Author Affiliations
Abstract
Breast cancer remains the most common malignancy among women worldwide, with survival rates now exceeding 90% in high-income countries. However, persistent post-surgical pain (PPSP) following breast cancer surgery remains a significant yet under-recognized complication, affecting up to 60% of patients. This narrative review synthesizes recent evidence on the prevalence, risk factors, pathophysiology, and management of PPSP in breast cancer survivors. The literature highlights that surgical factors such as axillary dissection, nerve injury, and radiotherapy, combined with inadequate acute pain control, contribute substantially to chronic pain syndromes. Regional anesthesia techniques, including thoracic paravertebral blocks and perioperative intravenous lidocaine infusions, show promise in reducing PPSP incidence, though evidence for pharmacologic adjuncts like pregabalin is inconclusive. Beyond pain, symptom clusters including fatigue, nausea, and sleep disturbance significantly affect recovery and quality of life. Emerging research suggests the gut microbiota may influence pain pathways, offering novel preventive avenues. This review emphasizes the need for holistic, multidisciplinary management, incorporating evidence-based nursing, rehabilitation, and psychosocial support. Ongoing high-quality research is essential to develop tailored, patient-centered strategies that effectively prevent and manage PPSP, ensuring that advances in breast cancer survival translate into improved long-term well-being and functional outcomes for survivors.
Keywords
Breast cancer, Persistent post-surgical pain (PPSP), Post-mastectomy pain syndrome (PMPS), Neuropathic pain, Paravertebral block, Nerve injury, Gut microbiota.
Introduction
Breast cancer remains the most common malignancy among women worldwide, accounting for approximately one-quarter of all cancers diagnosed in women and representing a significant source of global morbidity and mortality. With ongoing advances in screening and multimodal treatments, including surgery, chemotherapy, radiotherapy, and targeted therapies, survival rates for breast cancer patients have improved remarkably over the past decades. In high-income countries, the five-year survival rate now exceeds 90%.[1] However, these improvements in survival have brought to the forefront an equally important issue, the burden of treatment-related complications that continue to impact the long-term quality of life for survivors. Among these complications, post-surgical pain has emerged as one of the most common, persistent, and under-recognized sequelae of breast cancer treatment.[2]
Surgical resection, whether through breast-conserving surgery (lumpectomy) or mastectomy, remains the cornerstone of early-stage breast cancer management. Axillary lymph node dissection (ALND) or sentinel lymph node biopsy (SLNB) is often performed concurrently for staging and disease control. Despite refinements in surgical techniques toward more minimally invasive approaches, injury to peripheral nerves such as the intercostobrachial nerve remains a frequent cause of neuropathic pain syndromes.[3] It is now well established that PPSP. It is defined as pain that develops after a surgical procedure and persists for at least two to three months. PPSP is a prevalent complication following breast cancer surgery, with estimates suggesting that 20% to 60% of patients continue to suffer from moderate to severe pain months or even years after their initial operation.[4]
Surgical factors such as the extent of axillary dissection, nerve injury, and post-operative radiotherapy can exacerbate neuropathic pain by promoting tissue fibrosis, muscle contracture, and local edema.[5] Indeed, radiotherapy, despite significant technological advancements such as intensity-modulated radiotherapy (IMRT), continues to pose risks of tissue injury that may compound post-surgical pain and upper limb dysfunction. Moreover, patient-related factors such as younger age, higher body mass index, psychological vulnerability, pre-existing pain conditions, and the severity of acute post-operative pain are well-documented risk factors that modulate pain trajectories.[6]
The relationship between acute post-operative pain and the later development of chronic pain has been described as a dose-response association: inadequately controlled acute pain is an independent predictor for PPSP.[7] This underscores the critical role of perioperative pain management strategies. Multimodal analgesia, including regional anesthesia techniques such as ultrasound-guided thoracic paravertebral block (PVB), has gained attention for its potential to not only provide effective perioperative analgesia but also to mitigate the transition from acute to chronic pain.[8] While meta-analyses and randomized controlled trials (RCTs) suggest that PVB may reduce the incidence of chronic pain at six months post-surgery, methodological limitations and inconsistent findings call for larger, rigorously designed trials to confirm these effects.
Pharmacologic interventions such as perioperative intravenous lidocaine infusion and gabapentinoids like pregabalin have also been investigated as potential strategies to prevent PPSP. Lidocaine, an amide local anesthetic, suppresses spontaneous ectopic discharges through its action on voltage-gated sodium channels, and experimental evidence suggests it can reduce central sensitization.[9] Several small trials and systematic reviews have indicated that lidocaine may lower the risk of developing persistent neuropathic pain following breast cancer surgery; however, the evidence remains limited due to small sample sizes and heterogeneity in study design. Similarly, pregabalin, a calcium channel modulator initially developed for epilepsy, has been studied for its possible protective role against chronic post-surgical neuropathic pain. Although some Cochrane reviews have shown promising results, especially in cardiac surgery contexts, evidence for its effectiveness in breast cancer patients remains inconclusive.[10]
Equally important, the symptom burden for women undergoing breast cancer surgery extends beyond pain alone. Postoperative nausea and vomiting (PONV) and post-discharge nausea and vomiting (PDNV) have been identified as prevalent and distressing co-occurring symptoms.[11] Despite advances in antiemetic prophylaxis and enhanced recovery protocols, many women continue to report nausea and vomiting well into the recovery period, with significant implications for patient satisfaction, adherence to analgesic regimens, and overall quality of life.[12] Interestingly, several studies have noted that nausea and vomiting may be even more problematic than pain in the early post-discharge phase. This clustering of symptoms like pain, nausea, fatigue, and sleep disturbances suggests an intricate interplay that is still poorly understood and largely under-researched in the context of breast cancer recovery.[13]
Emerging areas of research have also begun to explore novel contributors to PPSP, including the role of the gut microbiota and its impact on pain modulation through the gut-brain axis.[14] Preclinical and clinical studies indicate that gut microbial composition may influence central and peripheral sensitization pathways, suggesting a potential avenue for preventive strategies using probiotics, prebiotics, or dietary modifications. While this remains an exploratory area, it underscores the need for an expanded, multidisciplinary approach to understanding and managing persistent pain.[15]
From a nursing and rehabilitation perspective, the concept of evidence-based nursing (EBN) has gained traction as a means of addressing post-surgical complications, including upper limb dysfunction and pain. EBN emphasizes the use of the best available scientific evidence to guide individualized care, moving beyond traditional empiric practice to adopt interventions that have demonstrated efficacy in enhancing recovery, improving function, and minimizing complications.[16] Rehabilitation programs, including physiotherapy and psychosocial support, play a vital role in mitigating the impact of PPSP and its associated functional impairments.[17]
In summary, despite significant advances in surgical techniques, anesthetic management, and supportive care, persistent post-surgical pain following breast cancer treatment remains a substantial clinical challenge.[18] Its multifactorial etiology, which involves surgical trauma, nerve injury, radiation-induced tissue changes, poorly controlled acute pain, and complex symptom clusters, highlights the necessity for comprehensive, multidisciplinary strategies that address both prevention and management.[19] High-quality research is urgently needed to refine predictive models, identify effective interventions, and develop individualized approaches that optimize recovery while minimizing long-term morbidity. This literature review aims to critically synthesize current evidence on the prevalence, risk factors, pathophysiology, and management of post-surgical pain in breast cancer survivors, with the goal of informing future research and improving patient outcomes.[20,21]
Methodology
A systematic literature search was conducted using PubMed and Google Scholar databases to identify relevant studies addressing post-surgical pain following breast cancer treatment. The search strategy incorporated the terms: “post-surgical pain” AND “breast cancer”, and filters were applied to include clinical trials, randomized controlled trials, observational studies, and human Studies. The search included publications from January 1, 2015, to June 30, 2025, and was limited to studies published in English.
Inclusion criteria:
- Clinical trials, observational studies, and randomized controlled trials
- Studies involving human subjects
- Articles published in English
- Studies including female participants
- Studies published between January 1, 2015, and June 30, 2025
Exclusion criteria:
- Books, commentaries, editorials, letters, documents, and book chapters
- Case reports, case series, and literature reviews
- Articles published in languages other than English
- Animal studies and in vitro (laboratory) studies
- Articles published before January 1, 2015, or after June 30, 2025
- Studies lacking a reported results section
The initial search yielded a total of 28,702 articles. After applying the inclusion and exclusion criteria, 1,539 references were downloaded from PubMed and Google Scholar. Prior to screening, 1,255 records were removed due to duplication, technical errors, or irrelevance. In the screening phase, the remaining 284 records were assessed based on titles and abstracts, resulting in the exclusion of 204 studies. Of the 80 reports selected for retrieval, 58 could not be accessed due to the unavailability of full text or restricted access.
The remaining 22 full-text articles were evaluated for eligibility. Of these, 16 studies were excluded due to a scale for the assessment of narrative review articles (SANRA) score of <10, indicating insufficient quality. Ultimately, six studies met all inclusion criteria and were included in the final review. This entire process is visually summarized in Figure 1, ensuring a transparent and systematic approach to study selection.
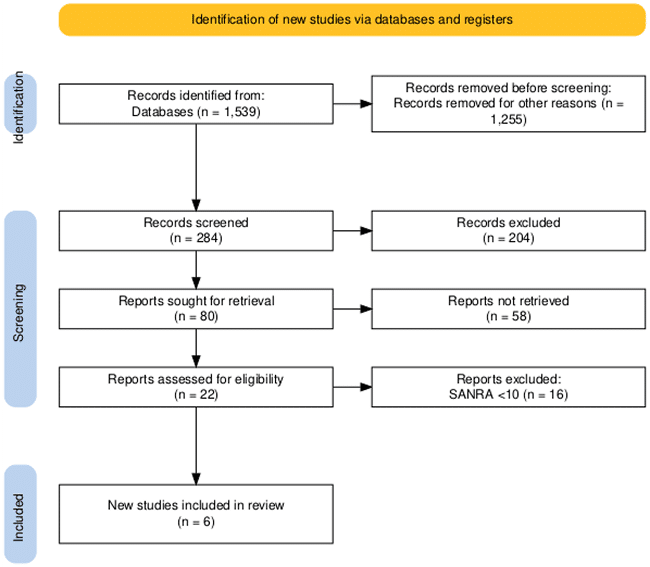
Figure 1: PRISMA flow diagram
Results
Paravertebral block lowers breast cancer post-operative pain
The study conducted by Lin ZM et al., in 283 female patients scheduled for primary breast cancer surgery, was screened; 218 consented and were randomized. During the study, four withdrew consent, three died within six months, two more died within 12 months, three were lost at six-month follow-up, and two more at 12 months. Thus, 214 patients were included in the intention-to-treat and safety analyses; 208 and 204 patients were included in the six- and 12-month analyses, respectively. Baseline characteristics were comparable between groups. PVB achieved a median sensory blockade of six dermatomes (Interquartile range (IQR): 5–6), covering T2–T6. Intraoperative sufentanil use (P < 0.001) and rescue analgesia within three days (P = 0.027) were significantly lower in the PVB group; other perioperative variables were similar.
At six months, chronic postsurgical pain (CPSP) incidence was significantly lower in the PVB group (12.5% (13/104)) than in the control group (24.0% (25/104); relative risk (RR) = 0.52, 95% confidence interval (CI) = 0.28–0.96, P = 0.031). Subgroup analysis showed PVB was more effective in patients with a body mass index (BMI) <25 kg/m² and those undergoing mastectomy (P = 0.060 and P = 0.032, respectively). Numeric rating scale (NRS) pain scores at rest and with movement within 48 hours were significantly lower in the PVB group (P = 0.006 and P < 0.001). Neuropathic pain was also significantly reduced in the PVB group at six months (10.6% (11/104) vs 23.1% (24/104), RR = 0.46, 95% CI = 0.24–0.89, P = 0.016) and at 12 months (10.9% (11/101) vs 22.3% (23/103), RR = 0.49, 95% CI = 0.25–0.95, P = 0.028). Other parameters showed no significant differences. Thoracic PVB was successfully performed in all patients, with no significant differences in adverse events between groups.[22]
Pain, fatigue, and opioids drive post-op nausea risk
Wesmiller SW et al., study included 334 women with a mean age of 60.0 ± 12.0 years (range 26–88); most were White (88%) and non-smokers (90%). Two days post-discharge, 84 women (25.1%) reported some nausea. Fewer than 10% had nausea in the PACU, but many experienced it during the car ride home. Only 17 participants (5%) reported vomiting after discharge, compared to 85 (25.4%) who reported nausea, with 40 (52%) rating their nausea as four or higher on a 0–10 scale. Given the low rate of vomiting, the analysis focused on post-discharge nausea (PDN).
Women with PDN had significantly higher mean pain scores than those without (4.46 ± 2.32 vs 2.82 ± 2.55, P<0.001). There was no significant difference between White and Black women, but those with PDN were significantly younger (P<0.007). Over half of those with PDN had a history of motion sickness, compared to 35% of those without. Opioid use was also significantly associated with PDN (P<0.001). Of the 290 women prescribed opioids, 159 took at least one dose, 54 (33%) did not fill their prescription, and 77 filled it but did not use it or felt it was unnecessary for pain control.
Unadjusted odds ratios showed significant associations between PDN and anxiety, depressive symptoms, fatigue, sleep disturbance, and pain. Due to high correlation, only anxiety was included in the adjusted regression model, which showed no multicollinearity concerns. The final adjusted analysis identified pain, fatigue, opioid use, type of surgery, and history of motion sickness as significant predictors of PDN. Age and history of PONV were not significant factors, and smoking was excluded due to the low proportion of smokers (10%).[23]
Memantine may reduce neuropathic pain after mastectomy
Morel V et al., demonstrated that of 207 patients assessed, 104 declined and 60 did not meet inclusion criteria; 43 consented and were randomized (memantine: n = 20; placebo: n = 20; after three withdrawals). Recruitment was from March 2012 to April 2013, with follow-up completed by November 2013. No adverse events or dropouts occurred during the six-month follow-up.
At three months post-mastectomy (M3), NRS pain was significantly lower with memantine than placebo (0.2 ± 0.4 vs 1.3 ± 1.8; P = 0.017); this was not significant at six months (M6) (0.5 ± 0.8 vs 0.9 ± 2.0; P = 0.10). The primary outcome effect size was 0.76 (95% CI: 0.12–1.40). Pain decreased within the memantine group from baseline to M3 (1.2 ± 2.0 vs 0.2 ± 0.4; P = 0.016). No group differences in surgery-induced neuropathic pain (DN4) were seen at M3 or M6 (placebo: 45% at M3, 30% at M6; memantine: 35% at both). Six placebo patients had DN4 scores ≥4 (5–8) vs two in the memantine group (score 5).
All patients used non-opioids and short-term opioids for two days post-surgery. Fewer memantine patients required antiepileptics for neuropathic pain at M3 (1 (5%) vs 6 (30%); P = 0.040), persisting at M6 (P = 0.040; time effect p = 0.041). McGill pain scores showed improved affective components at M3 for memantine (1.4 ± 1.9) vs placebo (10.0 ± 13.2; P = 0.032); other questionnaires showed no differences.
Half had prior chemotherapy (memantine: n = 11; placebo: n = 10). In this subgroup, pain scores at M3 and M6 were significantly lower for memantine (M3 ΔNRS: -1.5 ± 2.2 vs placebo 1.0 ± 2.3, P = 0.004; M6 ΔNRS: -1.2 ± 2.0 vs placebo 1.2 ± 3.2, P = 0.013), but the interaction was not significant (ΔNRS P = 0.06). Neuropathic pain from chemotherapy (DN4) was lower for memantine (M3 ΔDN4: -2.1 ± 1.6 vs placebo -0.5 ± 0.8, p = 0.001; M6 ΔDN4: -2.4 ± 2.0 vs placebo -1.0 ± 1.3, P = 0.009) without significant interaction (P = 0.73). Chemotherapy-induced paresthesia and dysesthesia dropped by 55% at M3 with memantine (Baseline: 9 (82%); M3: 3 (27%); P = 0.01) but not placebo (Baseline: 5 (50%); M3: 4 (40%); P = 0.32).
No significant group differences were seen in cognitive tests or overall quality of life at M3 or M6. Leeds sleep scores showed no significant differences except “behavior following wakefulness” at M6, which improved for memantine vs baseline (Baseline: 6.8 ± 3.8 vs 4.6 ± 5.5; M6: 4.8 ± 4.9 vs 5.8 ± 3.8; p = 0.038).[24]
Microbiome diversity predicts pain after breast cancer surgery
Masaud K et al., demonstrated that the data were collected from 68 patients (mean age 42.5 ± 10.6 years). Of these, 45 (66.17%) provided a stool sample and completed the study; 15 (22.05%) declined after consent, one (1.47%) did not have surgery, and seven (10.29%) could not provide a sample. Surgeries included wide local excision (WLE) with Sentinel Lymph Node Biopsy (SLNB) in 21 (46.6%), mastectomy with SLNB in 10 (22.2%), and unilateral mastectomy in two (4.4%). Of the 45 who provided a sample, one (2.2%) pre-op, two (4.4%) post-op, and four (8.8%) at 12 weeks did not respond. Pre-op, 68.8% reported no pain; pain peaked 60 min post-op (33% with moderate pain) and decreased by 12 weeks (51.2% no pain, 48.8% some pain).
Psychological Measures: One patient (2.2%) did not complete pre-/post-op questionnaires. State anxiety ≥40 was 40% pre-op vs 28.8% at 12 weeks; trait anxiety ≥40 was 24.4% both times. Catastrophising was low pre-op (98%) and post-op (95%); stress increased post-op.
Microbiome Diversity: Alpha diversity was significantly lower in severe vs mild pain at 60 min post-op, with a similar trend at 12 weeks; no differences for patients without PPSP. Bray–Curtis dissimilarity showed that sample type and PPSP affected clustering (p < 0.028).
Bifidobacteria were more abundant in PPSP patients at 12 weeks; Bacteroides were higher without PPSP. LEfSe found species like Anaerostipes hadrus, Ruminococcus lactaris, and Alistipes shahii enriched in no PPSP; Bifidobacterium longum, Megamonas hypermegale, Bifidobacterium breve, and Streptococcus vestibularis in PPSP. Bifidobacterium longum abundance was significantly linked to pain severity at 12 weeks.
Differential gene expression showed pathways up- and up-/downregulated between the pain/no pain groups. MAGs mapped to 253 species; Faecalibacterium prausnitzii and Eubacterium rectale made up 70.56% of % total abundance. Bray–Curtis dissimilarity showed PPSP grouping effect (p < 0.036, explaining 4.41% variation). Linear discriminant analysis effect size (LEfSe) highlighted Ethanoligenens harbinense and Clostridium cellulosi with severe pain at 60 min, Bifidobacterium angulatum and Bifidobacterium dentium at 12 weeks; six species, including Bifidobacterium longum and Bifidobacterium bifidum, were linked to PPSP.
Spearman correlations showed pain links with species like Bacteroides cellulosilyticus, Flavonifractor plautii, and Clostridioides difficile. A Random Forest model clearly distinguished PPSP status based on species distribution.[25]
PMPS impacts functioning and quality of life
Kakati B et al. demonstrated that 523 women underwent mastectomy with axillary clearance or selective lymph node biopsy/sampling. Sixty patients enrolled in another study involving pectoral nerve block (PECS) blocks and were excluded. Additionally, 115 were illiterate, 114 declined consent, 105 were unwilling to complete and mail questionnaires, four had prior chest wall surgery, and five had emergency surgery. Ultimately, 120 women met the criteria and consented to participate.
The mean age of participants was 52.45 ± 10.2 years, and the mean BMI was 25.83 ± 4.63. Of these, 39.2% (47) had stage IIB or higher disease, and 53.3% (64) received preoperative chemotherapy. Surgeries included standard axillary dissection in 100 women (83.3%) and selective lymph node dissection with intercostobrachial nerve (ICBN) preservation in 20 (16.7%). Postoperatively, 102 (85%) received chemotherapy and 76 (63.3%) had radiotherapy. Complications were minimal: four (3.3%) developed hematoma, 12 (10%) had wound infections, and two (1.7%) had local recurrence within six months.
All patients received regular postoperative analgesics: 87 (72.5%) were given NSAIDs and paracetamol, eight (6.7%) paracetamol alone, and 25 (20.8%) a combination including tramadol. On discharge (postoperative day 1), pain scores were mostly mild (47.5%) or moderate (45.8%), with three patients (2.5%) reporting severe pain. The median average pain score was 2 (IQR: 1–3), and the worst pain was 3 (IQR: 3–5).
At six months, 51 women (42.5%) still reported pain, with 43 (35.8%) meeting criteria for post-mastectomy pain syndrome (PMPS). Pain was most often located in the anterior chest wall (41.8%), axilla (32.6%), and medial upper arm (25.6%). Most described the pain as dull aching (48.8%) and mild in intensity (55.8%). No significant links were found between PMPS incidence and age, body mass index (BMI), surgery type, ICBN dissection, postoperative pain severity, history of headache or dysmenorrhea, or radiotherapy. However, those with PMPS had worse daily functioning and quality of life scores on the BPI and European Organisation for Research and Treatment of Cancer quality of life questionnaire core 30 (EORTC QLQ-C30).[26]
Lidocaine lowers neuropathic pain after breast surgery
Khan JS et al., demonstrated that of 296 eligible patients, 165 declined and 31 could not be contacted preoperatively. A total of 100 patients were randomized: 51 to active lidocaine, 49 to placebo lidocaine, 50 to active pregabalin, and 50 to placebo pregabalin. All completed follow-ups were included in secondary analyses. The mean age was 54.3 ± 11.1 years. Most underwent unilateral lumpectomy (75%), unilateral mastectomy (18%), or bilateral procedures (7%). The majority (94%) had surgery for suspected cancer; six had prophylactic mastectomies. Sentinel lymph node biopsy was done in 63% and axillary node dissection in 7%.
Recruitment ran from December 2014 to October 2015, enrolling 100 patients across two sites in 42 weeks (planned: 52 weeks). Three-month follow-up was 100% complete; acute follow-up was 98% due to minor missed visits. Study drug compliance was high: 92% received lidocaine/placebo intraoperatively; eight did not due to anesthesiologist refusal (4), oversight (1), time constraints (1), patient refusal (1), or unclear administration (1). Pregabalin/placebo compliance exceeded 88% perioperatively; preoperative compliance was 98%, postoperative ≥92% for placebo but dropped to 80% for active pregabalin by day 9 due to mild side effects, refusal, or lost medication.
At three months, 53% had persistent neuropathic pain: 77.4% mild at rest (NRS 0–3), 15.1% moderate (NRS 4–6), and 7.5% severe (NRS 7–10); with arm movement, 69.8% had mild pain, 17.0% moderate, and 13.2% severe.
Lidocaine reduced persistent neuropathic pain significantly (43.1% vs 63.3%; RR = 0.68; 95% CI = 0.47–1.0; P = 0.049) but not moderate to severe pain (7.8% vs 16.3%; RR = 0.48; P = 0.205). Pregabalin did not significantly reduce persistent pain (60% vs 46%; RR = 1.3; P = 0.166) or moderate to severe pain (12% vs 12%; RR = 1.00; P = 1.00). There was no significant lidocaine–pregabalin interaction (52% vs 58.3%; RR = 0.89; P = 0.656).
Pain scores at rest or with movement did not differ between lidocaine and placebo (P = 0.295, P = 0.220) or pregabalin and placebo (P = 0.734, P = 0.929). Opioid use was similar for both comparisons.
No lidocaine toxicity occurred. One placebo patient had opioid-related respiratory depression, which resolved with naloxone. One patient in the active lidocaine/active pregabalin group needed vasopressors. Nausea rates were similar, but vomiting was higher with pregabalin (12% vs 0%; P = 0.027). Infection risk, major complications, or reoperation rates were similar. No deaths, strokes, heart failure, or arrhythmias occurred.[27]
| Study / Authors | Sample size | Intervention | Key findings | Safety |
| Lin ZM et al.[23] | 218 randomized; 214 analysed | Thoracic PVB vs control | PVB reduced CPSP at 6 months (12.5% vs 24%; RR 0.52, P = 0.031); less neuropathic pain at 6 & 12 months; less intraoperative opioids & rescue analgesia needed | No significant differences in adverse events between groups |
| Wesmiller SW et al.[24] | 334 women | Observational study: no intervention; analyzed factors related to PDN | PDN incidence 25.1%; higher pain, fatigue, opioid use, history of motion sickness increased risk; pain score significantly higher in PDN group (4.46 vs 2.82) | Not applicable — observational only, no intervention |
| Morel V et al.[25] | 43 randomized (20 memantine, 20 placebo after withdrawals) | Memantine vs placebo for 4 weeks post-mastectomy | Significant pain reduction at 3 months (NRS 0.2 vs 1.3; P = 0.017); fewer required antiepileptics for neuropathic pain; benefit especially in chemo subgroup | No adverse events or dropouts during the 6-month follow-up |
| Masaud K et al.[26] | 68 enrolled; 45 completed | Observational: gut microbiome sampling pre/post breast surgery | Lower microbiome diversity linked to severe acute/post-surgical pain; certain species (e.g., Bifidobacterium longum) correlated with PPSP; potential for microbiome as biomarker | Not applicable — observational only, no intervention |
| Kakati B et al.[27] | 120 women | Observational survey on PMPS after mastectomy + axillary clearance | 42.5% had pain at 6 months; 35.8% met PMPS criteria; PMPS linked to worse daily functioning and QoL scores; pain was commonly dull aching, mild | Minimal surgical complications: few hematomas/wound infections; good follow-up; no new intervention |
| Khan JS et al.[28] | 100 randomized | IV lidocaine and/or oral pregabalin vs placebo | Lidocaine reduced persistent neuropathic pain at 3 months (43.1% vs 63.3%; RR 0.68; P = 0.049); pregabalin did not show a significant benefit; no significant difference in moderate–severe pain. | No lidocaine toxicity; mild pregabalin side effects; some nausea/vomiting with pregabalin; no major complications |
Table 1: Studies comparing postoperative pain management in breast cancer
Discussion
This review underscores that while advances in breast cancer screening, surgical techniques, and multimodal therapies have dramatically improved survival rates, especially in high-income countries where five-year survival now exceeds 90%, PPSP remains a prevalent and under-recognized challenge affecting the quality of survivorship. Across the studies reviewed, the incidence of PPSP varies widely, with estimates ranging from 20% to 60%, highlighting that surgical trauma, nerve injury, radiotherapy, and poorly controlled acute pain are key drivers of chronic pain syndromes.[28]
Findings from Lin et al. and Khan et al. demonstrate the promise of perioperative interventions such as thoracic PVB and intravenous lidocaine infusion. Both showed reductions in chronic pain incidence at six and twelve months postoperatively. However, while PVB reduced intraoperative opioid requirements and improved early pain control, its benefits appear more pronounced in certain subgroups, such as patients with lower BMI or those undergoing mastectomy, suggesting the need for patient-tailored approaches. Likewise, lidocaine infusion significantly reduced neuropathic pain incidence but did not fully mitigate moderate to severe pain, underlining the complex pathophysiology behind PPSP.[29]
The potential of pharmacologic adjuncts, such as pregabalin, remains inconclusive. Khan et al. found no significant benefit for pregabalin in preventing PPSP despite its mechanistic rationale as a modulator of calcium channels involved in neuropathic pain pathways. Similarly, Morel et al. explored memantine, showing some early promise in reducing neuropathic pain at three months, particularly for patients with prior chemotherapy exposure, but with no significant effect at six months. These mixed findings call for larger, well-powered trials to clarify which subgroups might benefit most.[30]
Beyond direct analgesia, the reviewed evidence emphasizes the multidimensional nature of post-surgical recovery. Wesmiller et al. found a striking association between pain, opioid use, fatigue, and the risk of PDNV, highlighting how unmanaged symptom clusters can complicate recovery and reduce patient satisfaction. This supports the growing view that perioperative symptom management must be holistic, extending beyond pain to encompass related factors such as nausea, sleep disturbance, and fatigue.
Emerging research by Masaud et al. suggests that gut microbiota diversity may play an underappreciated role in pain modulation through the gut–brain axis. Specific bacterial species were associated with the severity and persistence of PPSP, hinting at novel avenues for prevention through microbiome-targeted interventions. While exploratory, this line of inquiry aligns with the broader trend of precision medicine in oncology care.
Finally, functional outcomes and quality of life impacts remain major concerns. Kakati et al. found that women suffering from post-mastectomy pain syndrome (PMPS) reported significant limitations in daily activities and worse quality of life scores, reinforcing the need for comprehensive rehabilitation strategies. Evidence-based nursing, physiotherapy, and psychosocial support are crucial components in minimizing disability and facilitating reintegration into daily life.
The persistence of post-surgical pain following breast cancer treatment reflects a multifactorial problem requiring integrated solutions. Current evidence supports the role of regional anesthesia, perioperative analgesics, and symptom cluster management, but also points to critical research gaps, especially around personalized risk prediction and innovative preventive strategies like microbiome modulation. A multidisciplinary, patient-centered approach is essential to ensure that gains in breast cancer survival are not overshadowed by enduring pain and functional impairment.
Limitations
Limited sample sizes and generalizability: Many of the included studies had relatively small sample sizes and were conducted in single centers, which may limit the generalizability of their findings to broader, more diverse breast cancer populations.
Heterogeneity of study designs: There was significant variability in interventions, outcome measures, pain assessment tools, follow-up durations, and patient populations across the studies. This heterogeneity complicates direct comparisons and meta-analysis.
Short follow-up for chronic outcomes: Some trials had follow-up periods limited to six months to one year, which may not fully capture the persistence and long-term trajectories of PPSP.
Potential publication bias: The review only included studies published in English and accessible in selected databases, which may introduce publication and language bias, potentially overlooking relevant research published elsewhere.
Underrepresentation of emerging areas: While the role of the gut microbiome and psychosocial factors is increasingly recognized, current evidence remains exploratory and lacks robust, large-scale clinical trials.
Recommendations
- Run larger, multicenter trials to confirm promising pain management strategies.
- Standardize outcome measures for PPSP to compare studies better.
- Identify patient subgroups to tailor pain prevention plans.
- Address symptom clusters like fatigue, nausea, and sleep issues alongside pain.
- Expand microbiome research for innovative pain control options.
- Strengthen nursing and rehab care to improve recovery and daily functioning.
- Educate patients on pain management, early reporting, and self-care after surgery.
Conclusion
PPSP remains a significant and under-recognized challenge for breast cancer survivors, despite remarkable improvements in survival rates. This review highlights that PPSP is driven by multiple factors, including surgical nerve injury, extent of axillary dissection, radiotherapy effects, and inadequate acute pain management. Regional anesthesia techniques like paravertebral blocks and perioperative intravenous lidocaine show promise in reducing chronic pain, but evidence for other interventions like pregabalin remains inconsistent. Beyond pain, co-existing symptoms such as fatigue, nausea, and functional impairment further affect recovery and quality of life. Novel areas, including gut microbiota modulation, offer exciting directions for future research. Comprehensive, multidisciplinary approaches such as integrating evidence-based nursing, physiotherapy, psychological support, and patient education are essential to prevent and manage PPSP effectively. Continued high-quality research is needed to refine strategies, tailor interventions, and ensure that gains in breast cancer survival translate into better long-term well-being for survivors.
References
- Martinez V, Lehman T, Lavand’homme P, et al. Chronic postsurgical pain: A European survey. Eur J Anaesthesiol. 2024;41(5):351-362. doi:10.1097/EJA.0000000000001974
PubMed | Crossref | Google Scholar - Salati SA, Alsulaim L, Alharbi MH, et al. Postmastectomy Pain Syndrome: A Narrative Review. Cureus. 2023;15(10):e47384. doi:10.7759/cureus.47384
PubMed | Crossref | Google Scholar - Amiegbereta EE, Ibitoye AZ, Esi OE. Computational Analysis of a Suitable Antenna and Optimized Frequency for Microwave Ablation of Breast Cancerous Tissue. medtigo J Med. 2024;2(3):e13752246. doi:10.63096/medtigo3062249
Crossref - Vila MR, Todorovic MS, Tang C, et al. Cognitive flexibility and persistent post-surgical pain: the FLEXCAPP prospective observational study. Br J Anaesth. 2020;124(5):614-622. doi:10.1016/j.bja.2020.02.002
PubMed | Crossref | Google Scholar - Palesh O, Kamen C, Sharp S, et al. Physical Activity and Survival in Women With Advanced Breast Cancer. Cancer Nurs. 2018;41(4):E31-E38. doi:10.1097/NCC.0000000000000525
PubMed | Crossref | Google Scholar - Pradeep KRK, Raj KB, Shrivastava SP, et al. An Observational Study on Drug-Related Problems in the Treatment of Cancer Patients. medtigo J Med. 2025;3(2):e3062326. doi:10.63096/medtigo3062326
Crossref - Xiao Y, Li L, Xie Y, et al. Effects of aroma therapy and music intervention on pain and anxiety for breast cancer patients in the perioperative period. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2018;43(6):656-661. doi:10.11817/j.issn.1672-7347.2018.06.013
PubMed | Google Scholar - Wang M, Xiong HP, Sheng K et al. Perioperative Administration of Pregabalin and Esketamine to Prevent Chronic Pain After Breast Cancer Surgery: A Randomized Controlled Trial. Drug Des Devel Ther. 2023;17:1699-1706. doi:10.2147/DDDT.S413273
PubMed | Crossref | Google Scholar - Reyad RM, Omran AF, Abbas DN, et al. The Possible Preventive Role of Pregabalin in Postmastectomy Pain Syndrome: A Double-Blinded Randomized Controlled Trial . J Pain Symptom Manage. 2019;57(1):1-9. doi:10.1016/j.jpainsymman.2018.10.496
PubMed | Crossref | Google Scholar - Thabet TS, Khedr SA. Stellate Ganglion Destruction With Alcohol Versus Thermal Ablation for Chronic Post-Mastectomy Pain: A Randomized Trial. Pain Physician. 2024;27(2):E231-E238.
Stellate Ganglion Destruction With Alcohol Versus Thermal Ablation for Chronic Post-Mastectomy Pain: A Randomized Trial - Elvir-Lazo OL, White PF, Yumul R et al. Management strategies for the treatment and prevention of postoperative/postdischarge nausea and vomiting: an updated review. F1000Res. 2020;9:F1000 Faculty Rev-983. doi:10.12688/f1000research.21832.1
PubMed | Crossref | Google Scholar - Bennedsgaard K, Grosen K, Attal N, et al. Neuropathy and pain after breast cancer treatment: a prospective observational study. Scand J Pain. 2022;23(1):49-58. doi:10.1515/sjpain-2022-0017
PubMed | Crossref | Google Scholar - Sri Ramya T, Samatha A, Swarnalatha K, et al. Virtual Screening of Potent Phytochemicals of Murraya Paniculata for Anti-Breast Cancer Activity: Molecular Docking and Dynamics Approaches. medtigo J Pharmacol. 2025;2(2):e3061223. doi:10.63096/medtigo3061223
Crossref - González-Rubino JB, Martín-Valero R, Vinolo-Gil MJ. Physiotherapy protocol to reduce the evolution time of axillary web syndrome in women post-breast cancer surgery: a randomized clinical trial. Support Care Cancer. 2025;33(4):326. doi:10.1007/s00520-025-09373-1
PubMed | Crossref | Google Scholar - Torres-Lacomba M, Prieto-Gómez V, Arranz-Martín B, et al. Manual Lymph Drainage With Progressive Arm Exercises for Axillary Web Syndrome After Breast Cancer Surgery: A Randomized Controlled Trial. Phys Ther. 2022;102(3):pzab314. doi:10.1093/ptj/pzab314
PubMed | Crossref | Google Scholar - Zhang X, Zhang D, Yu P, Li X. Effects of Continuous Care Combined with Evidence-Based Nursing on Mental Status and Quality of Life and Self-Care Ability in Patients with Liver from Breast Cancer: A Single-Center Randomized Controlled Study. Comput Math Methods Med. 2022; doi:10.1155/2022/3637792
Crossref | Google Scholar - Aydin M, Kose E, Odabas I, et al. The effect of exercise on life quality and depression levels of breast cancer patients. Asian Pac J Cancer Prev. 2021;22(3):725-732. doi:10.31557/APJCP.2021.22.3.725
PubMed | Crossref | Google Scholar - Spivey TL, Gutowski ED, Zinboonyahgoon N, et al. Chronic Pain After Breast Surgery: A Prospective, Observational Study. Ann Surg Oncol. 2018;25(10):2917-2924. doi:10.1245/s10434-018-6644-x
PubMed | Crossref | Google Scholar - Hiensch AE, Monninkhof EM, Schmidt ME, et al. Design of a multinational randomized controlled trial to assess the effects of structured and individualized exercise in patients with metastatic breast cancer on fatigue and quality of life: the EFFECT study. Trials. 2022;23(1):610. doi:10.1186/s13063-022-06556-7
PubMed | Crossref | Google Scholar - Andersen KG, Kehlet H. Persistent pain after breast cancer treatment: a critical review of risk factors and strategies for prevention. J Pain. 2011;12(7):725-746. doi:10.1016/j.jpain.2010.12.005
PubMed | Crossref | Google Scholar - Al-Hilli Z, Wilkerson A. Breast Surgery: Management of Postoperative Complications Following Operations for Breast Cancer. Surg Clin North Am. 2021;101(5):845-863. doi:10.1016/j.suc.2021.06.014
PubMed | Crossref | Google Scholar - Lin ZM, Li MH, Zhang F, et al. Thoracic Paravertebral Blockade Reduces Chronic Postsurgical Pain in Breast Cancer Patients: A Randomized Controlled Trial. Pain Med. 2020;21(12):3539-3547. doi:10.1093/pm/pnaa270
PubMed | Crossref | Google Scholar - Wesmiller SW, Bender CM, Grayson SC, et al. Postdischarge Nausea and Vomiting and Co-occurring Symptoms in Women Following Breast Cancer Surgery. J Perianesth Nurs. 2023;38(3):478-482. doi:10.1016/j.jopan.2022.08.014
PubMed | Crossrefv | Google Scholar - Morel V, Joly D, Villatte C, et al. Memantine before Mastectomy Prevents Post-Surgery Pain: A Randomized, Blinded Clinical Trial in Surgical Patients. PLoS One. 2016;11(4):e0152741. doi:10.1371/journal.pone.0152741
PubMed | Crossref | Google Scholar - Masaud K, Collins JM, Rubio RC, et al. The gut microbiota in persistent post-operative pain following breast cancer surgery. Sci Rep. 2024;14(1):12401. doi:10.1038/s41598-024-62397-1
PubMed | Crossref | Google Scholar - Kakati B, Nair N, Chatterjee A. Post mastectomy pain syndrome at an Indian tertiary cancer centre and its impact on quality of life. Indian J Cancer. 2023;60(2):275-281. doi:10.4103/ijc.ijc_861_21
PubMed | Crossref | Google Scholar - Khan JS, Hodgson N, Choi S, et al. Perioperative Pregabalin and Intraoperative Lidocaine Infusion to Reduce Persistent Neuropathic Pain After Breast Cancer Surgery: A Multicenter, Factorial, Randomized, Controlled Pilot Trial. J Pain. 2019;20(8):980-993. doi:10.1016/j.jpain.2019.02.010
PubMed | Crossref | Google Scholar - Gherghe M, Bordea C, Blidaru A. Sentinel lymph node biopsy (SLNB) vs. axillary lymph node dissection (ALND) in the current surgical treatment of early stage breast cancer. J Med Life. 2015;8(2):176-180.
Sentinel lymph node biopsy (SLNB) vs. axillary lymph node dissection (ALND) in the current surgical treatment of early stage breast cancer - Bruce J, Mazuquin B, Canaway A, et al. Exercise versus usual care after non-reconstructive breast cancer surgery (UK PROSPER): multicentre randomised controlled trial and economic evaluation. BMJ. 2021;375:e066542. doi:10.1136/bmj-2021-066542
PubMed | Crossref | Google Scholar - Mohite PP, Kanase SB. Effectiveness of Scapular Strengthening Exercises on Shoulder Dysfunction for Pain and Functional Disability after Modified Radical Mastectomy: A Controlled Clinical Trial. Asian Pac J Cancer Prev. 2023;24(6):2099-2104. doi:10.31557/APJCP.2023.24.6.2099
PubMed | Crossref | Google Scholar
Acknowledgments
Not applicable
Funding
Not applicable
Author Information
Corresponding Author:
Samatha Ampeti
Department of Pharmacology
University College of Pharmaceutical Sciences, Kakatiya University, Warangal, India
Email: ampetisamatha9@gmail.com
Co-Authors:
Sonam Shashikala BV, Mansi Srivastava, Shubham Ravindra Sali, Patel Nirali Kirankumar, Raziya Begum Sheikh
Independent Researcher
Department of Content, medtigo India Pvt Ltd, Pune, India
Author Information
Corresponding Author:
Samatha Ampeti
Department of Pharmacology
University college of Pharmaceutical Sciences, Kakatiya University, Warangal, India
Email: ampetisamatha9@gmail.com
Co-Authors:
Sonam Shashikala BV, Mansi Srivastava, Shubham Ravindra Sali, Patel Nirali Kirankumar, Raziya Begum Sheikh
Independent Researcher
Department of Content, medtigo India Pvt Ltd, Pune, India
Ethical Approval
Not applicable
Conflict of Interest Statement
None
Guarantor
None
DOI
Cite this Article
Sonam SBV, Samatha A, Mansi S, Shubham RS, Patel NK, Raziya BS. Post Surgical Pain Following Breast Cancer. medtigo J Anesth Pain Med. 2025;1(1):e3067117. doi:10.63096/medtigo3067117 Crossref









