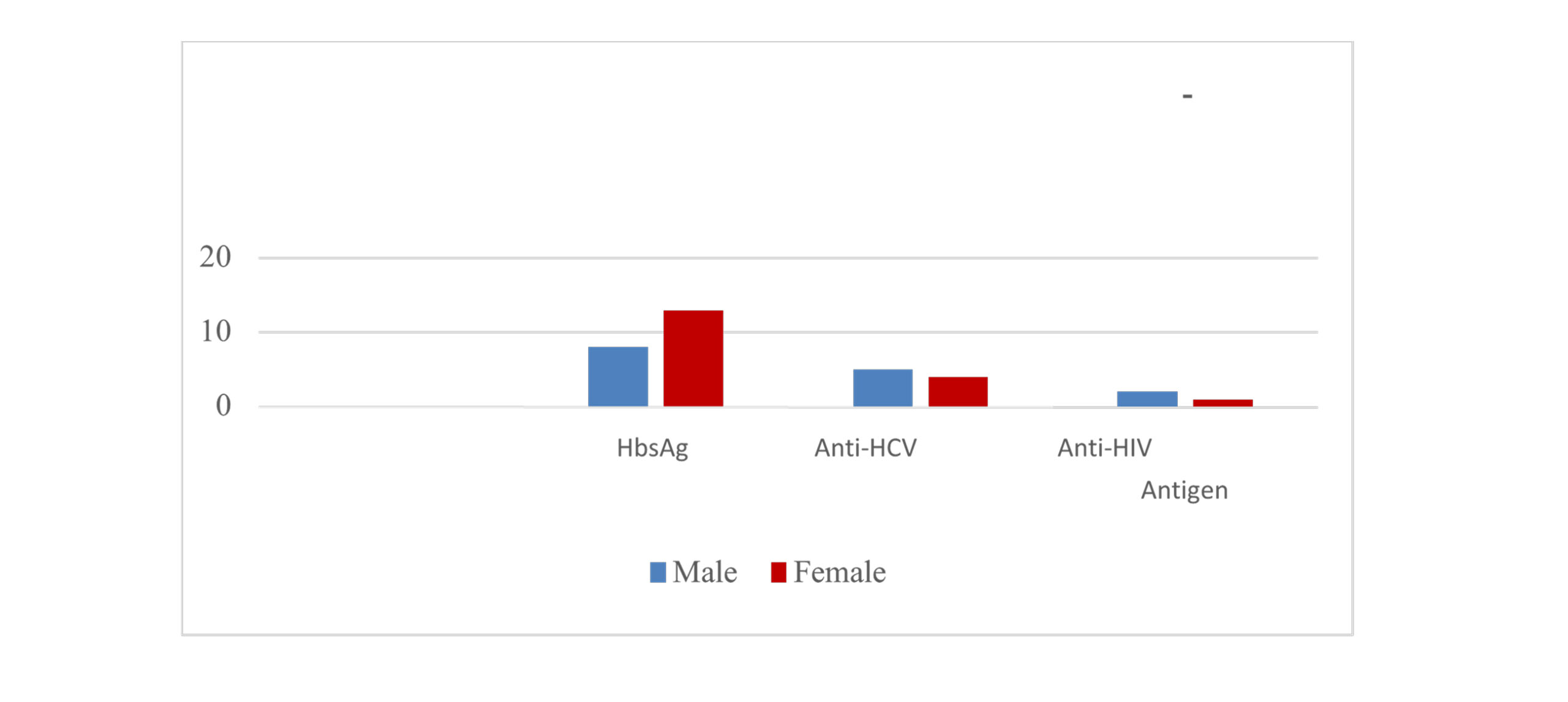
Figure 2: The number (n) of positive patients for HbsAg, anti-HCV antibody, and anti-HIV antibody/HIV antigen among males and females
Discussion
The number of patients who tested positive for HBsAg was higher than the number of patients with a positive serostatus for HCV and HIV. This may reflect the fact that hepatitis B is considerably more common than hepatitis C worldwide. Reasons for that include that HBV is 510 times more infectious than HCV and more stable in terms of surviving and remaining highly contagious outside of the body. In contrast to hepatitis B, hepatitis C is curable.[19] Furthermore, the transmission of HBV is 100-fold more likely than that of HIV.[20] Considering the results, comparable to our seroprevalence of HBV (0.53%), can be found in a similar study conducted in Japan, which determined that 0.39% of pre-operative patients were positive for HBsAg, and in France, with a seroprevalence of 0.38% for HBV among pre-operative patients.[7,21] Regarding the seroprevalence of HCV among preoperative patients, results similar to ours (0.23%) have been reported in a Turkish study, which reported 0.3%.[12] Also, in the same study conducted in Japan, a seroprevalence of 0.78% for HCV among preoperative patients was reported, which is similar to our result. Regarding the seroprevalence of HIV (0.076%), similar results were found in a different study that was conducted in Turkey with a seroprevalence of 0% and in the same study conducted in Japan with 0.08%.[10]
In Basrah, Iraq, 2.45% of preoperative patients tested positive for HBV, a rate higher than ours.[22] Another study conducted in India, which reported higher seroprevalence for all three viruses, reported a seroprevalence of 2.08% for HBV.[23] Regarding the seroprevalence of HCV, another study conducted in India reported 1.91%, whereas another study conducted in Pakistan reported as high as 21.38%.[24,25] These results exceed the HCV seroprevalence reported in the current study. The seroprevalence for HIV in this current study is smaller than the seroprevalence of 3.0% among pre-operative patients in a study conducted in Nigeria.[26] The previously mentioned study conducted in France also determined a higher seroprevalence of 0.635% for HIV.[7]
One may argue that preoperative virology screening should be a routine investigation in a developing country with a high prevalence of these viruses in the general population. Due to a lack of data on the prevalence of each virus in the general population of the Autonomic Region Kurdistan, data from Iraq were applied to the region. As previously mentioned, Iraq was regarded as a low-prevalence and moderately high-prevalence country for HBV and HCV, respectively, in 2019.[2] Also, Iraq was considered a country with low HIV prevalence.[4] Thus, only screening for the hepatitis C virus should be used as a routine preoperative investigation. However, the seroprevalence of HCV was relatively low in the current study, questioning the necessity to perform routine pre-operative virology screening in Iraq and the Kurdistan Region. The results of the current study are comparable to studies that were conducted in either developed countries, such as Japan and France, or a developing country that has an overall low to moderate prevalence of each virus within the general population, such as Turkey.[10,27] Even the developed countries listed above have a relatively low prevalence of each virus in the general population.
Data from 2019 indicated that France had a low overall prevalence of hepatitis B (<2%) and a very low prevalence of hepatitis C (0- <1.0%). Similarly, Japan was regarded as a low-prevalence country for hepatitis B and hepatitis C (1.0-<1.5%).[2] In 2021, the overall prevalence of HIV/AIDS in France was 0.3%.[28]Additionally, because most studies reporting higher seroprevalences for the viruses were conducted in developing countries with higher prevalence of each virus in the general population, the Kurdistan Region may be considered to have relatively low to moderate seroprevalence of blood-borne viruses among pre-operative patients compared with other developing countries. For instance, India is among the countries with intermediate HBV endemicity, with a prevalence of 2-4% in the general population.[29] The prevalence of HCV in the general population of India is estimated to range from 0.09% to 15%.[30] India showed an adult prevalence of 0.21% for HIV in 2021.[31]
Pakistan and Nigeria, which also showed higher seropositivity rates, are considered to be countries with a high prevalence of HBV (≥8%) and HCV (≥5%).[2] The estimated prevalence of HIV in the general population of Pakistan is 0.1%, and in the adult population of Nigeria in 2019 is 1.4%.[32] Given the low seroprevalences among pre-operative patients and the overall low to moderate prevalences in the general population for the three viruses in Iraq, the Kurdistan Region, Turkey, and the aforementioned developed countries, routine virology screening in pre-operative patients does not appear essential for preventing transmission. Instead, selective screening of patients with risk factors and a clinical presentation suggestive of infection with a blood-borne virus appears sufficient in these countries. Routine pre-operative virology screening may be essential in specific developing countries or in any country with high seroprevalence among pre-operative patients and high prevalence in the general population.
Greater prevalence of HBV, HCV, and HIV in the general population of any country may be explained by the lack of precautions for transmission prevention. To prevent hepatitis B transmission, the WHO recommends that all neonates, children, and adults at high risk of infection receive the safe and effective hepatitis B vaccine.[33] Additional precautions include avoiding exposure to blood and bodily fluids; thoroughly washing hands after potential exposure; using condoms; using sterile needles for tattoos and piercings; avoiding sharing needles and sharps; and avoiding the use of illegal street drugs.[34] The same measures apply to preventing hepatitis C, except that no vaccine is available. The risk of HIV transmission can be prevented by the use of protected intercourse, including latex condoms, not sharing needles, taking pre-exposure prophylaxis if at high risk of exposure, getting tested for other sexually transmitted illnesses, and appropriate treatment of infected individuals with antiretroviral therapy.[35,36] Spreading awareness about these precautions is imperative.
Considering the high risk of exposure among health care workers, the CDC/Healthcare Infection Control and Prevention Advisory Committee introduced the concept of “Standard Precautions” in their “1996 Guideline for Isolation Precautions in Hospitals” according to which certain infection preventative measures should be applied to all patients, regardless of their infection status in order to prevent the spread of pathogens via blood and bodily fluids.[14] The following should be applied to all settings in which healthcare is delivered:
- Personal protective equipment
- Hand hygiene
- Respiratory hygiene
- Aseptic technique
- Needle-stick and sharps injury prevention
- Cleaning and disinfection
- Waste disposal
Compliance with this guideline, which treats every patient as a positive case, is imperative to reduce the risk of transmission. This raises the question of whether investing solely in standard precautions is more cost-effective than implementing additional precautions only for patients deemed positive by routine pre-operative virology screening.[37]
Strengths and limitations: Our study has several strengths, including a large sample size, data from tertiary hospitals that serve a wide range of patients, and a recent data collection period. About limitations, the retrospective nature of this study precluded the collection of sufficient data. To elaborate, aside from the name and serostatus, each test result contained no additional information about the patient or the procedure performed. This did not allow the exclusion of ineligible subjects, thereby limiting the sample’s representativeness of the preoperative population in the Kurdistan Region. Furthermore, gender was inferred based on nomenclature, which may introduce a marginal risk of classification error. Additionally, because the data were collected only in Erbil, the sample did not adequately represent the entire Kurdistan Region. Finally, it should be acknowledged that ELISA-based manual testing is subject to a diagnostic window period, during which recent infections may yield false-negative results when compared with more sensitive molecular techniques such as polymerase chain reaction (PCR) testing.
Recommendations:
- The investigation of the seroprevalence of HBV, HCV, and HIV in the pre-operative population in the Kurdistan region by a prospective cross-sectional study design to survey each patient who is screened for the viruses pre-operatively.
- The conduction of such a study over a longer period of time, with a greater coverage of hospitals in the remaining provinces in the Kurdistan Region. This enables more accurate determination of the cost-effectiveness of routine preoperative virology screening in subsequent studies.
- The conduction of a study that investigates the adherence to standard precautions within hospitals in the Kurdistan Region.
- We recommend the implementation of Electronic Health Records (EHR) to facilitate integrated clinical auditing, which would allow for better access to detailed patient information.
Conclusion
In Conclusion, routine pre-operative virology screening does not appear to be essential in the prevention of HBV, HCV, and HIV transmission to healthcare staff involved in surgery in countries that have a low prevalence of these viruses, both among pre-operative patients and the general population. Rather, raising awareness of bloodborne pathogens and their prevention among the general population and adherence to standard precautions appear sufficient. Even in countries with relatively high prevalence, it is important to determine whether adherence to standard precautions is sufficient to prevent transmission and is more cost-effective than additional routine preoperative virology screening to avoid unnecessary use of limited resources.
References
- World Health Organization. Hepatitis.
Hepatitis - Centers for Disease Control and Prevention. Global Viral Hepatitis. 2025.
Global Viral Hepatitis - World Health Organization. HIV and AIDS. 2025.
HIV and AIDS - Saleh I, Numan H, Khalil N, Koubaisy H. HIV/AIDS status in Baghdad, Iraq over ten years. Indian J Forensic Med Toxicol. 2021;15(1):2253. doi:10.37506/ijfmt.v15i1.13738
Crossref | Google Scholar - National Institute for Health and Care Excellence (NICE). Routine preoperative tests for elective surgery. 2016.
Routine preoperative tests for elective surgery - LADR Der Laborverbund Dr. Kramer & Kollegen. LADR Startseite.
LADR Der Laborverbund - Assoumou L, Thormann F, Soulié C, et al. Routine screening for HIV, hepatitis B virus and hepatitis C virus in individuals undergoing oral and maxillofacial surgery. HIV Med. 2019;20(5):353-358. doi:10.1111/hiv.12732
PubMed | Crossref | Google Scholar - United Kingdom Government. Countries Defined as Developing by the OECD. 2021.
Countries defined as developing by the OECD - Karim HMR, Mistry T. Influence of Workplace, Location and Experience on Preoperative Routine Viral Screening Tests: A Post-Hoc Observation from an Online Survey. Ulutas Med J. 2020;6(2):74-79. doi:10.5455/umj.20200311082916
Crossref | Google Scholar - Yılmaz Türkoğlu E, Konuk ŞG. Assessment of hepatitis B, hepatitis C and human immunodeficiency virus screening results performed before elective eye surgery. MKÜ Tıp Dergisi. 2022;13(47):303‑308. doi:10.17944/mkutfd.1022708
Crossref | Google Scholar - Deuffic-Burban S, Delarocque-Astagneau E, Abiteboul D, Bouvet E, Yazdanpanah Y. Blood-borne viruses in health care workers: prevention and management. J Clin Virol. 2011;52(1):4-10. doi:10.1016/j.jcv.2011.05.016
PubMed | Crossref | Google Scholar - Onerci Celebi O, Araz Server E, Hamit B, Yiğit Ö. The seroprevalence of hepatitis B, hepatitis C, and human immunodeficiency virus in patients undergoing septoplasty. Braz J Otorhinolaryngol. doi:10.1016/j.bjorl.2016.10.008
PubMed | Crossref | Google Scholar - Occupational Safety and Health Administration (OSHA). Bloodborne Pathogens — Worker protections against occupational exposure to infectious diseases. OSHA, U.S. Department of Labor. Accessed January 30, 2026.
Bloodborne Pathogens — Worker protections against occupational exposure to infectious diseases - Kurdsitan 24. KRG Minsitry of Health publishes healthcare statistics. 2023.
KRG Minsitry of Health publishes healthcare statistics - World Health Organization. Hepatitis B. Updated July 23, 2025.
Hepatitis B - World Health Organization. Hepatitis C. Updated July 25, 2025.
Hepatitis C - Goto K, Roca Suarez AA, Wrensch F, Baumert TF, Lupberger J. Hepatitis C Virus and Hepatocellular Carcinoma: When the Host Loses Its Grip. Int J Mol Sci. 2020;21(9):3057. doi:10.3390/ijms21093057
PubMed | Crossref | Google Scholar - World Health Organization. WHO releases first‑ever global guidance for country validation of viral hepatitis B and C elimination. 2021. WHO releases first-ever global guidance for country validation of viral hepatitis B and C eliminati…
- Hepatitis B Foundation. What’s the difference: Hepatitis B vs Hepatitis C? – Hepatitis B Foundation.
What’s the Difference: Hepatitis B vs Hepatitis C? – Hepatitis B Foundation - Thio CL. Hepatitis B and human immunodeficiency virus coinfection. Hepatology. 2009;49(5 Suppl):S138-145. doi:10.1002/hep.22883
PubMed | Crossref | Google Scholar - Sukegawa S, Sukegawa Y, Hasegawa K, et al. The Effectiveness of Pre-Operative Screening Tests in Determining Viral Infections in Patients Undergoing Oral and Maxillofacial Surgery. Healthcare (Basel). 2022;10(7):1348. doi:10.3390/healthcare10071348
PubMed | Crossref | Google Scholar - Hayder T, Hasan T. Prevalence of hepatitis virus B and C in patients in Al Najaf Governorate, Iraq. Int J Pharm Res. 2020;12:195. doi:10.31838/ijpr/2020.12.03.195
Crossref | Google Scholar - Mohan M, Sharma M, Pandey CP, Agarwal A. Preoperative screening of HIV, HBV, HCV essential for surgical team and patients both: a research study in Department of Surgery, Tertiary Care Institute of North India, Rohilkhand Medical College and Hospital, Bareilly (U.P.), India. Int J Contemp Med Res. 2018;5:22. doi:10.21276/ijcmr.2018.5.7.22
Crossref | Google Scholar - Nahvi N, Farooq S. Seroprevalence of hepatitis B, hepatitis C, and HIV 1/2 in patients undergoing surgery in a tertiary care hospital in north India (a hospital-based study). Clin Epidemiol Glob Health. 2020. doi:10.1016/j.cegh.2019.04.004
Crossref | Google Scholar - Malik SJ, Melwani R, Bano F, et al. The Frequency and Risk Factors Identified for HBsAg and Anti-HCV in a Preoperative Screening of Patients Admitted in the Surgical Department at Al-Tibri Medical College Hospital. Cureus. 2020;12(7):e8938. doi:10.7759/cureus.8938
PubMed | Crossref | Google Scholar - Odogu V, Mercieca K, Thampy R, Fiebai B. HIV seroprevalence in patients undergoing ophthalmic surgery in the Niger/Delta region of Nigeria. Niger J Ophthalmol. 2009;16(2). doi:10.4314/njo.v16i2.46742
Crossref | Google Scholar - Asialink Business. Doing Business Guide: Japan. 2024.
Doing Business Guide: Japan - Statista. HIV/AIDS in France – Statistics & Facts. 2024.
HIV/AIDS in France – Statistics & Facts - Premkumar M, Kumar Chawla Y. Chronic Hepatitis B: Challenges and Successes in India. Clin Liver Dis (Hoboken). 2021;18(3):111-116. doi:10.1002/cld.1125
PubMed | Crossref | Google Scholar - National Health Mission. National Viral Hepatitis Control Program (NVHCP).
National Health Mission - National AIDS Control Organization & ICMR‑National Institute of Medical Statistics. India HIV Estimates 2021: Fact Sheets. National AIDS Control Organisation, Ministry of Health and Family Welfare, Government of India; 2022.
India HIV Estimates 2021 - Joint United Nations Programme on HIV/AIDS (UNAIDS). Country Progress Report — Pakistan; Global AIDS Monitoring 2020. 2020.
Country Progress Report — Pakistan; Global AIDS Monitoring 2020 - Hepatitis B Foundation. Prevention Tips for Hepatitis B.
Prevention Tips for Hepatitis B - Hepatitis B Foundation. Hepatitis B Vaccination.
Hepatitis B Vaccination - Cleveland Clinic. HIV & AIDS: Causes, Symptoms, Treatment & Prevention.
HIV & AIDS: Causes, Symptoms, Treatment & Prevention - Centers for Disease Control and Prevention. Treating HIV. 2024.
Treating HIV - Clinical Excellence Commission (NSW Government). Infection Prevention and Control Precautions.
Infection Prevention and Control Precautions
Acknowledgments
The authors would like to acknowledge Maternity Teaching Hospital and Rozhhalat Emergency Hospital for the provision of data.
Funding
The authors received no funds, grants, or financial support for this study.
Author Information
Corresponding Author:
Issra Sorani
Department of Medicine
University of Kurdistan Hewlêr, Iraq
Email: issrasorani@gmail.com
Co-Authors:
Ali Nizar Ali
Department of Medicine
University of Kurdistan Hewlêr, Iraq
Srwa Khalid
Department of Obstetrics and Gynecology
University of Kurdistan Hewlêr, Iraq
Author Contribution
Ali Nizar Mawti contributed to data collection, data curation, preparation of graphs and tables, data analysis, methodology development, and drafting the conclusion. Issra Sorani was responsible for data collection and data analysis, and contributed to writing the introduction, results, discussion, recommendations, and references. Srwa Khalid contributed to the conceptualization of the study, prepared the abstract, and was involved in the review and editing of the manuscript.
Ethical Approval
This study was approved by the Research Ethics Committee of the University of Kurdistan–Hewlêr.
Conflict of Interest Statement
The authors declare no conflicts of interest.
Guarantor
Issra Sorani takes full responsibility for the integrity of the work, from inception to published article.
DOI
Cite this Article
Mawti AN, Sorani I, Khalid S. Preoperative Virology Screening in the Kurdistan Region: Is it Necessary to be Routine? A Cross-sectional Descriptive Study . medtigo J Med. 2026;4(1):e3062419. doi:10.63096/medtigo3062419 Crossref









