Author Affiliations
Abstract
Background: Neck of femur (NOF) fractures are a major public health issue for Fiji’s aging population, with surgical site infections (SSIs) being a devastating complication that increases morbidity, mortality, and healthcare costs. This study provides the first detailed analysis of SSI incidence and its associated risk factors in NOF fracture surgeries at Lautoka Hospital.
Methodology: A retrospective, cross-sectional study was conducted on 56 patients who underwent NOF fracture surgery at Lautoka Hospital. Data on patient demographics, clinical factors, and postoperative outcomes were collected from hospital electronic medical records (EMR), registers, and clinical staff interviews. The study received full ethical approval from the Fiji Human Health Research Ethics Review Committee (FNHRERC) and Aspen Medical.
Results: The study identified an alarming SSI rate of 8.9%. The patient cohort was characteristically geriatric, with a mean age of 69.1 years. Significant risk factors included a high burden of comorbidities (nearly 80% of patients had at least one chronic illness), with diabetes and hypertension being prevalent. Systemic delays were a critical issue, with 71.4% of patients waiting over 48 hours for surgery. The median length of hospital stay was 6 days, but outliers extended up to 56 days, often linked to infection. Staphylococcus aureus was a primary causative organism identified.
Conclusion: The high SSI rate is driven by a combination of a vulnerable, comorbid patient population and significant, modifiable systemic delays in care. The findings underscore an urgent need for targeted, low-cost interventions focused on streamlining preoperative pathways, ensuring protocol adherence, and enhancing postoperative care to improve patient outcomes in Fiji.
Keywords
Neck of femur fracture, Surgical site infection, Fiji, Geriatric patients, Comorbidities, Diabetes.
Introduction
Neck of femur (NOF) fractures represent a growing and complex challenge for healthcare systems worldwide, particularly among the elderly population, where osteoporosis and frailty are common. These fractures are associated with a significant financial and clinical burden, leading to high rates of disability and mortality. Surgical intervention remains the cornerstone of treatment to restore function and reduce mortality.
However, the success of these surgeries is often compromised by postoperative complications, chief among them being SSIs. SSIs are a frequent cause of readmission, prolonged hospital stays, and increased healthcare costs. Globally, SSI incidence in hip surgeries ranges from 2% to 7%. Risk factors are multifactorial and include patient-related factors like Advanced age, poor nutritional status, and comorbidities, as well as procedural factors like prolonged operative duration (>107 minutes).
In Fiji, the challenge is amplified by an aging population and a high prevalence of non-communicable diseases (NCDs) like diabetes and obesity. A previous study at Suva’s CWM Hospital found an 8.7% prevalence of healthcare-associated infections, with SSIs being the most common type, highlighting this as a national issue. This study aims to provide the first comprehensive analysis of the incidence, risk factors, and outcomes associated with SSIs following NOF surgeries at Lautoka Hospital, providing evidence to guide targeted, locally relevant prevention strategies.
Methodology
Study Design and Setting A hospital-based, cross-sectional observational study was conducted at Lautoka Hospital. Lautoka Hospital is a 305-bed divisional teaching hospital, serving over 330,000 people in Fiji’s Western Division and affiliated with Fiji National University.
Study Population The study included 56 consecutive patients who underwent surgery for a neck of femur fracture. This included both male and female patients of varying ages and comorbidities to capture a comprehensive risk profile.
Data Collection: Data was collected retrospectively from October 2023 to October 2024. Information was sourced from multiple channels to ensure a robust dataset, including:
- Hospital EMR and operating theatre logs.
- Trauma ward inpatient registers.
- Questionnaires and interviews with the clinical staff involved in patient care.
Ethical considerations: The study was conducted with strict adherence to ethical guidelines.
- Facility approval to conduct the research was granted by Health Care (Fiji) Pte Ltd T/A Aspen Medical.
- Full scientific, technical, and ethical approval was granted by the FNHRERC on April 24, 2025 (FNHRERC Number: MI 7 2 2025).
- Informed consent was obtained from all participating clinical staff, and patient data was de-identified to ensure confidentiality. Data was stored securely, with access limited to the primary research team.
Results
Part I: Analysis of a retrospective study on surgical site infections (N=56)
This section provides a meticulous, question-by-question breakdown of the data from the ‘Questionnaire for Retrospective Study on SSIs in NOF Fracture Surgeries’.[1] The analysis will serve as the empirical foundation for the subsequent discussion and recommendations.
Patient cohort and baseline characteristics:
Demographic profile: The retrospective study cohort consists of 56 patients who underwent surgery for a neck of femur (NOF) fracture.[1] The gender distribution shows a slight female predominance.
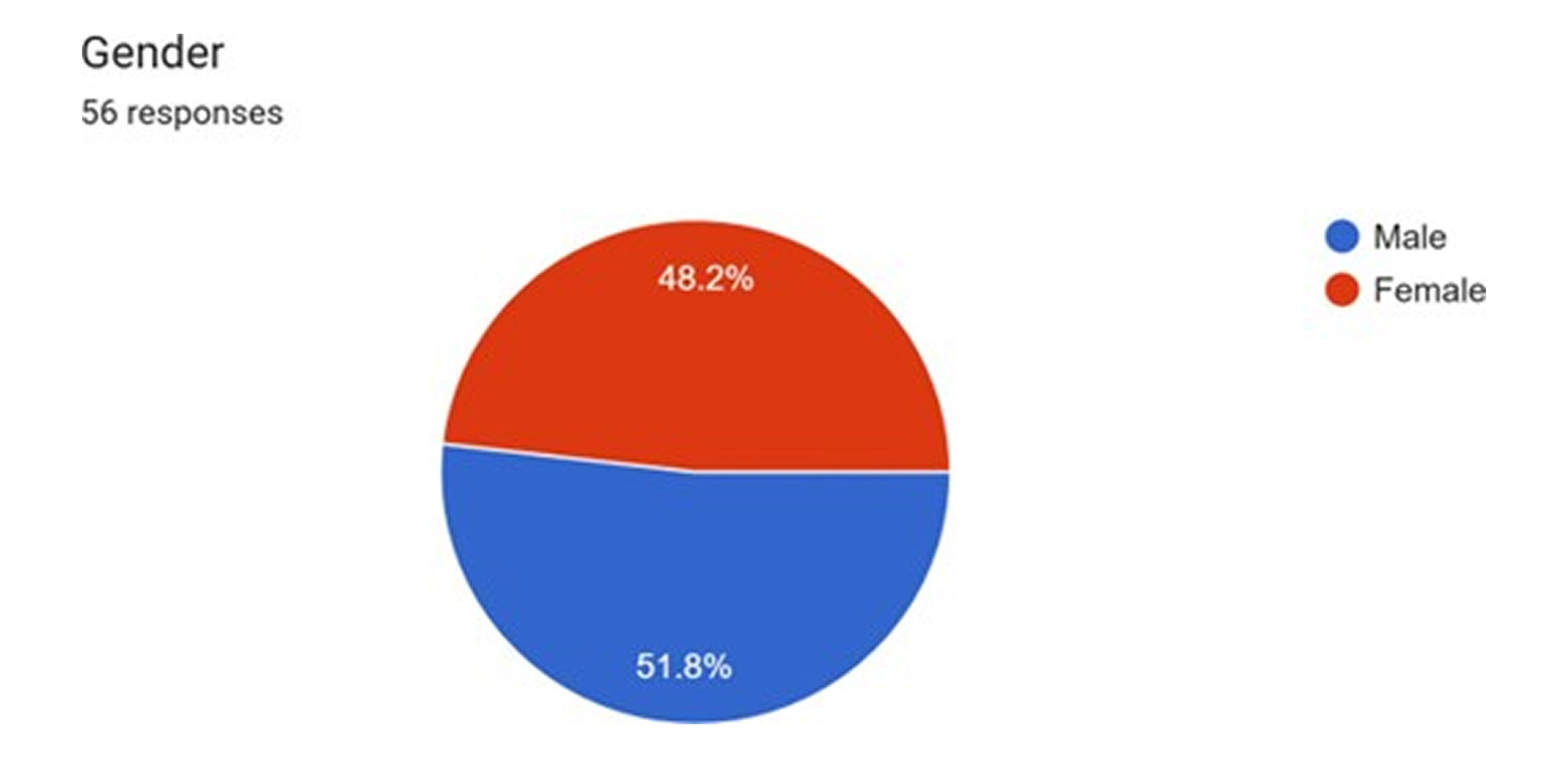
Figure 1: Gender distribution
with 51.8% of patients being female and 48.2% male. This aligns with global epidemiological trends for hip fractures, which are more common in women due to a higher prevalence of osteoporosis.[2]
The age of the patients spans a wide range, from 41 to 96 years. However, the distribution is heavily skewed towards the geriatric population, which is characteristic of NOF fractures, as they are predominantly low-energy fragility fractures resulting from falls in older adults.[2] Key age clusters within the cohort include patients aged 71 (10.7%), 76 (8.9%), and 68 (7.1%).1 The advanced age of this patient population is a critical baseline characteristic, as it is a well-established independent risk factor for a host of adverse postoperative outcomes, including surgical site infections (SSIs), delayed recovery, and postoperative delirium.[3] The physiological reserves of elderly patients are often diminished, making them more vulnerable to the stress of surgery and subsequent complications.
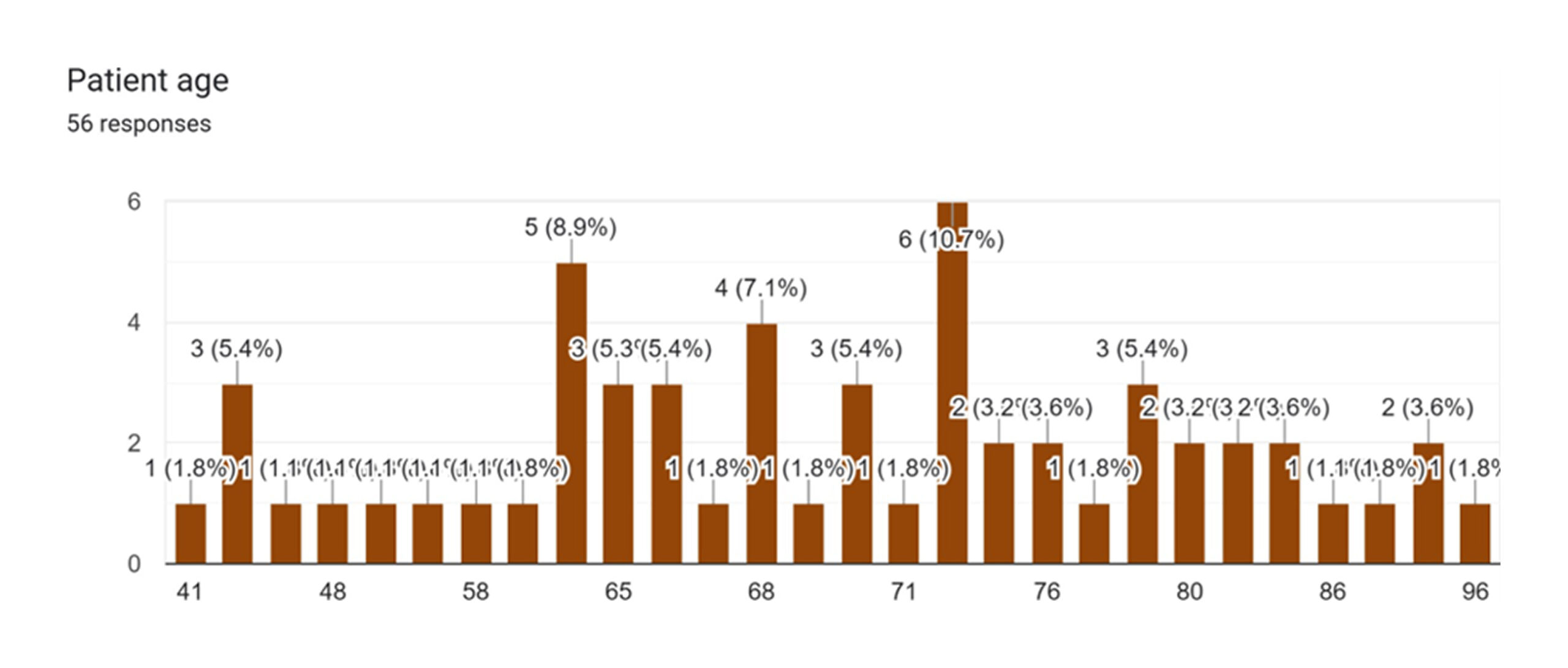
Figure 2: Distribution as per patient age
Analysis of pre-existing comorbidities: The patient cohort demonstrates a profound burden of pre-existing non-communicable diseases (NCDs), a factor with significant implications for surgical risk and outcomes. Based on the 56 responses, hypertension was the most prevalent comorbidity, affecting 57.1% of patients. This was followed closely by diabetes mellitus, present in 48.2% of the cohort. Other significant conditions included chronic kidney disease (17.9%) and cardiovascular disease (14.3%). Critically, only 13 patients, representing 23.2% of the cohort, reported having no pre-existing comorbidities.[4]
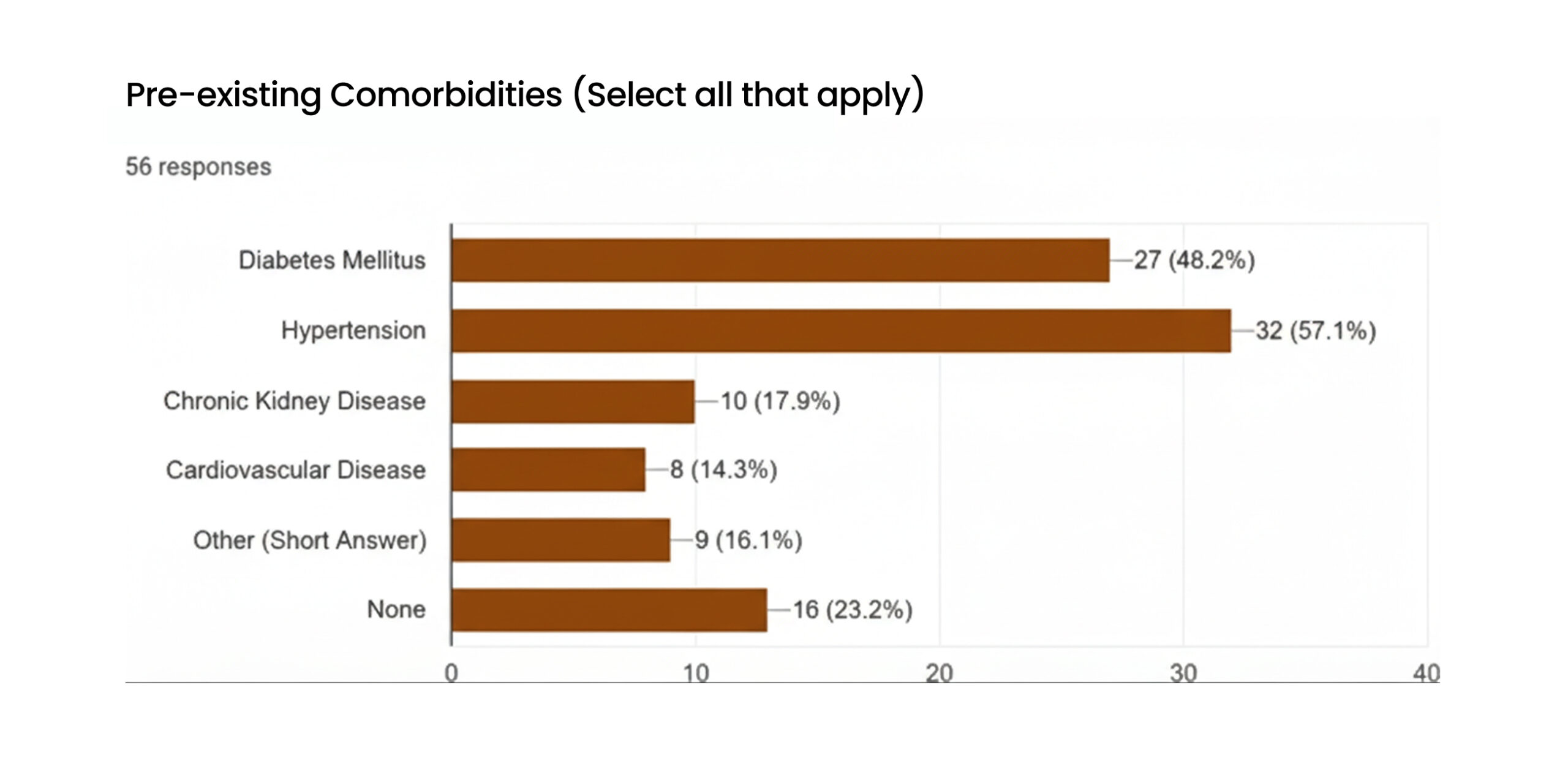
Figure 3: Pre-existing comorbidities
The prevalence of diabetes and hypertension in this surgical cohort is exceptionally high and serves as a central theme for understanding SSI risk in this context. These conditions are not passive background characteristics; they are potent, active drivers of infection risk. Diabetes, in particular, compromises patient outcomes by impairing immune function, reducing microvascular circulation essential for tissue repair, and delaying wound healing, thereby creating a more favorable environment for bacterial proliferation.[5] Similarly, hypertension can be a marker of underlying vascular disease that compromises tissue perfusion at the surgical site.[6,7]
Lifestyle-associated risk factors: Lifestyle-related factors further compound the baseline risk profile of the cohort. Analysis of Body Mass Index (BMI) data, available for 45 patients, reveals that a substantial majority are either overweight (60%) or obese (8.9%). Combined, these two categories account for 68.9% of the patients with available data, while only 28.9% fall within the normal weight range, and 2.2% are underweight.[8-10] This high prevalence of excess body weight is consistent with national data for Fiji, which identifies obesity as a major public health challenge.[8] Obesity is a critical independent risk factor for SSI. Adipose tissue is characterized by relative avascularity, which leads to poor penetration of prophylactic antibiotics and diminished oxygen tension at the wound site. Furthermore, obesity is associated with a state of chronic low-grade inflammation, which can impair the normal physiological processes of wound healing and immune response, making patients more susceptible to infection.[11]
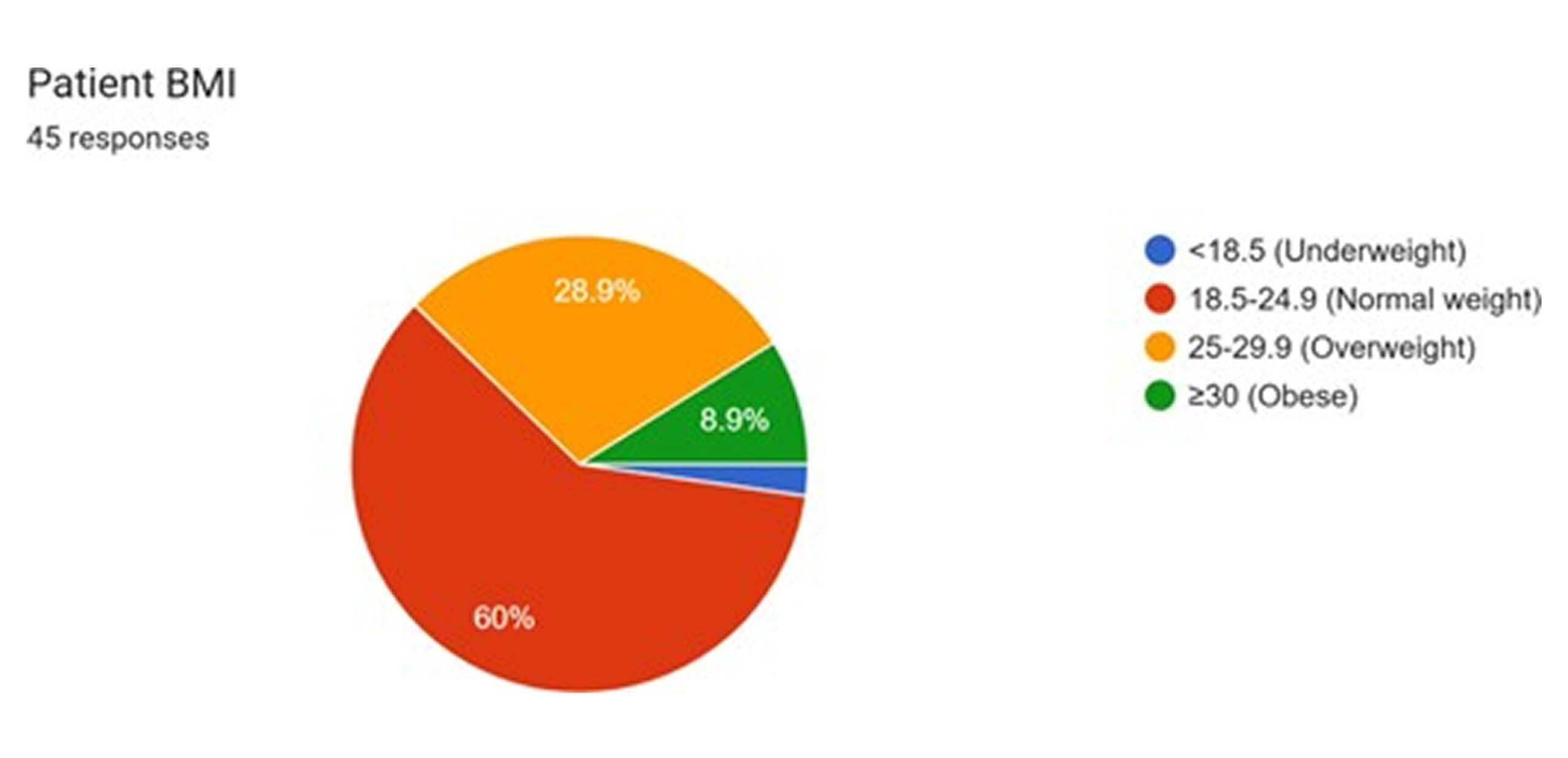
Figure 4: Patient BMI
Data on smoking and alcohol use, available for 53 and 54 patients respectively, indicate that a notable portion of the cohort engages in these behaviors. Current smokers constitute 13.2% of respondents, with a much larger group of former smokers (30.2%). Regular alcohol use was reported by 9.3% of patients, with an additional 27.8% reporting occasional use.[12,13] Both smoking and chronic alcohol consumption are known to impact surgical outcomes negatively. Smoking impairs wound healing by causing vasoconstriction and reducing oxygen delivery to tissues, while both can affect nutritional status and immune function.[14]
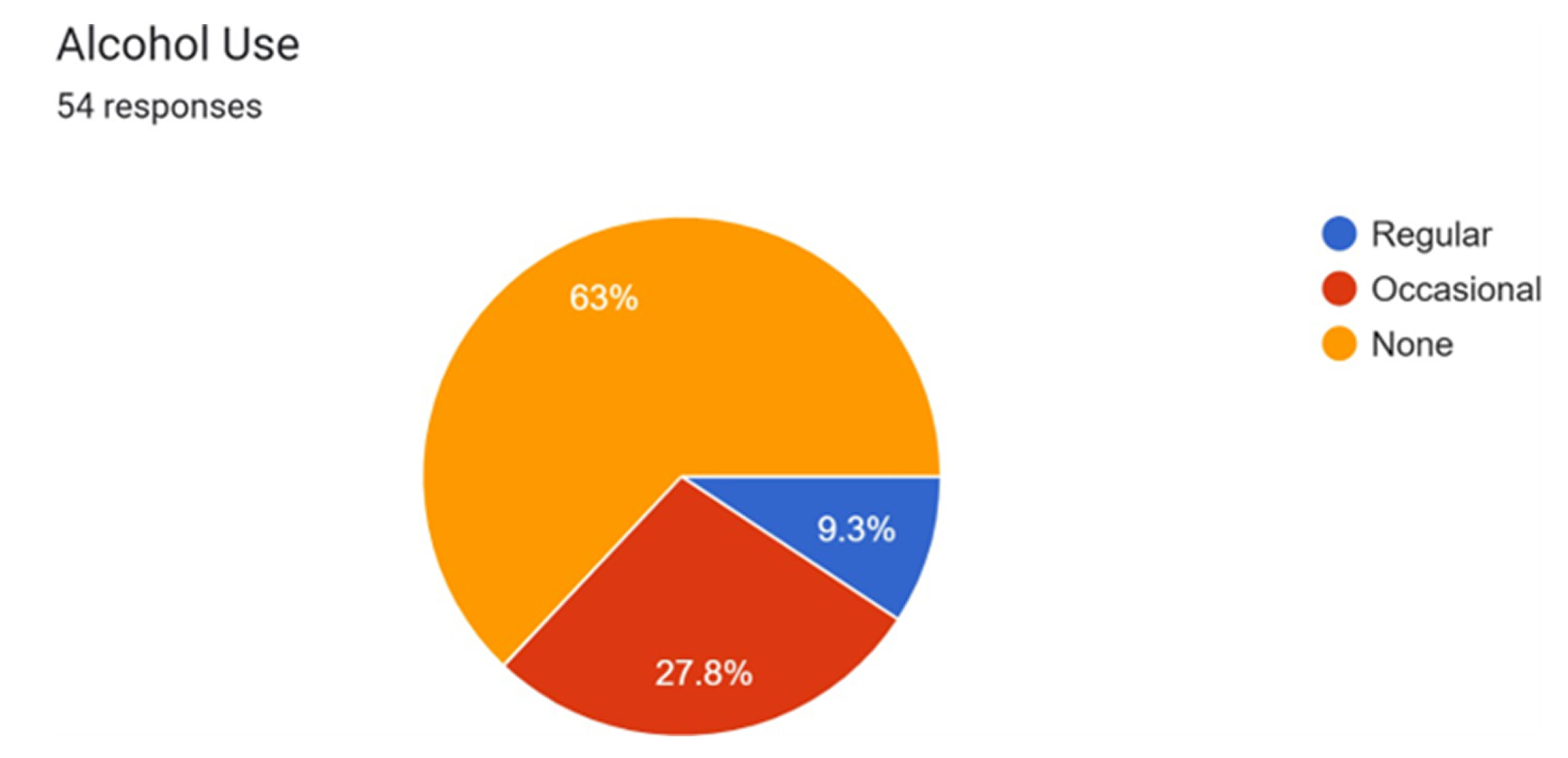
Figure 5: Alcohol use
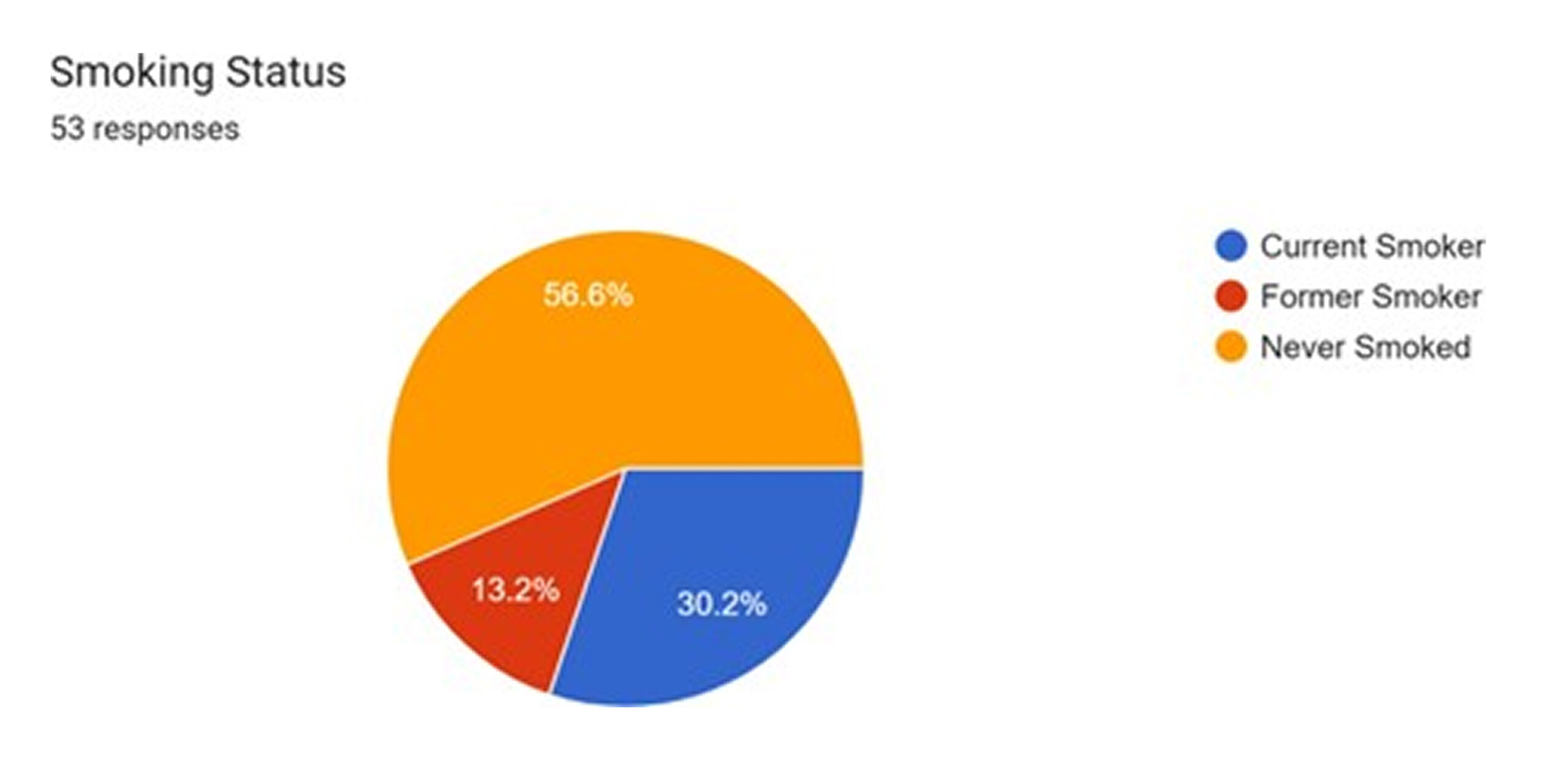
Figure 6: Smoking status
| Characteristic | Category | Frequency (n) | Percentage (%) | Source |
| Gender | Male | 27 | 48.2 | 1 |
| Female | 29 | 51.8 | 1 | |
| Age (years) | 41-64 | 14 | 25.0 | 1 |
| 65-79 | 26 | 46.4 | 1 | |
| ≥80 | 16 | 28.6 | 1 | |
| BMI (N=45) | <18.5
(Underweight) |
1 | 2.2 | 1 |
| 18.5-24.9
(Normal) |
13 | 28.9 | 1 | |
| 25-29.9
(Overweight) |
27 | 60.0 | 1 | |
| ≥30 (Obese) | 4 | 8.9 | 1 | |
| Comorbidities | Diabetes Mellitus | 27 | 48.2 | 1 |
| Hypertension | 32 | 57.1 | 1 | |
| Chronic Kidney Disease | 10 | 17.9 | 1 | |
| Cardiovascular Disease | 8 | 14.3 | 1 | |
| None Reported | 13 | 23.2 | 1 | |
| Smoking status (N=53) | Current Smoker | 7 | 13.2 | 1 |
| Former Smoker | 16 | 30.2 | 1 | |
| Never Smoked | 30 | 56.6 | 1 | |
| Alcohol use
(N=54) |
Regular | 5 | 9.3 | 1 |
| Occasional | 15 | 27.8 | 1 | |
| None | 34 | 63.0 | 1 |
Table 1: Summary of patient demographics and baseline characteristics (N=56)
Part 2: Injury and surgical intervention profile
Fracture characteristics and mechanism of injury: The questionnaire data show that the majority of fractures were intracapsular (62.5%), with the remaining 37.5% being extracapsular.[1] This distinction is clinically important, as intracapsular fractures disrupt the blood supply to the femoral head, carrying a significantly higher risk of complications such as avascular necrosis (AVN) and nonunion, which often necessitates arthroplasty rather than internal fixation.[2]
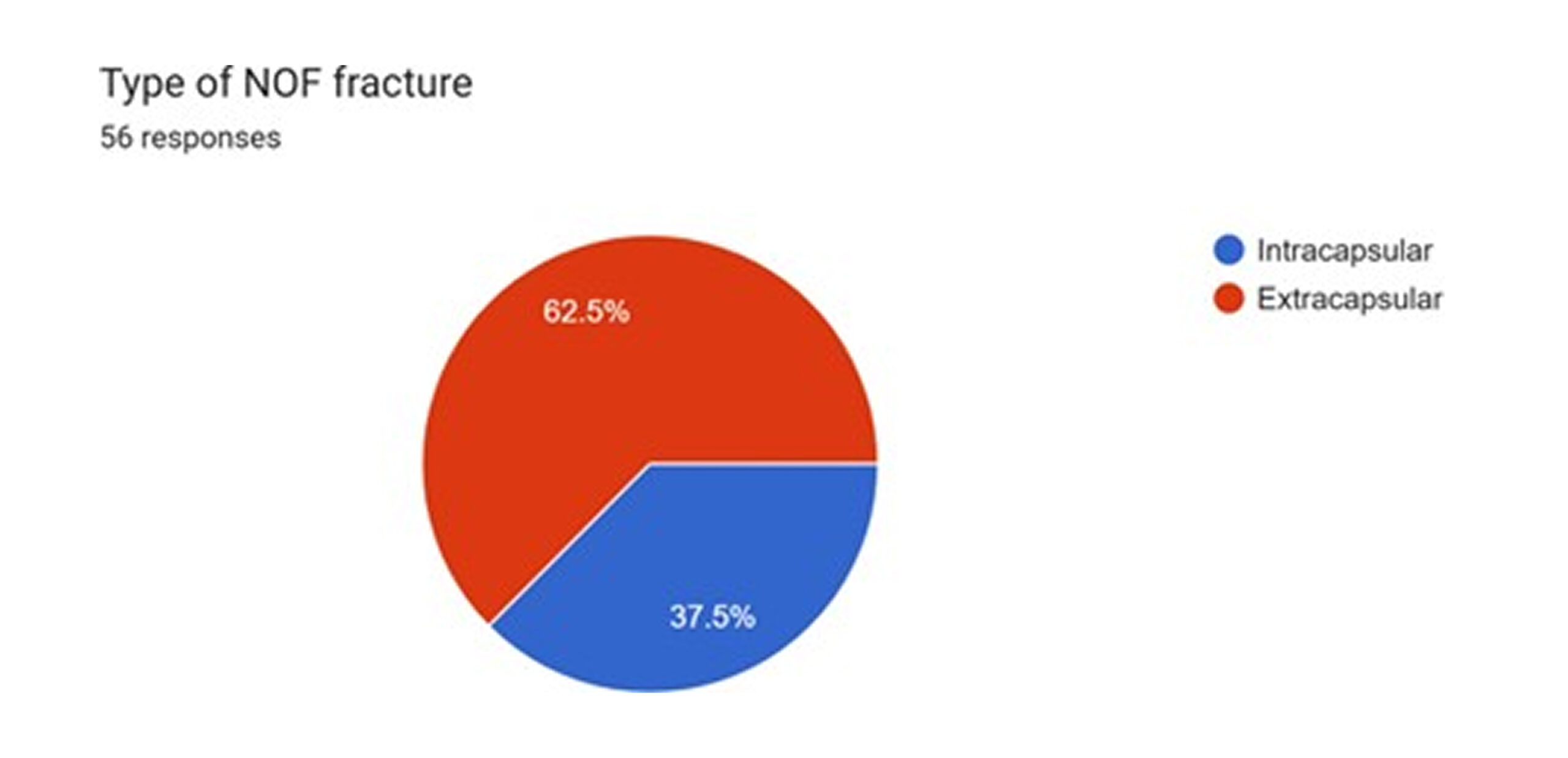
Figure 7: Types of NOF fracture
The predominant mechanism of injury was a fall from standing height, accounting for 92.9% of cases. High-impact trauma, such as from a motor vehicle accident, was responsible for only 7.1% of fractures.[1] This paper strongly reinforces the characterization of this cohort’s injuries as fragility fractures, which are hallmarks of osteoporosis in the elderly population and are consistent with the demographic profile previously established.[2]
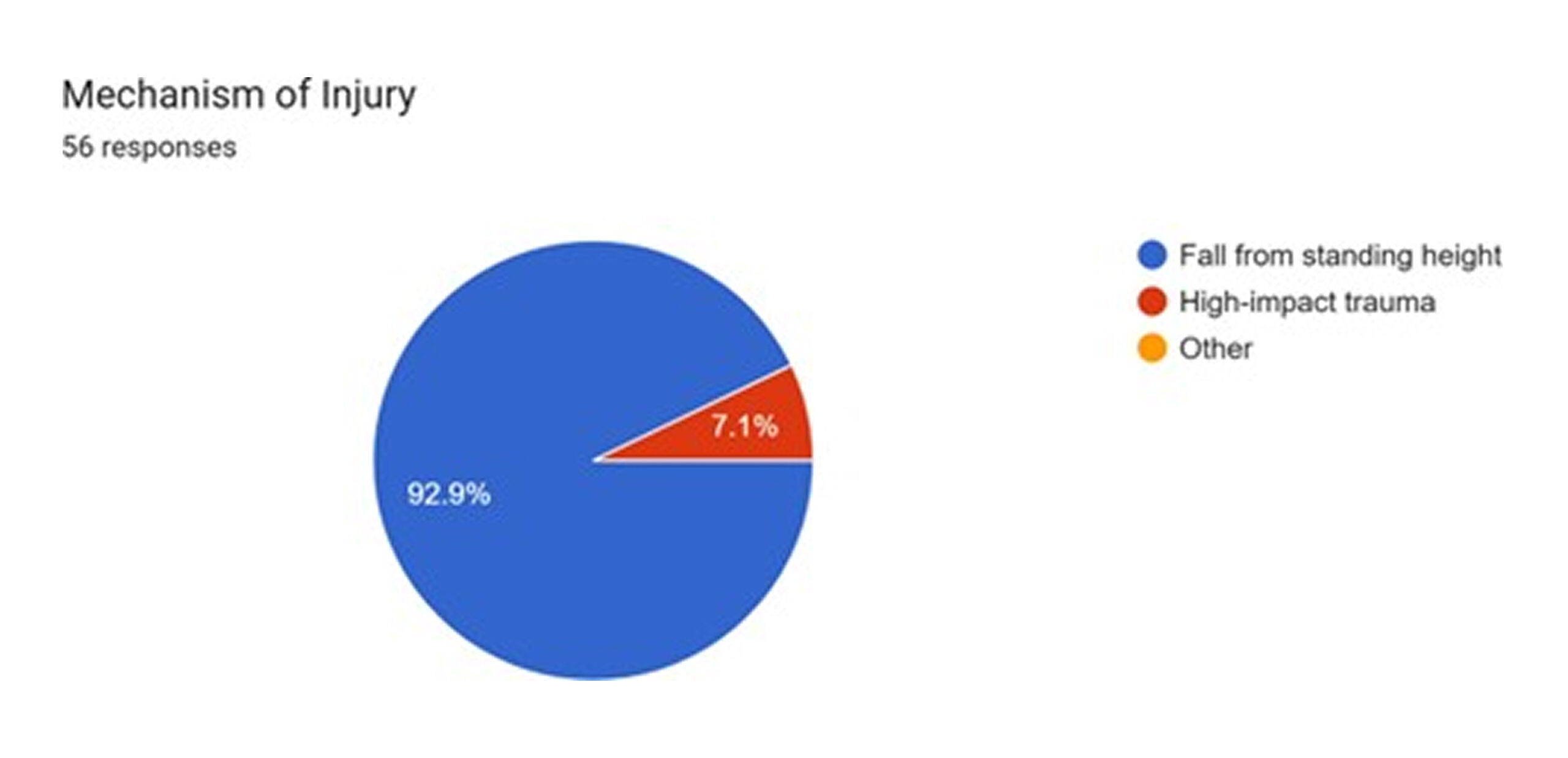
Figure 8: Mechanism of injury
Surgical procedures and perioperative timelines: The choice of surgical procedure reflects the fracture patterns and patient demographics. Internal fixation with the use of rods and screws was the most frequently performed operation, used in 60.7% of cases, followed by Hemiarthroplasty in 26.8%.[1] These arthroplasty procedures are typically indicated for displaced intracapsular fractures in the elderly. Analysis of the free-text responses for the surgical procedure provides crucial additional detail. The mention of “DHS” (Dynamic Hip Screw) and similar terms in 7.2% of cases indicates that internal fixation was used, which is appropriate for extracapsular fractures.[1]
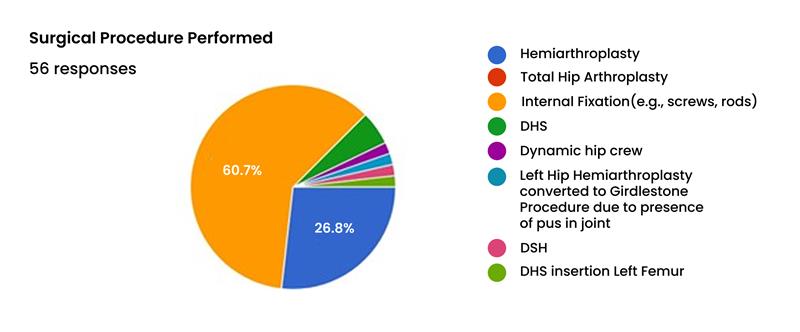
Figure 9: Surgical procedure performed
Most critically, “Left Hip Hemiarthroplasty converted to Girdlestone Proc…”.[1] A Girdlestone procedure, or excision arthroplasty, involves the removal of the femoral head and neck without replacement. It is a salvage operation of last resort, typically performed in cases of severe, uncontrolled periprosthetic infection where revision arthroplasty is not feasible.[15] The presence of even a single Girdlestone procedure (1.8% of the cohort) is a sentinel event, signaling a catastrophic infectious complication that resulted in a functionally devastating outcome for the patient.
Regarding timelines, the majority of patients (71.4%) received their surgery within 48 hours of admission. A further 23.2% were operated on between 24 and 48 hours, while 5.4% waited less than 24 hours.[16,17] Adherence to a <48-hour timeline for surgery is a key quality indicator in hip fracture care, as delays are associated with increased pain, higher risk of complications like pressure sores and pneumonia, and potentially increased mortality.[18] While the performance for most patients appears to align with best practice, the delay experienced by over a quarter of the cohort is a point of concern. The duration of surgery was highly variable, with recorded times ranging. A detailed examination of surgical interventions demonstrates a substantial commitment to the prompt execution of procedures subsequent to patient admission. A considerable proportion, specifically 71.4%, of all surgical operations were conducted within the initial 24-hour timeframe following admission. This underscores an effective framework for urgent and critical cases, prioritizing swift surgical action to enhance patient prognosis and mitigate the length of hospitalizations, potentially.
Moreover, an additional 23.2% of surgical procedures were undertaken within the subsequent 24-hour interval, equating to between 24- and 48-hours post-admission. This indicates that, while not immediate, these cases were nevertheless addressed within a comparatively brief period, likely encompassing elective procedures or those necessitating a short duration of pre-operative stabilization or diagnostic evaluation.
Only a minor segment of patients, precisely 5.4%, experienced a deferment exceeding 48 hours for their surgical intervention. This diminutive percentage implies that protracted delays are uncommon, presumably reserved for exceptionally intricate cases requiring extensive pre-operative preparation, transference to specialized institutions, or instances where patient co-morbidities mandate a more circumspect approach to surgery. The efficacy in performing the vast majority of surgeries within the initial 48 hours highlights a proficiently managed and adaptive surgical department. From just over one hour to more than four hours, reflecting the complexity of individual cases and potentially other systemic factors.[1]
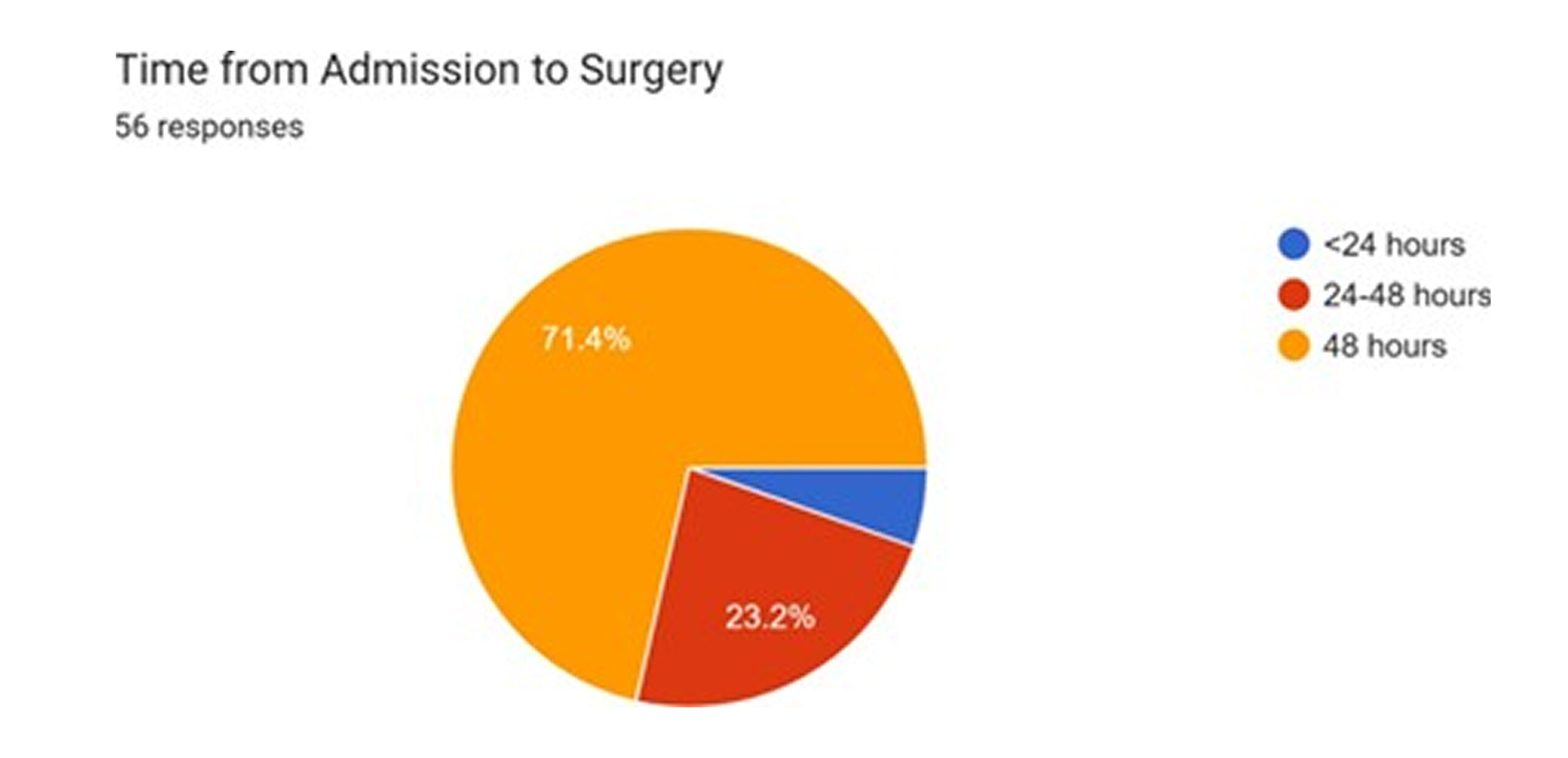
Figure 10: Time from admission to surgery
Analysis of intraoperative practices: The questionnaire reveals that no surgical drain was used in any of the 56 cases. This practice aligns with current evidence-based guidelines, which suggest that routine drain placement after hip fracture surgery provides no significant benefit in preventing hematoma or reducing SSI rates. In fact, drains may increase the need for blood transfusions and can act as a retrograde portal of entry for bacteria, potentially increasing infection risk. This finding indicates clinical practice that is consistent with current recommendations.[19,20]
Second, the data on antibiotic prophylaxis, while indicating widespread use, show variability and potential gaps. Of the 52 responses, Cefazolin was the most commonly specified agent. (32.7%), with others, including Flucloxacillin and Ceftriaxone, are also mentioned.[21,22] The use of a first-generation cephalosporin like Cefazolin is a standard and appropriate choice for prophylaxis against the most common pathogen in orthopedic surgery, Methicillin-Sensitive Staphylococcus aureus (MSSA). However, its effectiveness is entirely dependent on the local prevalence of Methicillin-Resistant S. aureus (MRSA), against which it offers no coverage.[23] Given that recent data from Fiji’s main tertiary hospital suggests an MRSA rate of 11-13%, a significant portion of patients may be receiving suboptimal prophylaxis.[24] The inconsistent reporting of dose, timing, and duration in the questionnaire responses also points to a potential lack of standardization in this critical preventive measure.[1]
Postoperative course and outcomes
Postoperative mobilization and length of hospital stay: The data on postoperative mobilization reveal significant delays. Only a minority of patients (24.4%) were mobilized within the first 24 hours post-surgery. A larger group (28.9%) was mobilized at 48 hours, while a combined majority of 51.1% experienced delays of 72 hours or more.1 Early mobilization, ideally on the day of or the day after surgery, is a cornerstone of modern hip fracture care. It is crucial for preventing a cascade of immobility-related complications, including deep vein thrombosis (DVT), pulmonary embolism (PE), pneumonia, and pressure ulcers. Furthermore, delayed mobilization is strongly associated with poorer functional outcomes, increased length of stay (LOS), and higher mortality.[25] The observed delays in this cohort are therefore a major concern and likely contribute to adverse outcomes.
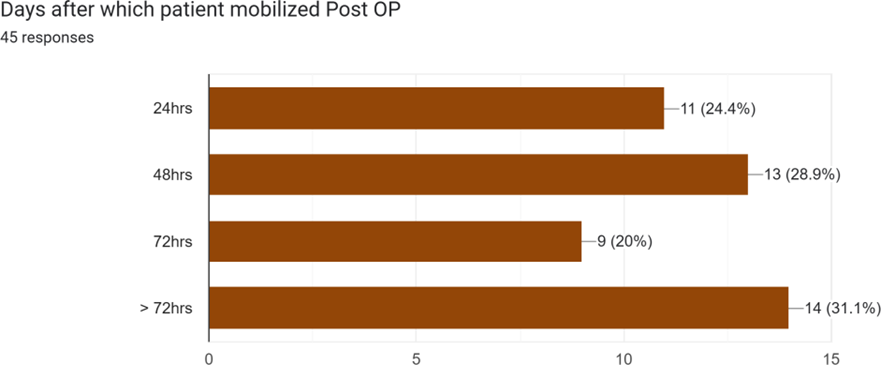
Figure 11: Days after which patient mobilized post OP
The length of hospital stay was highly variable, reflecting the heterogeneity of patient recovery trajectories. While some patients were discharged in as few as 2-3 days, others had prolonged stays, with responses including “16 days,” “25 DAYS,” “43 days,” and “56 days.[1] Such extended hospitalizations are often indicative of postoperative complications, such as the SSIs being investigated, and place a substantial burden on healthcare resources.
Incidence and classification of SSIs: A surgical site infection was documented in 8.9% of the cohort (5 out of 56 patients).[1] This rate is a key finding of the study and will be benchmarked against national and international data in Part II of this report.
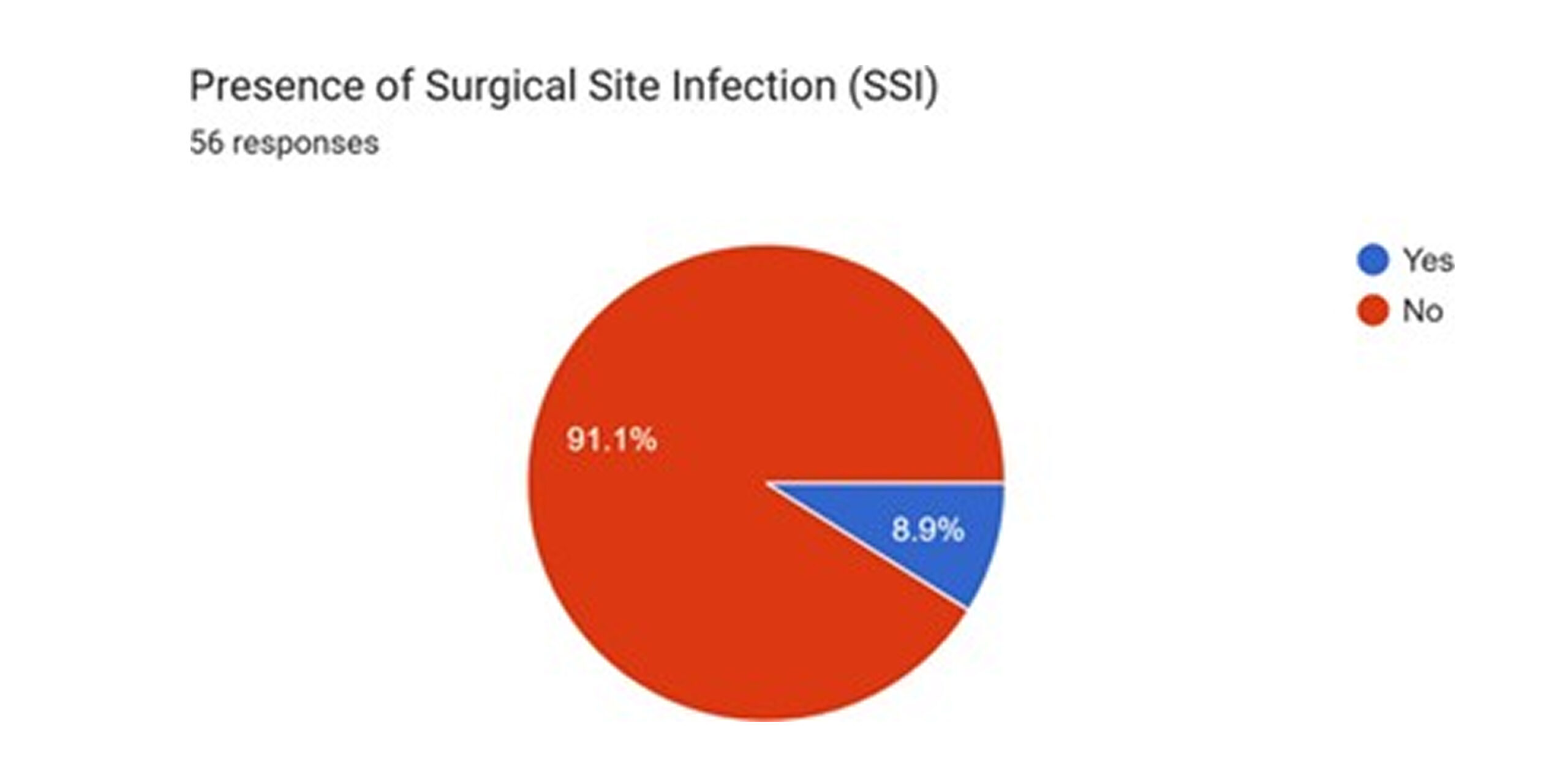
Figure 12: Presence of SSI
When the type of SSI was specified (based on 34 responses, many of which were “NA” for the non-infected patients), the majority were classified as infection involving bone and cartilage (64.7% of those with SSI). However, there was also one case of Deep Incisional SSI (8.8%) reported.1 Further reveals a more severe picture. One response describes osteomyelitis infection that is difficult to treat and can lead to implant failure.[1] This, combined with the previously noted Girdlestone procedure, indicates that the 8.9% overall rate may underrepresent the clinical severity and impact of the infections that are occurring.
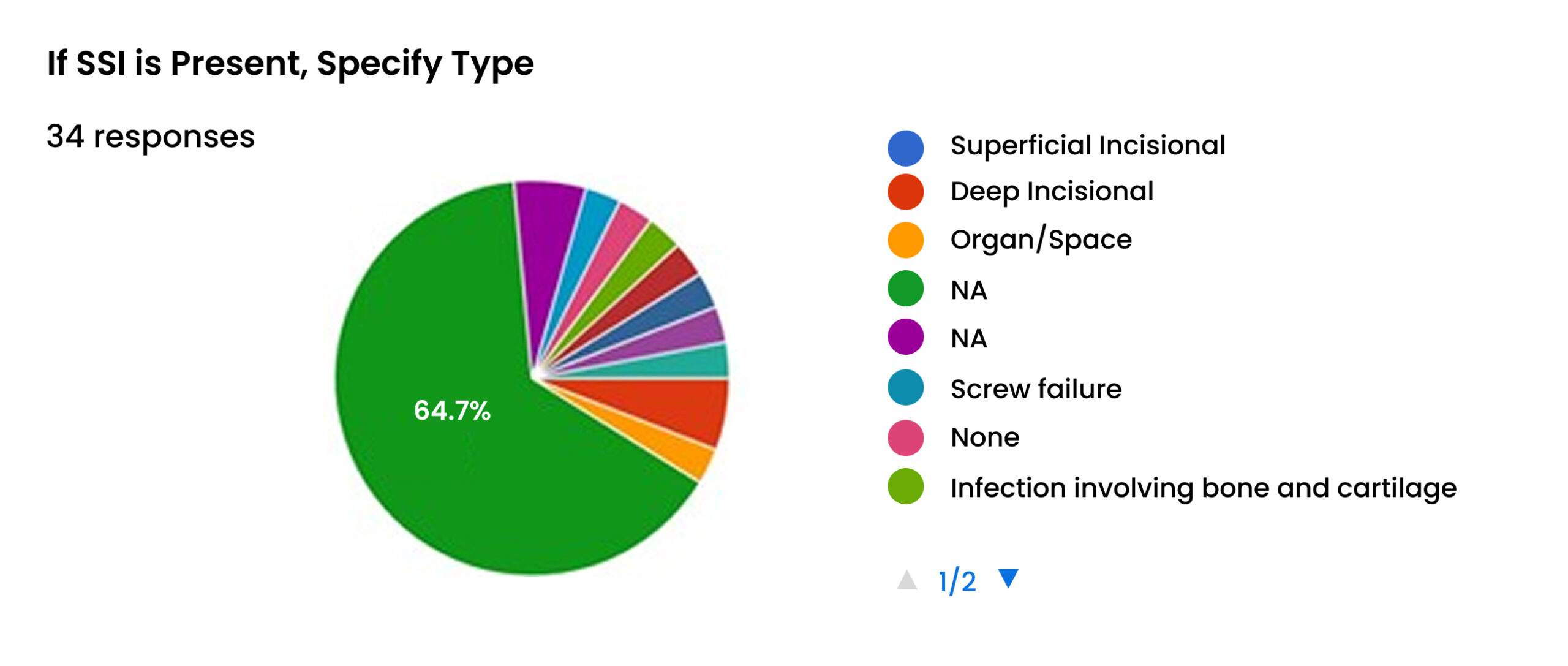
Figure 13: Specific type of SSI
Microbiology of identified infections: Microbiological data, where available, point overwhelmingly to Staphylococcus aureus as the primary causative pathogen, identified in 75% of cases with a specified organism.[1] This is consistent with its status as the leading cause of SSIs in orthopedic surgery worldwide.[26]
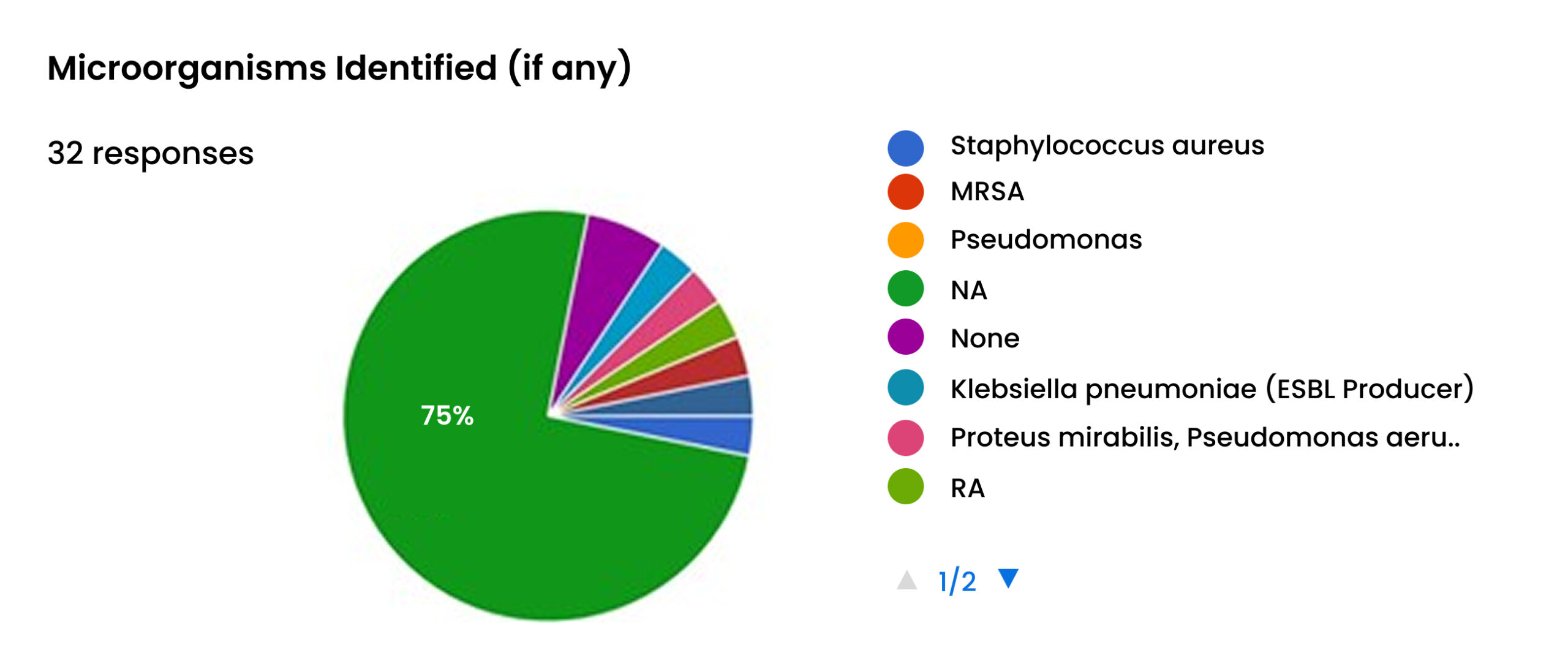
Figure 14: Microorganisms identified
Critically, the data also provides evidence of antimicrobial resistance (AMR). One case of MRSA was explicitly identified, along with one case of Pseudomonas and one polymicrobial infection involving Proteus mirabilis and an ESBL (Extended-Spectrum-Beta-Lactamase)-producing Klebsiella pneumoniae.[1] The presence of these highly resistant organisms, even within this small sample, is a significant red flag. It aligns with broader regional concerns about rising AMR in the Pacific and suggests that standard empiric antibiotic regimens may be failing in a growing number of cases.[24]
Management and complications of SSI: The management of SSIs in the cohort involved a range of interventions, including courses of antibiotics, surgical debridement of the wound, and, in at least one case, the application of a vacuum dressing to promote healing.[26,27] These interventions highlight the additional healthcare resources consumed by SSIs.
The questionnaire also captured other postoperative complications that occurred in the cohort. The most frequently cited complication was Delayed Wound Healing, reported in 64.7% of the relevant responses. However, more severe systemic complications were also noted, including Deep Vein Thrombosis (8.8%), Pneumonia (8.8%), and Implant Failure (8.8%).[28] These complications illustrate the significant morbidity associated with SSIs, which can lead to a cascade of further adverse events, prolonged hospitalization, poor functional outcomes, and substantially increased healthcare costs.[29] The Girdlestone procedure, as the ultimate management for an uncontrolled infection represents the most severe end of this complication spectrum.[1]
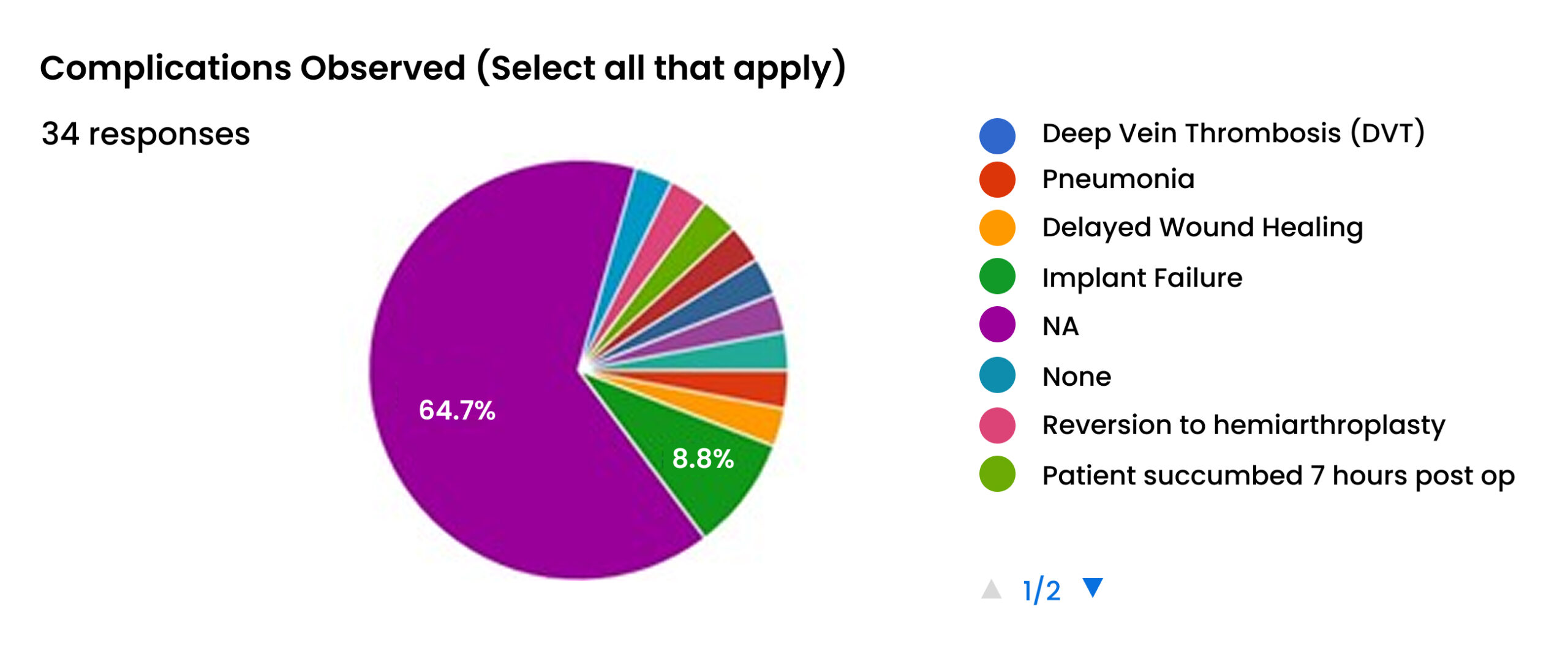
Figure 15: Complications observed
| Outcome metric | Finding / Frequency | Percentage (%) | Source |
| Overall SSI rate | 5 / 56 patients | 8.9 | 1 |
| SSI type (among infected) | Superficial Incisional | 11 / 17 responses* | 64.7 |
| Deep Incisional | 1 / 17 responses* | 8.8 | |
| Organ/Space (Osteomyelitis) | 1 case implied** | – | |
| Causative organisms (among infected) | Staphylococcus aureus | 3 / 4 responses*** | 75.0 |
| MRSA | 1 case noted | – | |
| Polymicrobial (ESBL
Klebsiella) |
1 case noted | – | |
| Management of SSI | Antibiotics, Debridement, Vacuum Dressing, | Multiple instances | – |
| Girdlestone Procedure | |||
| Associated complications | Delayed Wound Healing | 11 / 17 responses* | 64.7 |
| Implant Failure | 3 / 34
responses**** |
8.8 | |
| Deep Vein Thrombosis (DVT) | 3 / 34
responses**** |
8.8 | |
| Pneumonia | 3 / 34
responses**** |
8.8 |
Table 2: Summary of surgical site infection outcomes and management
Discussion
This section transitions from the presentation of data to its interpretation, synthesizing the questionnaire findings with the extensive body of provided research to construct a comprehensive analysis of the drivers, context, and consequences of surgical site infections in Fiji.
Benchmarking the 8.9% SSI Rate: A critical appraisal
The observed SSI rate of 8.9% in this cohort of 56 NOF fracture patients is a finding of considerable clinical and public health significance.[1] To understand its true weight, this figure must be benchmarked against established international and regional data. Globally, SSI rates in orthopaedic surgery vary dramatically, particularly between high-income countries (HICs) and low- and middle-income countries (LMICs). A systematic review indicates that while cumulative SSI incidence in the USA may be around 2.6 per 100 procedures, the range in developing countries is far broader and higher, from 2.5% to as much as 41.9%.[30]
Within this wide spectrum, the 8.9% rate found in the Fijian cohort appears moderate. However, when focusing specifically on the more comparable sub-category of hip fracture surgery, the rate appears alarmingly high. For instance, a large study of proximal femoral fractures reported a total SSI rate of 2.8%.[31] Another study focusing on femoral neck fractures found an incidence of 3.67%, and a study of hip hemiarthroplasty in an elderly Asian population—a cohort very similar to this one—reported a rate of 4.3%.[32,33] A recent UK-based study documented an overall SSI rate of 4.3% in NOF surgeries.[34]
This comparison demonstrates that the 8.9% rate is approximately double the rate reported in several large, comparable international series. It is not merely a number but a clear signal of underlying systemic issues contributing to preventable patient harm. While it falls short of the most severe rates reported in some LMIC settings, it is substantially higher than achievable standards of care for this common and high-stakes procedure. This elevated rate represents a significant and urgent target for quality improvement initiatives within the Fijian healthcare system.
The clinical spectrum of infection: Beyond superficial complications
Focusing solely on the aggregate 8.9% incidence rate risks masking the true clinical severity of these infections. The data from the questionnaire reveals a spectrum of infections that extends far beyond simple superficial wound problems.[1] While most of the specified infections were superficial, the documentation of at least one deep incisional SSI, one case strongly suggestive of osteomyelitis, and, most strikingly, one Girdlestone procedure, presents a much graver picture.[35]
| Study/Region | Surgical procedure | SSI rate (%) | Source |
| Current study (Fiji) | NOF fractures | 8.9 | |
| Asia (Multi-center) | Proximal femoral fractures | 2.8 | 31 |
| China | Femoral neck fractures | 3.67 | 32 |
| Asia (Single institution) | Hip hemiarthroplasty | 4.3 | 33 |
| United Kingdom | NOF fractures | 4.3 | 34 |
| Developing Countries (General) | Orthopedic surgery | 2.5 – 41.9 | 30 |
| Turkey (Single institution) | Orthopedic surgery | 22.7 | 26 |
| USA (Nationwide) | All surgical procedures | 2.6 | 30 |
Table 3: Comparative incidence of SSI in hip fracture surgery
The Impact of endemic comorbidities: Diabetes and obesity as primary catalysts
The analysis reveals a profound synergy between Fiji’s national non-communicable disease (NCD) crisis and the incidence of postoperative infections in this cohort. The patient characteristics are not merely a collection of risk factors; they are a direct reflection of the population’s health profile. The rate of diabetes mellitus in the surgical cohort, at a staggering 48.2%, far surpasses even the high national prevalence estimates for Fiji, which range from 16.6% to 30%.[1] Similarly, the combined overweight and obesity rate of 68.9% among patients with BMI data mirrors the national epidemic of obesity.[36]
These comorbidities are not passive bystanders in the development of SSI. The link is causal and potent. Extensive research and meta-analyses confirm that diabetes significantly increases the risk of SSI, with odds ratios typically in the range of 1.53 to 1.63, effectively increasing a patient’s risk by over 50%.5 Hyperglycemia impairs neutrophil function, compromises microvascular circulation, and creates a glucose-rich environment conducive to bacterial growth, all of which delay wound healing and increase susceptibility to infection.38 Likewise, obesity is a major independent risk factor. The relative hypovascularity of adipose tissue hinders the delivery of prophylactic antibiotics and oxygen to the surgical site, while the chronic inflammatory state associated with obesity can disrupt the normal healing cascade.[37]
This confluence of data leads to a critical conclusion: the orthopedic SSI problem in Fiji is, in large part, a surgical manifestation of the national NCD epidemic. The operating theatre represents a critical nexus where the systemic metabolic dysregulation caused by years of uncontrolled diabetes and obesity manifests as a localized, preventable, and costly surgical complication. This understanding has profound implications for prevention strategies. It suggests that interventions confined solely to the operating theatre and postoperative ward, while necessary, will be insufficient. A truly effective approach must bridge acute hospital care with public health initiatives, placing a heavy emphasis on aggressive preoperative identification and optimization of these high-risk patients.[38]
Perioperative practices under scrutiny: Antibiotics and mobilization
The choice of antibiotic prophylaxis, while appropriate in many cases, may be inadequate for the local microbiological landscape. The primary reliance on Cefazolin is standard for covering MSSA.[39] However, its efficacy is nullified in the presence of MRSA.
S. aureus isolates, with older studies reporting even higher rates.[24] This creates a scenario where more than one in ten patients may be receiving ineffective prophylaxis, a critical failure in the first line of defense against SSI. This highlights a pressing need for robust, local, department-specific antimicrobial surveillance to guide empirical prophylaxis protocols. The significant delays in postoperative mobilization are a major concern. With over half the cohort remaining in bed for 72 hours or longer after surgery, patients are exposed to a heightened risk of a cascade of complications, including pneumonia. These delays could stem from a variety of factors, including inadequate pain control, patient frailty, or systemic issues such as shortages of physiotherapy staff or competing nursing priorities, as hinted at in reports from volunteer organizations.[40-42] Regardless of the cause, this gap between best practice (mobilization within 24 hours) and observed practice represents a critical area for process improvement to enhance recovery and reduce complications.
These findings collectively point not to isolated errors, but to potential systemic issues in the clinical care pathway. They represent ingrained protocols, data deficits, and resource constraints that are tangible, measurable, and, most importantly, addressable through a targeted quality improvement program.[43]
The microbiological landscape: The specter of antimicrobial resistance
The microbiological data from the study, though limited, provides a concerning snapshot of the challenges posed by antimicrobial resistance (AMR) in Fiji. The identification of S. aureus as the pathogen in 75% of cultured infections is expected.[1] The critical issue lies in its resistance paflerns. The explicit notation of one MRSA case and, perhaps more alarmingly, one polymicrobial infection involving an ESBL-producing bacteria. Klebsiella pneumoniae, confirms that multi-drug-resistant organisms (MDROs) are present and causing SSIs in this patient population.[44,45]
This finding must be contextualized within the broader landscape of AMR in Fiji and the Pacific. Molecular epidemiology studies have confirmed the presence of both community-acquired (CC1) and formidable hospital-acquired, multi-drug resistant (CC239) MRSA lineages in Fiji, indicating a complex and dual-front threat.[42] Recent data from CWMH not only confirms an 11-13% MRSA rate but also shows a worrying increase in ESBL production among K. pneumoniae (from 30% in 2019 to 43% in 2022) and the emergence of carbapenem-resistant pathogens.[46-48]
This evidence strongly suggests that the orthopedic department cannot operate in an epidemiological vacuum. Without access to a robust, continuously updated, and department-specific antibiogram, the selection of surgical prophylaxis remains a practice based on historical assumptions rather than current local data. The current standard of care (Cefazolin) is likely failing in a significant and growing percentage of cases. This elevates the risk of SSI and drives the use of broader-spectrum, more expensive second-line antibiotics for treatment, further fueling the cycle of resistance. Establishing a reliable system for local microbiological surveillance is therefore not an academic exercise but a fundamental prerequisite for providing safe and effective surgical care.[48-50]
Economic implications of SSIs for patients and the Fijian health system
The economic consequences of the high SSI rate are profound, impacting the Fijian health system, the national economy, and individual households. Fiji’s health system is already under immense strain from the NCD epidemic, with these diseases consuming a disproportionate share of the health budget and NCD-related mortality costing the nation an estimated 5-10% of its GDP.[51]
Surgical site infections add a significant and largely preventable layer of cost onto this already burdened system. International data from LMIC settings show that the additional cost of treating a single SSI is substantial, ranging from a few thousand to over $29,000 per patient.[29] These costs are driven by multiple factors: prolonged length of hospital stay, the need for revision surgeries and debridement, extended courses of expensive, often imported, broad-spectrum antibiotics. The 8.9% SSI rate observed in this cohort, therefore, represents a significant and continuous drain on the finite resources available. Every infection prevented is a hospital bed freed, scarce foreign exchange saved, and clinical staff time reallocated to other needs.[52-54]
For patients and their families, the economic shock of an SSI can be catastrophic. In a nation where 24.1% of the population lives below the national poverty line and out-of-pocket expenditures for healthcare can be significant, the direct and indirect costs of a complicated hospital stay are often insurmountable.[55] These costs include not only medical bills but also lost wages for the patient and family caregivers, transportation expenses, and the long-term cost of disability. An SSI can easily push a vulnerable family deeper into poverty, creating a vicious cycle of ill health and economic hardship.[52] Therefore, investing in SSI prevention is not merely a clinical quality improvement initiative; it is a potent economic strategy with high returns for the health system, the national economy, and the welfare of the Fijian people.[53]
Strategic recommendations for SSI reduction in Fiji
This section translates the preceding analysis into a multi-level, actionable strategy designed to address the identified drivers of surgical site infections within the specific context of the Fijian healthcare system.[54-56]
Foundational strengthening of infection prevention and control (IPC)
Optimizing national IPC guidelines: From policy to bedside
Fiji has made a commendable step forward by launching its revised national Infection Prevention and Control (IPC) guidelines in 2022, developed in collaboration with regional partners like the Pacific Community (SPC) and WHO.[57] These guidelines, aligned with the Pacific Public Health Surveillance Network (PPHSN) framework, provide a crucial high-level policy foundation.[58,59] However, the most significant challenge, identified not only in Fiji but across the Pacific region, is the effective translation of these comprehensive policy documents into consistent, everyday practice at the patient’s bedside.[60] The gap between policy and implementation is where prevention efforts often fail.
To bridge this gap, a targeted, practical approach is required. It is recommended that a formal gap analysis be conducted, specifically comparing the tenets of the 2022 Fiji National IPC Guideline against the observed, on-the-ground practices within the surgical departments and operating theatres of Lautoka. This initiative should be a collaborative effort. led by the hospital-level IPC compiles, and nursing departments. The primary output of this analysis should not be another report, but rather the development of practical, context-specific Standard Operating Procedures (SOPs), checklists, and visual aids. These tools must be designed for the busy clinical environment, simplifying the core principles of the national guideline into clear, actionable steps for tasks such as preoperative skin preparation, operating room setup, instrument handling, and postoperative wound care. This focus on creating user-friendly implementation tools is essential to move from policy awareness to behavioral change and improved patient outcomes.
Enhancing SSI surveillance: A low-cost, technology-enabled approach
Effective prevention requires robust data, and SSI surveillance is a well-documented area of need across the Pacific.[61,62] Lautoka Hospital has already taken a positive step by piloting a standardized SSI surveillance methodology with support from the SPC and the Doherty Institute, which began in December 2021.[63] However, experiences from similar pilots in the region, such as in the Solomon Islands, highlight persistent challenges, particularly in capturing post-discharge infections and managing data efficiently with limited resources.[64] A significant proportion of SSIs, especially superficial ones, manifest after the patient has left the hospital, leading to a systematic underestimation of the true infection burden if surveillance is purely inpatient-based.[65]
To overcome this, it is recommended to leverage one of Fiji’s most ubiquitous resources: mobile technology. Fiji has high mobile phone penetration, which can be harnessed to create a low-cost, effective post-discharge SSI surveillance system.[66] This system does not require a complex electronic health record. Instead, it can be built using simple, accessible technologies:
- SMS-based follow-up: Automated text messages sent to patients at set intervals (e.g., day 7, 14, and 30 post-op) with simple questions about their wound (e.g., “Is your wound red? Y/N,” “Is there any pus from your wound? Y/N”).
- Dedicated nurse-led telephone calls: A trained IPC or orthopedic nurse can conduct structured telephone interviews with patients’ post-discharge to screen for signs and symptoms of infection.
- Simple mobile imaging: Patients with smartphones can be instructed to send a photo of their wound to a secure number if they have concerns, allowing for remote triage by a clinical professional.
These methods have been shown to be feasible for SSI surveillance in other LMIC settings and directly address the critical gap in post-discharge data capture.[66] This technology-enabled approach should be integrated with the paper-based, in-hospital surveillance tools already trialed at Lautoka to create a comprehensive, end-to-end surveillance system that is both effective and sustainable within a resource-constrained environment.[67-70]
Human factors engineering in the operating theatre: Designing for safety
The high rate of SSIs cannot be attributed solely to individual lapses; it is often a symptom of system failures. With the significant infrastructure investments made by Aspen Medical at Lautoka Hospital, the focus of system analysis shifts. Rather than addressing basic deficits, the challenge is now to optimize state-of-the-art environments. Even in modern facilities, the intricate workflow, the placement of new equipment, and the processes for managing sterile instruments can create subtle, system-induced error traps if not designed with human factors in mind. Human Factors Engineering (HFE) is a scientific discipline that analyzes the interaction between people, tasks, tools, and the environment to design systems that make it easier to do the right thing and harder to do the wrong thing.[71]
It is recommended that a formal HFE assessment of the orthopaedic operating theatre workflow be conducted at both Lautoka and CWM Hospitals. This is not a fault-finding exercise but a systems analysis. It would involve direct observation of surgical procedures by a trained team (which could include an IPC professional, a senior surgeon, and a senior nurse) to map the entire workflow from patient entry to exit. The goal is to identify latent hazards and inefficiencies that increase the risk of contamination. Based on this assessment, simple, low-cost, high-impact interventions can be designed, such as:
Workflow optimization: Reorganizing the physical layout of the theatre to create a logical, unidirectional flow of staff and equipment, minimizing unnecessary traffic and cross-contamination.
Instrument and supply management: Redesigning surgical instrument trays to be organized in the sequence of use. Using visual management systems (e.g., color-coding, shadow boards) to ensure all necessary supplies are present and correct before the procedure begins.
Visual cues and nudges: Placing highly visible reminders and hand hygiene stations at critical decision points in the workflow (e.g., immediately before touching the patient or sterile field).
Ergonomic design: Ensuring that equipment and furniture are positioned to reduce physical strain and allow for correct sterile technique without awkward maneuvering.
This HFE approach shifts the focus from blaming individuals to redesigning the system for safety. It is a novel but powerful methodology that can uncover and mitigate risks that traditional IPC audits might miss, leading to more resilient and reliable safety practices.
A multi-modal, evidence-based prevention bundle for NOF fracture surgery: To translate evidence into reliable practice, it is strongly recommended that Fiji develop and implement a mandatory, multi-modal “Fiji NOF Fracture SSI Prevention Bundle.” A “bundle” is a structured set of 3-5 high-impact, evidence-based interventions that, when implemented collectively and reliably for every patient, every time, result in significantly better outcomes than when implemented individually.[72] The proposed bundle targets the specific risk factors and practice gaps identified in the Part II analysis.
Preoperative bundle elements:
- Aggressive glycemic control: Given that nearly half the cohort has diabetes, this is the single most important preoperative intervention.[1] A mandatory protocol should be instituted for all patients with known diabetes or who are found to be hyperglycemic on admission. This includes checking HbA1c to assess long-term control and implementing a standardized protocol for perioperative insulin administration to maintain blood glucose levels below a target threshold (e.g., <180-200 mg/dL or <10-11 mmol/L). This directly mitigates a primary driver of infection risk.[6]
- Targeted aureus decolonization: In light of the high prevalence of S. aureus infections in the cohort and the documented presence of MRSA in Fiji, universal preoperative decolonization should be implemented for all NOF fracture patients.[1] This involves a simple, relatively low-cost, high-impact regimen of administering intranasal mupirocin ointment and having the patient wash with chlorhexidine gluconate (CHG) soap or wipes prior to surgery. This measure is strongly recommended by international guidelines to reduce the patient’s own bacterial load, a major source of SSIs.[72]
- Nutritional screening and optimization: Malnutrition is a known risk factor for poor wound healing. All elderly hip fracture patients should undergo a simple nutritional screening on admission. For those identified as malnourished or at risk, preoperative and postoperative oral nutritional supplementation should be provided to improve their physiological capacity to heal.
Intraoperative bundle elements:
- Cessation of routine surgical drain use: A formal clinical directive should be issued to discontinue the routine placement of surgical drains in primary hip arthroplasty for fractures. This aligns with strong evidence of no benefit and potential for harm, and represents an immediate, zero-cost practice improvement that can reduce resource use (e.g., blood transfusions).[20]
- Evidence-based antibiotic prophylaxis: The choice of prophylactic antibiotic must be guided by local data. An urgent priority is the development and dissemination of an orthopedic-specific antibiogram for Lautoka and CWM hospitals. Based on this data, the standard prophylaxis protocol should be updated. If the MRSA rate in orthopedic SSIs is confirmed to be significant (e.g., >10-15%), the protocol should include the addition of an agent with MRSA coverage, such as vancomycin or teicoplanin, for all arthroplasty cases.[23] The protocol must also mandate correct timing (within 60 minutes of incision), appropriate weight-based dosing, and re-dosing for procedures exceeding two half-lives of the antibiotic.
Postoperative bundle elements:
- Protocol-driven early mobilization: To combat the significant delays in mobilization observed in the study, a formal, protocol-driven approach is needed.[1] A standard of care should be established requiring all NOF fracture patients to be mobilized (e.g., sitting out of bed, standing) by a physiotherapist or trained nurse within 24 hours of surgery, unless specific medical contraindications exist. Achieving this will require a review of physiotherapy staffing and weekend coverage, but it is a critical investment to prevent a cascade of costly complications.[25]
- Systematic delirium prevention and management: Given the elderly, frail nature of the patient cohort, a non-pharmacological, multi-component delirium prevention protocol should be implemented as standard care on the orthopedic ward. This is a low-cost, nursing-led intervention that includes strategies such as frequent reorientation, ensuring patients have their glasses and hearing aids, maintaining a normal sleep-wake cycle (e.g., opening blinds during the day, minimizing noise at night), managing pain effectively, and ensuring adequate hydration and nutrition. Early identification and management of delirium can significantly reduce its duration and severity, leading to better outcomes and shorter hospital stays.[73]
| Phase of care | Intervention | Rationale / Evidence source |
| Preoperative | Glycemic control: Standardized protocol to maintain blood glucose
<180 mg/dL. |
Targets the extremely high prevalence of diabetes (48.2%) in the patient cohort, a major independent risk factor for SSI.[1] |
| S. aureus decolonization: Universal nasal mupirocin and CHG body wash. | Addresses the primary pathogen (S. aureus) and the presence of MRSA in Fiji; a high-impact, evidence-based preventive measure.[24] | |
| Nutritional screening & support: Screen all patients and provide supplements if malnourished. | Improves physiological
reserve and wound healing capacity in a frail, elderly population.[14] |
|
| Data-driven antibiotic prophylaxis: Standardize regimen based on a local antibiogram, ensuring MRSA coverage if needed, correct timing, and
re-dosing. |
Addresses the potential for prophylaxis failure due to local AMR pathogens (MRSA, ESBLs) and ensures consistency.[23] | |
| Postoperative | Early mobilization protocol: Mobilize the patient out of bed within 24 hours of surgery. | Combats significant observed delays and prevents a cascade of immobility-related complications (DVT, pneumonia, delirium).[25] |
| Delirium prevention bundle: Implement non-pharmacological strategies (orientation, sleep hygiene, sensory aids). | Proactively manages a common and debilitating complication in this
high-risk elderly population, improving outcomes and reducing LOS.[74] |
Table 4: Proposed evidence-Based SSI prevention bundle for NOF surgery in Fiji
Empowering stakeholders for sustainable change
The successful and lasting reduction of SSIs requires more than just technical interventions; it necessitates a cultural shift that involves and empowers all stakeholders, from patients and their families to frontline clinicians and hospital leadership. The following recommendations are designed to foster this environment of shared responsibility and continuous improvement.[75]
Engaging and educating patients as partners in prevention
Patient engagement is a powerful but often underutilized tool in infection prevention. An informed and empowered patient can become an active participant in their own safety, improving adherence to care protocols and serving as an early warning system for potential complications.[76]
It is recommended to develop a standardized patient engagement program for orthopedic surgery in Fiji. This program should include:
- Culturally appropriate educational materials: Create simple, low-literacy pamphlets and short videos in Fijian, Hindi, and English. These materials should explain in clear terms what an SSI is, the patient’s role in prevention (e.g., preoperative CHG washing, not touching the wound), how to care for their incision and any drains after discharge, and the key signs of infection to watch for (redness, swelling, pus, fever).[77]
- Standardized discharge kits: Upon discharge, every patient should receive a “wound care kit” containing a small supply of necessary dressings, antiseptic wipes, and a copy of the instructional pamphlet. This removes barriers to proper home care.[78]
- Empowerment to “Speak Up”: Patients and their families should be explicitly encouraged and empowered to ask questions and to remind healthcare staff about hand hygiene. A simple campaign with posters saying, “It’s OK to ask: Have you washed your hands?” can help normalize this interaction and reinforce hand hygiene as a shared priority.[79] This active involvement can transform the patient from a passive recipient of care into a vital partner in the safety process.[80]
Developing IPC champions and fostering a safety culture
Top-down mandates for practice change are often met with resistance or transient compliance. Sustainable improvement requires local ownership and peer-to-peer leadership. The concept of identifying and empowering local “champions” is a proven strategy for driving change, particularly in LMIC settings where formal resources may be limited.[81]
An implementation roadmap using the consolidated framework for implementation research (CFIR)
To ensure the successful rollout and sustainability of the SSI Prevention Bundle and associated practice changes, a structured implementation strategy is essential. Simply introducing the recommendations is not enough. The Consolidated Framework for Implementation Research (CFIR) provides a comprehensive, evidence-based roadmap for guiding complex health interventions, ensuring that potential barriers and facilitators are systematically identified and addressed.[82] The CFIR has been successfully adapted for use in LMIC settings and provides a robust structure for this initiative.[83]
It is recommended that the MoHMS and hospital leadership use the five domains of the CFIR to guide the implementation process:
- Intervention characteristics: The SSI Prevention Bundle must be presented clearly, emphasizing its strong evidence base, its low complexity, and its clear relative advantage over current practices in terms of improved patient outcomes and reduced costs.
- Outer setting: The initiative must be aligned with external factors, such as MoHMS national health priorities (e.g., combating NCDs and AMR) and the needs and expectations of the patient population.
- Inner setting: This is a critical domain. Success requires securing visible and sustained support from hospital leadership (Leadership Engagement), creating a Tension for Change by clearly communicating the unacceptably high SSI rate and its consequences, allocating the necessary Available Resources (e.g., ensuring a reliable supply of mupirocin and CHG), and leveraging the newly appointed IPC Champions.
- Characteristics of individuals: The strategy must address the knowledge, beliefs, and self-efficacy of frontline staff through targeted training, addressing any misconceptions (e.g., about the utility of drains), and empowering them to adopt the new practices.
- Process: The implementation should be planned and executed systematically. This involves engaging all stakeholders early, executing the plan, starting with a well-supported pilot on a single orthopedic ward. Reflecting on the pilot data and feedback to refine the process, and then scaling up the initiative across the hospital.
Using a recognized implementation science framework like the CFIR provides a structured, thoughtful, and evidence-based methodology that moves beyond simply “what to do” and provides a clear roadmap for “how to do it,” dramatically increasing the probability of achieving a sustained reduction in surgical site infections in Fiji.
| CFIR domain | Key construct | Actionable strategy for Fiji |
| Intervention characteristics | Relative advantage | Clearly articulate the benefits of the SSI bundle over current practice, focusing on improved patient outcomes (lower morbidity, mortality) and economic benefits (reduced LOS, lower costs). |
| Complexity | Present the bundle as a simple, clear checklist of 6-7 core actions. Develop
easy-to-use visual aids and pocket cards for clinicians. |
|
| Outer setting | Patient needs & resources. | Develop patient education materials in local languages. Align the initiative with the national |
| Goal of reducing the burden of NCDs and AMR. | ||
| Inner setting | Leadership engagement | Present a formal business case to hospital and MoHMS leadership, highlighting the clinical and economic impact of the current SSI rate and the return on investment from prevention. |
| Available resources | Conduct a resource audit to ensure a reliable supply chain for bundle components (e.g., mupirocin, CHG, specific antibiotics) and physiotherapy staffing.
Secure a dedicated budget line item. |
|
| Champions | Formally appoint and train multidisciplinary IPC champions within the orthopedic department to lead peer education, conduct audits, and provide feedback. | |
| Characteristics of individuals | Knowledge & beliefs | Conduct targeted training sessions for all orthopedic staff (surgeons, nurses, anesthetists, physiotherapists) to explain the evidence behind each bundle element, particularly the rationale for discontinuing drains and using specific antibiotics. |
| Process | Planning & executing | Develop a detailed project
plan with a clear timeline. Launch the bundle as a formal Quality Improvement (QI) project, starting with a pilot on one orthopedic ward to test and refine the process before hospital-wide rollout. |
| Reflecting & evaluating | Implement the low-cost SSI surveillance system to track the SSI rate before and after implementation.
Regularly review compliance data from champion-led audits and provide feedback to staff to drive continuous improvement. |
Table 5: CFIR-based implementation strategy for SSI prevention initiatives
Conclusion
This comprehensive analysis, founded on a retrospective study of 56 neck of femur fracture patients and contextualized by extensive research on the Fijian healthcare system, reveals that surgical site infection is a significant, complex, and costly problem. The observed SSI rate of 8.9% is alarmingly high compared to achievable international benchmarks for this procedure and is associated with severe clinical outcomes, including deep infection, osteomyelitis, and functionally devastating salvage surgery.
The investigation identifies a multifactorial etiology for this high infection rate, rooted in three interconnected domains. First, the patient population itself carries an exceptionally high baseline risk, driven by Fiji’s national epidemic of non-communicable diseases; the staggering prevalence of diabetes (48.2%) and obesity (68.9%) in the surgical cohort are primary physiological drivers of infection susceptibility. Second, several perioperative clinical practices appear to be misaligned with current evidence-based medicine, including the universal use of surgical drains, potentially inadequate antibiotic prophylaxis due to emerging antimicrobial resistance, and significant delays in postoperative mobilization. Third, these clinical issues are exacerbated by systemic challenges within the healthcare environment, including foundational resource and infrastructure deficits that persist despite high-level modernization efforts, and a reliance on external teams that have not yet translated into sustainable, independent local capacity.
The economic consequences of this high SSI rate are profound, imposing a preventable burden on a health system already strained by NCDs and inflicting potentially catastrophic financial hardship on vulnerable patients and their families.
However, this analysis also reveals that the problem is addressable. The drivers of infection are identifiable and modifiable. A sustained reduction in SSIs is achievable through a concerted, multi-modal strategy that is tailored to the Fijian context. This strategy must be built on a foundation of strengthened Infection Prevention and Control systems, including the practical implementation of national guidelines and the establishment of robust, low-cost surveillance. The core of the clinical intervention should be the mandatory implementation of an evidence-based “SSI Prevention Bundle” for all hip fracture surgeries, targeting key risk factors in the preoperative, intraoperative, and postoperative phases. Finally, for this change to be sustainable, it must be driven by empowered local stakeholders, including IPC champions on the frontlines and engaged patients who are treated as active partners in their own safety. By adopting this structured, evidence-based, and contextually aware approach, Fiji can significantly reduce the burden of surgical site infections, improving patient outcomes, enhancing healthcare quality, and ensuring a more efficient and equitable use of its vital healthcare resources.
References
- Femoral neck fractures. Orthobullets. Updated May 2025.
Femoral Neck Fractures - Chen Y, Liang S, Wu H, et al. Postoperative delirium in geriatric patients with hip fractures. Front Aging Neurosci. 2022;14:1068278. doi:10.3389/fnagi.2022.1068278
Crossref | Google Scholar - Yang Y, Zhao X, Dong T, Yang Z, Zhang Q, Zhang Y. Risk factors for postoperative delirium following hip fracture repair in elderly patients: a systematic review and meta-analysis. Aging Clin Exp Res. 2017;29(2):115-126. doi:10.1007/s40520-016-0541-6
PubMed | Crossref | Google Scholar - He C, Zhou F, Zhou F, Wang J, Huang W. Impact of type 2 diabetes on surgical site infections and prognosis post orthopaedic surgery: A systematic review and meta-analysis. Int Wound J. 2023. doi:10.1111/iwj.14422
PubMed | Crossref | Google Scholar - Martin ET, Kaye KS, Knott C, et al. Diabetes and Risk of Surgical Site Infection: A Systematic Review and Meta-analysis. Infect Control Hosp Epidemiol. 2016;37(1):88-99. doi:10.1017/ice.2015.249
PubMed | Crossref | Google Scholar - Yang J, Zhang X, Liang W. A retrospective analysis of factors affecting surgical site infection in orthopaedic patients. J Int Med Res. 2020;48(4):300060520907776. doi:10.1177/0300060520907776
PubMed | Crossref | Google Scholar - Global Nutrition Report. Fiji nutrition profile. 2025.
Global Nutrition Report - Ministry of Health & Medical Services. What is obesity? MHMS Fiji.
What is obesity? - Nasokia A. Tackling the obesity epidemic. Fiji Times. Updated April 15, 2024.
Tackling the obesity epidemic - Specialty Wound Care. How does obesity impact wound healing? Specialty Wound Care. Published April 7, 2025.
How Obesity Affects Wound Healing? - Yang Y, Zhao Z, Wang Y, Gao Y, Sun H, Liu W. Impact of wound complications in obese versus non-obese patients undergoing total hip arthroplasty: A meta-analysis. Int Wound J. 2023;20(10):4200-4207. doi:10.1111/iwj.14318
PubMed | Crossref | Google Scholar - Hawes J, Hernandez C, Gowen L, Wagner R. Evaluation of fat distribution as a potential risk factor for infection and wound healing complications in total hip arthroplasty. J Am Osteopath Acad Orthop. 2025;Volume IV, Number 1.
Evaluation of Fat Distribution as a Potential Risk Factor for Infection and Wound Healing Complicat… - Najjar Y, Saleh M. Orthopedic surgical site infection: incidence, predisposing factors, and prevention. Int J Med Sci Clin Invention. 2017;4(2). doi:10.18535/ijmsci/v4i2.04
Crossref | Google Scholar - Cordero-Ampuero J. Girdlestone procedure: when and why. Hip Int. 2012;22 Suppl 8:S36-39. doi:10.5301/HIP.2012.9568
PubMed | Crossref | Google Scholar - Nester M, Rivera R, Kittur K, Alford N, Huynh TH. Girdlestone resection arthroplasty: indications, outcomes, and complications. Glob J Ortho Res. 2024;4(5).
Girdlestone Resection Arthroplasty: Indications, Outcomes, and Complications - mctlaw®. Girdlestone surgery after hip replacement complications. MCTLaw. Updated November 14, 2025.
Girdlestone Surgery After a Failed Hip Replacement complications - Gaski G. Femoral neck fractures in patients younger than 50 years. Orthopaedic Trauma Association; 2021.
Femoral neck fractures in patients younger than 50 years - Lee SH, Kwak DK, Yoo JH. Surgical drain has no benefits in hemiarthroplasty for femoral neck fractures in elderly patients. Sci Rep. 2023;13(1):21422. doi:10.1038/s41598-023-48799-7
PubMed | Crossref | Google Scholar - Siletz A, Childers CP, Faltermeier C, et al. Surgical Technical Evidence Review of Hip Fracture Surgery Conducted for the AHRQ Safety Program for Improving Surgical Care and Recovery. Geriatr Orthop Surg Rehabil. 2018;9:2151459318769215. doi:10.1177/2151459318769215
PubMed | Crossref | Google Scholar - Bucataru A, Balasoiu M, Ghenea AE, et al. Factors Contributing to Surgical Site Infections: A Comprehensive Systematic Review of Etiology and Risk Factors. Clin Pract. 2023;14(1):52-68. doi:10.3390/clinpract14010006
PubMed | Crossref | Google Scholar - Ahmed S, Zafar Z, Waseem T. To drain or not to drain in thyroidectomy: a meta-analysis of outcomes. Int J Surg. 2020.
To Drain or Not to Drain in Thyroidectomy: A Meta-analysis of Outcomes - Gupta B, Soman KC, Bhoir L, Gadahire M, Patel B, Ahdal J. The burden of methicillin resistant Staphylococcus aureus in surgical site infections: a review. J Clin Diagn Res. 2021;15:DC01-06. doi:10.7860/JCDR/2021/46922.14891
Crossref | Google Scholar - Getahun Strobel A, Prasad P, Lane CR, et al. The changing epidemiology of antimicrobial resistance in Fiji: a descriptive analysis of antimicrobial susceptibility and trends of endemic and emerging pathogens, 2019-2022. Lancet Reg Health West Pac. 2024;45:101036. doi:10.1016/j.lanwpc.2024.101036
PubMed | Crossref | Google Scholar - Horton I, Bourget-Murray J, Buth O, et al. Delayed mobilization following admission for hip fracture is associated with increased morbidity and length of hospital stay. Can J Surg. 2023;66(4):E432-438. doi:10.1503/cjs.006822
PubMed | Crossref | Google Scholar - Maksimović J, Marković-Denić L, Bumbasirević M, Marinković J, Vlajinac H. Surgical site infections in orthopedic patients: prospective cohort study. Croat Med J. 2008;49(1):58-65. doi:10.3325/cmj.2008.1.58
PubMed | Crossref | Google Scholar - Rajkumari N, Gupta AK, Mathur P, et al. Outcomes of surgical site infections in orthopedic trauma surgeries in a tertiary care centre in India. J Postgrad Med. 2014;60(3):254-259. doi:10.4103/0022-3859.138731
PubMed | Crossref | Google Scholar - Foxlee ND, Townell N, McIver L, Lau CL. Antibiotic Resistance in Pacific Island Countries and Territories: A Systematic Scoping Review. Antibiotics (Basel). 2019;8(1):29. doi:10.3390/antibiotics8010029
PubMed | Crossref | Google Scholar - Maraş G, Sürme Y. Surgical site infections: prevalence, economic burden, and new preventive recommendations. Explor Res Hypothesis Med. 2023;8(4):366‑371. doi:10.14218/ERHM.2023.00010
Crossref | Google Scholar - Rammohan N. Surgical site infections: causes and implication on healthcare systems and patients in low resource countries. J Student Res. doi:10.47611/JSRHS.V12I1.3781
Crossref - Silas U, Berberich C, Anyimiah P, et al. Risk of surgical site infection after hip hemiarthroplasty of femoral neck fractures: a systematic review and meta-analysis. Arch Orthop Trauma Surg. 2024;144(8):3685-3695. doi:10.1007/s00402-024-05384-5
Crossref | Google Scholar - Ji C, Zhu Y, Liu S, et al. Incidence and risk of surgical site infection after adult femoral neck fractures treated by surgery: A retrospective case-control study. Medicine (Baltimore). 2019;98(11):e14882. doi:10.1097/MD.0000000000014882
PubMed | Crossref | Google Scholar - Lau AC, Neo GH, Lee HC. Risk factors of surgical site infections in hip hemiarthroplasty: a single-institution experience over nine years. Singapore Med J. 2014;55(10):535-538. doi:10.11622/smedj.2014137
PubMed | Crossref | Google Scholar - Solari F, Joyner C, Dash K, et al. A Prospective Analysis of Surgical Site Infections in the Neck of Femur Fractures in a Trauma Unit: A Six-Year Analysis From 2019 to 2024. Cureus. 2025;17(5):e83545. doi:10.7759/cureus.83545
PubMed | Crossref | Google Scholar - Sheldon J, Campos A, Worsley C, et al. Girdlestone procedure. Reference article. Radiopaedia.org. 2024. doi:10.53347/rID-25401
Crossref - Ministry of Health & Medical Services, Fiji. What is diabetes? MHMS Fiji. Accessed December 30, 2025.
What is diabetes? - International Diabetes Federation. Fiji – IDF member information. IDF.org. Accessed December 30, 2025.
Fiji – International Diabetes Federation - Lee TC, Lee YL, Chen JC, Chen CH, Ho PS. Impact of type 2 diabetes on postoperative outcome after hip fracture: nationwide population-based study in Taiwan. BMJ Open Diabetes Res Care. 2020;8(1):e000843. doi:10.1136/bmjdrc-2019-000843
PubMed | Crossref | Google Scholar - Mashayekhi Y, Amadi‑Livingstone C, Timamy A, et al. Narrative review on the management of neck of femur fractures in people living with HIV: challenges, complications, and long‑term outcomes. Microorganisms. 2025;13(7):1530. doi:10.3390/microorganisms13071530
Crossref | Google Scholar - Patil SS, Suresh KP, Shinduja R, et al. Prevalence of Methicillin-resistant Staphylococcus Aureus in India: A Systematic Review and Meta-analysis. Oman Med J. 2022;37(4):e440. doi:10.5001/omj.2022.22
PubMed | Crossref | Google Scholar - Ministry of Health & Medical Services, Fiji. Lautoka Hospital. MHMS Fiji.
Lautoka Hospital - Jenney A, Holt D, Ritika R, et al. The clinical and molecular epidemiology of Staphylococcus aureus infections in Fiji. BMC Infect Dis. 2014;14:160. doi:10.1186/1471-2334-14-160
PubMed | Crossref | Google Scholar - Loftus MJ, Curtis SJ, Naidu R, et al. Prevalence of healthcare-associated infections and antimicrobial use among inpatients in a tertiary hospital in Fiji: a point prevalence survey. Antimicrob Resist Infect Control. 2020;9(1):146. doi:10.1186/s13756-020-00807-5
PubMed | Crossref | Google Scholar - Aspen Medical. Aspen Medical: comprehensive global healthcare solutions.
Aspen Medical: comprehensive global healthcare solutions - Aspen Medical. First phase of a $11 million upgrade program at Lautoka Hospital commences.
First phase of a $11 million upgrade program at Lautoka Hospital commences - Narayan V. First‑ever open‑heart surgery completed at the Lautoka Hospital under Aspen Medical. FijiVillage.
First-ever open-heart surgery completed at the Lautoka Hospital under Aspen Medical - Orthopaedic Outreach. Fiji. Orthopaedic Outreach Australasia.
Fiji - Stryker MedEd. Argo Registrar Ortho Education Program. Stryker MedEd.
Argo Registrar Ortho Education Program - HealthShare. Orthopaedic Surgeons in Brisbane 4000 QLD. HealthShare Directory.
Orthopaedic Surgeons in Brisbane QLD 4000 - Pacific Islands Orthopaedic Association. Apply for training. Pacific Islands Orthopaedic Association.
Apply for training - Vaitohi B. Inadequate healthcare in Pacific Islands. Ballard Brief. 2025.
Inadequate Healthcare in Pacific Islands - Monahan M, Jowett S, Pinkney T, et al. Surgical site infection and costs in low- and middle-income countries: A systematic review of the economic burden. PLoS One. 2020;15(6):e0232960. doi:10.1371/journal.pone.0232960
PubMed | Crossref | Google Scholar - Monahan M, Jowett S, Pinkney T, et al. Surgical site infection and costs in low- and middle-income countries: A systematic review of the economic burden. PLoS One. 2020;15(6):e0232960. doi:10.1371/journal.pone.0232960
PubMed | Crossref | Google Scholar - Piednoir E, Robert-Yap J, Baillet P, Lermite E, Christou N. The Socioeconomic Impact of Surgical Site Infections. Front Public Health. 2021;9:712461. doi:10.3389/fpubh.2021.712461
PubMed | Crossref | Google Scholar - The Borgen Project. The Links Between Poverty and Diabetes in Fiji. The Borgen Project. 2023.
The Links Between Poverty and Diabetes in Fiji – The Borgen Project - Kidd S. Poverty, vulnerability and social protection in the Pacific: The role of social transfers. AusAID Pacific Social Protection Series. 2012.
Poverty, vulnerability and social protection in the Pacific: The role of social transfers - Medical Dialogues. Health Ministry launches Red Line campaign to curb antibiotic misuse. Medical Dialogues. Published March 30, 2025.
Health Ministry Launches Red Line Campaign to Curb Antibiotic Misuse - Pacific Public Health Surveillance Network. Infection Prevention and Control Guidelines. Published February 2025.
Infection Prevention and Control Guidelines - Pacific Community (SPC). Updated regional infection prevention and control guidelines for the Pacific published. Pacific Community. Published October 6, 2021.
Updated regional infection prevention and control guidelines for the Pacific published - World Health Organization Regional Office for the Western Pacific. Infection prevention and control systems in six priority countries in the Western Pacific Region: regional report 2023. 2025.
Infection prevention and control systems in six priority countries in the Western Pacific Region: r… - Pacific Community (SPC). Technical Paper 6.5: Regional Infection Prevention and Control Updates. Presented at: Pacific Heads of Nursing and Midwifery (PHoNM) Meeting; November 14–16, 2023; Nadi, Fiji.
PHONM 6.5_Technical Paper_Regional IPC Updates - Leong M, Picton R, Wratten M, Mahe A, Zimmerman PA. Baseline evaluation of the World Health Organization (WHO) infection prevention and control (IPC) core components in Pacific Island Countries and Territories (PICTs). Antimicrob Resist Infect Control. 2024;13(1):108. doi:10.1186/s13756-024-01447-9
PubMed | Crossref | Google Scholar - Pacific Community (SPC). Improving healthcare associated infection surveillance. SPC Public Health Updates. Published March 18, 2022.
Improving healthcare associated infection surveillance - Pacific Community (SPC). 2023 Pacific Infection Control Network Meeting. SPC Public Health Division.
2023 Pacific Infection Control Network Meeting - Calderwood MS, Anderson DJ, Bratzler DW, et al. Strategies to prevent surgical site infections in acute-care hospitals: 2022 Update. Infect Control Hosp Epidemiol. 2023;44(5):695-720. doi:10.1017/ice.2023.67
PubMed | Crossref | Google Scholar - Sandberg CEJ, Knight SR, Qureshi AU, Pathak S. Using Telemedicine to Diagnose Surgical Site Infections in Low- and Middle-Income Countries: Systematic Review. JMIR Mhealth Uhealth. 2019;7(8):e13309. doi:10.2196/13309
PubMed | Crossref | Google Scholar - Pennathur PR, Herwaldt LA. Role of Human Factors Engineering in Infection Prevention: Gaps and Opportunities. Curr Treat Options Infect Dis. 2017;9(2):230-249. doi:10.1007/s40506-017-0123-y
PubMed | Crossref | Google Scholar - EPRA International Journal of Health Sciences. Exploring the work‑life balance of faculty through occupational stress and stress coping strategies. EPRA International Journal of Health Sciences. 2025;12(3). doi:10.36713/epra20470
Crossref | Google Scholar - Storr J, Wigglesworth N, Kilpatrick C. Integrating human factors with infection prevention and control. The Health Foundation; 2013.
Integrating Human Factors with Infection and Prevention Control - Gopathoti P, Siddharth V, Jamwal T, et al. A multicentric study on the Organization of Infection Prevention and Control Programme in tertiary care public sector hospitals of India: Perspectives from a Low- and Middle-Income country. J Hosp Infect. doi:10.1016/j.jhin.2025.11.047
PubMed | Crossref | Google Scholar - Arulampalam T, Barach P. Human factors in surgery: optimal surgical team proficiency and decision making. Bull R Coll Surg Engl. 2023;105(3). doi:10.1308/rcsbull.2023.45
Crossref | Google Scholar - World Health Organization. Global guidelines for the prevention of surgical site infection. 2nd ed. WHO; 2018. ISBN: 978‑92‑4‑155047‑5
Global guidelines for the prevention of surgical site infection - Fenta E, Teshome D, Kibret S, et al. Incidence and risk factors of postoperative delirium in elderly surgical patients 2023. Sci Rep. 2025;15(1):1400. doi:10.1038/s41598-024-84554-2
PubMed | Crossref | Google Scholar - Health in Aging Foundation. Ask the expert: prevention and treatment of post‑operative delirium.
Ask the Expert: Prevention and Treatment of Post-Operative Delirium - Mossie A, Regasa T, Neme D, Awoke Z, Zemedkun A, Hailu S. Evidence‑based guideline on management of postoperative delirium in older people for low resource setting: systematic review article. Int J Gen Med. 2022;15:4053‑4065. doi:10.2147/IJGM.S349232
Crossref | Google Scholar - Mirnoto J, Irwan AM. Patient knowledge and participation in preventing surgical site infections: an integrative review. Gulhane Med J. 2025;67(2):58‑66. doi:10.4274/gulhane.galenos.2025.00821
Crossref | Google Scholar - Oliveira MC, Dalcól C, de Carvalho REFL, Poveda VB. Patient participation in surgical site infection prevention: perceptions of nurses, physicians and patients. Rev Esc Enferm USP. 2023;57:e20220459. doi:10.1590/1980-220X-REEUSP-2022-0459en
PubMed | Crossref | Google Scholar - Michail KA, Tsitsi T, Charalambous M. Effect of patient education on surgical site infections rates: a systematic review of the literature. Int J Health Care Manage. 2024. doi:10.1080/20479700.2024.2403893
Crossref | Google Scholar - Centers for Disease Control and Prevention. Patient engagement in infection prevention. Safe Healthcare Blog. Posted March 15, 2023 by Lori Nerbonne, BSN, RN and Marie Moss, MPH, RN, BSN, CIC, CPHQ, FAPIC.
Patient Engagement in Infection Prevention - Cox J, Douglas L, Wemmer V, Kaminsky K. The Role of Patient Engagement in Surgical Site Infection Reduction: A Process Improvement Project. Adv Skin Wound Care. 2023;36(11):599-603. doi:10.1097/ASW.0000000000000055
PubMed | Crossref | Google Scholar - Mehtar S, Wanyoro A, Ogunsola F, et al. Implementation of surgical site infection surveillance in low- and middle-income countries: A position statement for the International Society for Infectious Diseases. Int J Infect Dis. 2020;100:123-131. doi:10.1016/j.ijid.2020.07.021
PubMed | Crossref | Google Scholar - The Consolidated Framework for Implementation Research (CFIR) – Technical Assistance for users of the CFIR framework. CFIR Guide.
The Consolidated Framework for Implementation Research – Technical Assistance for users of the CFIR… - Schweidenback JS, Rangachari P, D’Amato‑Palumbo S, Gladstone JS. Integrating the Consolidated Framework for Implementation Research (CFIR) and tensions into a novel conceptual model for telehealth advancement in healthcare organizations. J Healthc Leadersh. 2024;16:501‑510. doi:10.2147/JHL.S497875
Crossref | Google Scholar - Means AR, Kemp CG, Gwayi-Chore MC, et al. Evaluating and optimizing the consolidated framework for implementation research (CFIR) for use in low- and middle-income countries: a systematic review. Implement Sci. 2020;15(1):17. doi:10.1186/s13012-020-0977-0
PubMed | Crossref | Google Scholar
Acknowledgments
The authors acknowledge the support and supervision provided by Dr. Akhtar Ali and Dr. Rahul Krishna Reddy. The authors also acknowledge Lautoka Hospital (Aspen Medical Fiji) and the Surgical Department at Aspen Lautoka Hospital for facilitating this study and supporting access to relevant records and departmental processes.
Funding
This study was self-funded by the authors and supervisors.
Author Information
Corresponding Author:
Nikil Naveel Chand
Department of Surgery
Lautoka Hospital (Health Care (Fiji) Pte Ltd t/a Aspen Medical), Lautoka, Fiji
Email: nikilchand31@gmail.com
Co-Authors:
Anjali Devi, Shamal Kumar, Raymond Kumar, Shivnesh Shivam Krishna, Susprita Kumar, and Mohammed Moseb Ansar, Akhtar Ali (Supervisor), Rahul Krishna Reddy (Supervisor)
Department of Surgery
Lautoka Hospital (Health Care (Fiji) Pte Ltd t/a Aspen Medical), Lautoka, Fiji
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
Ethical approval for this study was granted by the FHHRERC, Ministry of Health and Medical Services, Fiji, under approval number MI_7_2_2025 (dated 24 April 2025), in conjunction with institutional approval processes at Lautoka Hospital (Aspen Medical Fiji).
Guarantor
None
DOI
Cite this Article
Chand NN, Devi A, Kumar S, et al. Postoperative Surgical Site Infections in Neck of Femur Fracture Surgeries: A Retrospective Study on Incidence, Risk Factors, and Outcomes at Lautoka Hospital. medtigo J Med. 2026;4(1):e3062412. doi:10.63096/medtigo3062412 Crossref









