Author Affiliations
Abstract
Background: The coronavirus disease of 2019 (COVID-19) was caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-COV-2). Research indicates that individuals who succumb to the disease are often older, predominantly male, and more likely to have underlying health conditions such as hypertension, diabetes, cardiovascular, or respiratory diseases. This highlights the importance of identifying risk factors across different populations and settings.
Objective: To assess the proportion of different outcomes, such as recovery, hospitalization, and mortality, among home-isolated COVID-19 patients and to determine various risk factors associated with COVID-19 adverse outcomes.
Methodology: This study was conducted at Shimoga Institute of Medical Sciences, Shivamogga, Karnataka. Patient details, including contact information, were obtained from the triage records. Data collection was done through structured telephonic interviews.
Results: A total of 168 people participated in this study, with 94 men (55.4%) and 74 women (44.6%). More than 90% of home-isolated patients recovered without complications. However, 10.75% required hospitalization, and 3% succumbed to the disease. Comorbidities were reported in 37% of the patients.
Conclusion: Hospitalization was more frequent among older individuals, males, and those with comorbidities. Mortality risk was particularly higher in hospitalized patients with advanced age and existing health conditions. Although most home-isolated patients recovered, the fact that over 10% required escalation of care underlines the importance of vigilant monitoring. Timely intervention for home-isolated patients could significantly reduce adverse outcomes and save lives.
Keywords
Coronavirus disease of 2019 (COVID-19), Risk factors, Home isolation, Hospitalization rate, Pandemic.
Introduction
COVID-19 is a current global pandemic caused by the newly discovered novel SARS-COV-2 affecting 220 countries and territories all around the world. In December 2019, the Wuhan Municipal Health Commission in China reported a slew of pneumonia cases in Wuhan, Hubei Province. COVID-19 was declared a “public health emergency of international concern” by the WHO in January 2020.[1] The total number of confirmed new cases and deaths continues to rise. There have been 141,536,837 confirmed cases and 3,026,938 deaths globally as of April 20, 2021.[2] The first incidence was reported in India on January 27, 2020, when a 20-year-old woman arrived at the General Hospital in Thrissur, Kerala, with a one-day history of dry cough and sore throat.[3] To date, 14,788,109 cases have been confirmed, and 177,150 deaths have been registered in the country, with a case fatality rate of 3.2% and a recovery rate of 85.86%.[4-6]
India, which has the highest population in the world, second only to China, is one of the global epicenters of the disease at the time of writing, and the causes are varied, but some effects can be attributed to the intricate interplay of mutant strains, as well as failure to follow COVID-19 procedure after the initial wave. India, like many other third-world countries, lacks access to resources such as beds, diagnostics, and ventilators, and this scarcity aids illness transmission and difficulties in patients, among other things. COVID-19 symptoms vary from patient to patient and may include fever, cough, breathing difficulties, and organ failures. And in severe cases, COVID-19 can lead to death, posing a concern to public health.[7,8]
According to studies, in comparison to those who have recovered, patients who have died thus far were older, more likely to be male, and to have a comorbidity such as hypertension, diabetes, cardiovascular disease, or lung disease.[9] Due to COVID-19, the rates of diabetes (10.6 percent) and chronic respiratory disease (4.8 percent), as well as obesity (4.4 percent), chronic heart disease (4.4 percent), and cancer (0.3 percent), appear to be associated to mortality and hospitalization rates thus necessitating the assessment of risk variables in various demographic groups or contexts.[10] Furthermore, data on illness outcomes and research into the risk variables that influence COVID-19 results may help policymakers prioritize non-pharmaceutical measures such as the need for general beds, ICU beds, ventilators, and screening. As substantial confounders, the risk variables may be essential for the design and interpretation of clinical trials on the efficacy of therapies.
This study aims to evaluate the relationship between comorbidities and COVID-19 outcomes, including hospitalization, recovery, and mortality, among home-isolated patients. Data of laboratory-confirmed COVID-19 cases with definitive outcomes were retrospectively collected from the triage records of McGann Hospital, Shivamogga.
Methodology
The study was carried out in the Shimoga Institute of Medical Sciences, Shivamogga, Karnataka.
Study design: Retrospective longitudinal study
Subjects: Home-isolated COVID-19 patients
Inclusion criteria: All COVID-19 patients registered in the triage of McGANN hospital, Shimoga Institute of Medical Sciences, Shimoga, from April 20th to June 20th, 2021.
Exclusion criteria: People who did not consent, or were suffering from mental illnesses, or did not respond, or patients whose contact details were not available.
Sample size: n=150. Telephonic interviews were conducted to collect the necessary information. The proportion of recovery from COVID-19 infection is p=85.86%, according to data from the government of India.
So, with the help of the formula 4pq/d 2,
where
p is 85.86%
d is precision= 6%
q= 100-p = 100-85.86= 14.14
n = 4*85.86*14.14/6*6 = 133, with 10% non-responders
n=146. It was rounded off to 150. So, the estimated sample size = 150
Time and duration of study: April 20 th – June 20 th, 2021; 2 months
Sampling: The basic information of all the home-isolated COVID-19 patients who reported to McGANN Hospital was collected. The patients were telephoned, and their basic information was confirmed, followed by the collection of outcome variables.
Ethical Permission: Ethical approval was obtained from the IEC of Shimoga Institute of Medical Sciences, which is recognized by the CDSCO (Registration No. ECR/952/Inst/KA/2017/RR-20) and registered with NECRBHR-DHR (File No. EC/NEW/INST/2020/922).
Variables:
- Outcome Variables:
a) Recovery
b) Hospital admission
c) Mortality/Death - Risk Factor Variables:
a) Diabetes
b) Chronic Respiratory Disease
c) Obesity
d) Chronic Heart Disease
e) Cancer
f) Any other chronic diseases
Analysis: Data was entered into an Excel spreadsheet and analyzed using Epi-Info software. Statistical tests used:
a) Proportion
b) Percentage
c) t-test
d) Chi-square test
Results
Patient’s clinical data: Data from 200 patients were accessible, of which 168 responded and were regarded as having a definitive outcome. The mean ± SD age of the patients was 41.6766 ± 16.6633. There were 75 females (44.64%) and 93 males (55.36%) in our study. Shimoga Taluk was home to all the patients. At least one comorbidity was present in 57 people (33.93%). 130 (77.38%) of the 168 patients had symptoms. Diabetes and hypertension were the most prevalent comorbidities (45.61%). A total of 15 people were admitted to the hospital, of whom 8 died. A total of 160 patients recovered, with 150 requiring no hospitalization and recovering at home. Patients above the age of 60 had a higher percentage of hospitalization (35.71%) than those under the age of 60. (3.57%). Furthermore, older individuals had the highest prevalence of comorbidities, the most prevalent of which were diabetes and hypertension. Symptoms such as fever, body aches, cough, and sore throat were also more common in them.
Clinical characteristics of COVID-19 patients
Age: In our study, 15 people were hospitalized, 13 of whom were over 60 years old and two of whom were under 60 years old. We found that there was an association between age and adverse outcomes such as hospitalization and death using the chi-square test. The association was found to be significant. The chi-square statistic is 58.4516. The p-value is < 0.00001. The result is significant at p < 0.05.
| Age | Non-Hospitalized | Hospitalized | Total |
| ≥ 60 | 10 (6.5%) | 13 (86.66%) | 23 (13.69%) |
| <60 | 140 (91.5%) | 2 (13.34%) | 142 (86.31%) |
| Total | 153 (100%) | 15 (100%) | 168 (100%) |
Table 1: Age distribution of hospitalized and non-hospitalized patients
Comorbidities: In our study, 57 participants had at least one comorbidity, the most prevalent of which were diabetes and hypertension. There were 18 people with diabetes, 18 with hypertension, 2 with cardiovascular disease, and 3 with pulmonary disease. 14 of the 15 people admitted to the hospital had at least one comorbidity, the most prevalent of which were diabetes and hypertension. 7 out of 8 patients who died had at least one comorbidity, the most prevalent of which were diabetes and hypertension.
| Comorbidities | Non-Hospitalized | Hospitalized |
| Diabetes | 18 | 8 |
| Hypertension | 18 | 8 |
| Cardiovascular diseases | 1 | 1 |
| Pulmonary diseases | 2 | 1 |
Table 2: Depicts the comorbidities that exist in both hospitalized and non-hospitalized patients
| Comorbidities | Non-Hospitalized | Hospitalized | Total |
| Yes | 43 (28.10%) | 14 (93.33%) | 57 (33.92%) |
| No | 110 (71.9%) | 1 (6.67%) | 111 (66.08%) |
| Total | 153 (100%) | 15 (100%) | 168 (100%) |
Table 3: Represents the descriptive table of comorbidities
Sex: The chi-square statistic is 0.1437. The p-value is .704663. The result is not significant at p < 0.05.
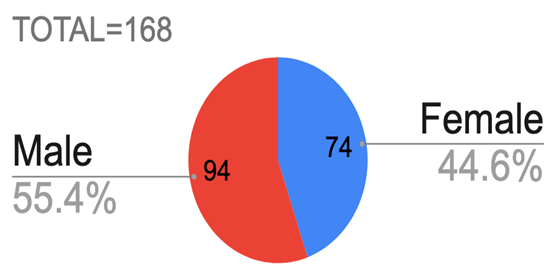
Figure 1: Pie chart showing the sex distribution of males and females infected
| Sex | Non-Hospitalized | Hospitalized | Total |
| Male | 84 (54.9%) | 10 (66.66%) | 94 (55.95%) |
| Female | 69 (45.1%) | 5 (33.34%) | 74 (44.05%) |
| Total | 153 (100%) | 15 (100%) | 168 (100%) |
Table 4: Sex distribution among hospitalized and non-hospitalized patients
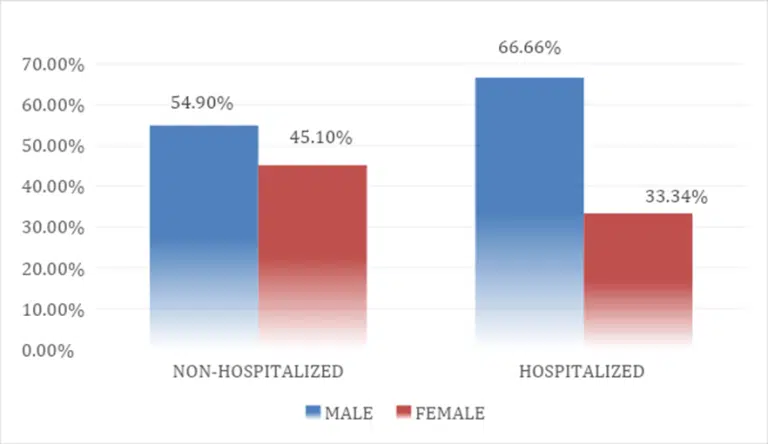
Figure 2: Complex chart showing the sex distribution among hospitalized and non-hospitalized patients
Because COVID-19 is a relatively new and understudied disease, the data available on it is limited. Tables 1 and 2 revealed, however, that elderly individuals with underlying comorbidities had a higher risk of experiencing illness severity. Table 1 demonstrates that elderly individuals had a higher risk of being hospitalized and possibly dying. The elderly, a vulnerable population with underlying chronic disease, were in danger of not only hospitalization but also death. This conclusion is supported by Tables 1 and 2. Males had a slightly higher rate of hospitalization, according to our study. 13 (86.66%) out of the 15 patients who were hospitalized were above 60. 8 of them had diabetes, and 8 had hypertension. Diabetes and Hypertension were the most associated chronic comorbidities among the elderly patients. Cardiovascular and pulmonary diseases were associated with the severity of the infection.
Signs and symptoms: Out of 168 patients, 40 (23.81%) were asymptomatic. Fever, cough, and sore throat were the most common symptoms among hospitalized patients, as shown in Table 4. The symptoms, on the other hand, were not linked to the severity of the condition.
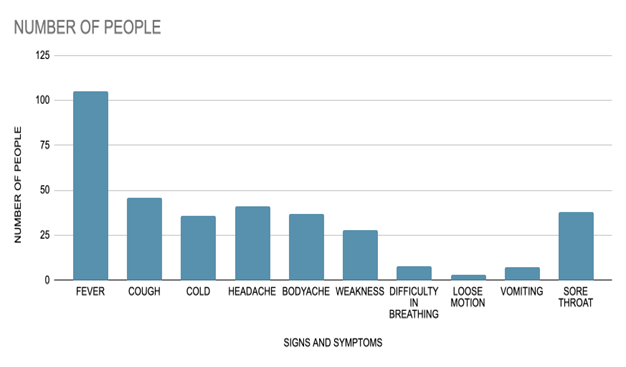
Figure 3: Bar graph showing the signs and symptoms present in the patients
Risk factors associated with death as the outcome: A total of 15 hospitalized patients were examined for death out of a total of 168 people. 8 of the 15 hospitalized patients died, according to the data. Death rates were higher among older patients (≥ 60). Males made up half of the group, while females made up the other half. 6 patients (75%) had at least one comorbidity, the most prevalent of which were diabetes and hypertension. The most prevalent symptoms were fever and cough. The analysis (Table 6) revealed that age was associated with death. 87.5% of the patients who died were elderly. 85.7% of them had an underlying disease.
| Age | Death | Recovered | Total |
| ≥ 60 | 7 (87.5%) | 16 (10%) | 23 (13.69%) |
| <60 | 1 (12.5%) | 144 (90%) | 145 (86.31%) |
| Total | 8 (100%) | 160 (100%) | 168 (100%) |
Table 5: Age distribution among people who died
| Sex | Death | Recovered | Total |
| Female | 4 (50%) | 70 (43.75%) | 74 (44.04%) |
| Male | 4 (50%) | 90 (56.25%) | 94 (55.96%) |
| Total | 8 (100%) | 160 (100%) | 168 (100%) |
Table 6: Sex distribution among people who died
| Comorbidities | Death | Recovered | Total |
| Diabetes | 5 (41.66%) | 21 (46.66%) | 26 (45.61%) |
| Hypertension | 5 (41.66%) | 21 (46.66%) | 26 (45.61%) |
| Cardiovascular diseases | 1 (8.34%) | 1 (2.22%) | 2 (3.51%) |
| Pulmonary diseases | 1 (8.34%) | 2 (4.46%) | 3 (5.27%) |
| Total | 12 (100%) | 45 (100%) | 57 (100%) |
Table 7: Comorbidities found among people who died
Discussion
The study on 168 patients revealed the recovery rate to be 91.07% and the mortality rate to be 4.765, which is higher than the recovery and fatality rate of the country at the time of the study.[5,6] Elderly male patients with underlying comorbidities had a greater mortality risk, according to a study conducted in Tamil Nadu, India.[11] The present study showed that the mortality rate in younger patients was less than that of older patients; this was consistent with the findings of the global evidence.[12,13] Our studies also showed that men were more severely infected than women, consistent with global evidence.[14] This finding was, however, inconsistent with the findings of the country, which showed that the mortality rate was higher among women, as opposed to our study, which showed that it was equal among men and women. The fatality ratlifestylemen is 3.07% as opposed to male being 2.62%.[15] This data is surprising since the global data revealed that several factors, including sex hormones and high expression of coronavirus receptors (ACE 2) in men, and also lifestyle, such as higher levels of smoking and drinking among men as compared to women, placed men at higher risk of complications and death.[16]
Multiple comorbidities are related to poorer outcomes. Hospitalized non-survivors had comorbidities. This was also shown in previous studies.[9,17] In our study, the most common comorbidities are diabetes and hypertension, which are similar to the findings of previous studies.[17,18] It was observed in a cohort study of 7337 COVID-19 patients with and without type 2 diabetes that those with type 2 diabetes required more interventions during their hospital stay than those who were not diabetic.[19] The study also showed that patients with uncontrolled blood glucose levels had higher fatality rates. The poor outcomes among diabetic patients infected with COVID-19 may be due to pulmonary dysfunction involving lung volume, pulmonary diffusing capacity, control of ventilation, bronchomotor tone, and noradrenergic bronchial innervation.[20] Additionally, a dysregulated immune response caused by diabetes is responsible for the poor outcomes.[21]
According to new research published in the European Heart Journal, people with high blood pressure had a two-fold greater risk of dying from the coronavirus COVID-19 compared to patients without high blood pressure.[22] Previous studies with univariate analysis reported that hypertension was associated with mortality, but this association disappeared with multivariate analysis.[23,24] Presently, our studies have shown that hypertension is independently associated with increased hospitalization and mortality. Cardiovascular diseases (CVDs) were outlined as a risk factor for poorer outcomes in the first reports.[25-27] Our studies revealed that patients with CVD had a higher risk of hospitalization and death once they got COVID. A report from 99 patients admitted to Northern Italy hospitals, with only 53 patients with CVDs, found higher mortality rates in a univariate analysis.[28] A larger report with 522 patients from 2 Spanish hospitals described an independent association of CVDs only with the combined endpoint of death or respiratory insufficiency.[29] In patients with SARS-CoV-2 infection, underlying CVDs can aggravate the pneumonia and increase the severity of symptoms.[30] Among other comorbidities, chronic obstructive pulmonary disease (COPD) was also linked with higher mortality. Meta-analysis of multiple studies in China found that there was a fourfold increase in mortality in patients with preexisting COPD who were diagnosed with COVID-19.[31,32] We reported a low prevalence of COPD patients in the COVID-19 case series compared to the latest COPD prevalence rate in China, which was 13.6% (95% confidence interval (CI) 12·0–15·2) and the global prevalence of COPD (9–10%).[33,34] The prevalence of COPD in our subjects was low. However, the hospitalization rate and fatality rate were high.
Chronic kidney disease appears to be associated with an increased risk of serious COVID-19 infection; however, our dataset didn’t have patients with CKD.[35] Another risk factor associated with obesity. A study of COVID-19 cases suggests that risks of hospitalization, intensive care unit admission, invasive mechanical ventilation, and death are higher with increasing body mass index (BMI).[36]
This cross-sectional study examining the relationship between COVID-19 outcomes and comorbidities (diabetes, hypertension, cardiovascular diseases, and pulmonary diseases) found that older age, male gender, and the presence of comorbidities were significantly associated with increased hospitalization rates. Among hospitalized patients, comorbidities and advancing age were further linked to a higher risk of mortality.
These findings highlight the critical need for regular monitoring of high-risk individuals, particularly elderly patients with chronic illnesses, to enable early detection of clinical deterioration. Proactive monitoring can help mitigate disease severity and improve survival outcomes. Managing underlying health conditions remains essential in reducing fatality rates.
Furthermore, individuals with comorbidities should be advised to follow stringent preventive measures, including frequent handwashing, use of alcohol-based sanitizers, wearing masks in public spaces, adherence to prescribed medications, and avoiding unnecessary outings. Although the recovery rate in our study was relatively high, a notable proportion (10.75%) of home-isolated patients required hospitalization during the disease course.
Implications: The research shed light on the estimation of mortality, hospitalization, and recovery rates, as well as the risk variables involved. The results of this study may aid policymakers in coming up with different ways to follow up on home-isolated patients. We don’t know much about COVID-19 because it’s a relatively new disease. Our study, on the other hand, has attempted to discuss the risk factors that have been identified with severe COVID-19 disease. Our research shows that even though there is a good recovery rate, significant hospitalization rate, and mortality rate among home-isolated COVID-19 patients, it emphasizes proper monitoring and follow-up of these patients.
Limitations: The research was conducted at a time when the country’s vaccination campaign was just getting started, and progress was slow. Because it was a telephonic interview, the amount of information gathered was limited. Furthermore, only a small fraction of patients had CVD or chronic lung illness. There was no further clinical or laboratory data available to assess the patient’s condition. The status of smoking was also unavailable.
Conclusion
In conclusion, the presence of comorbidities in elderly patients should be recognized as a key factor in future COVID-19 risk stratification models. Enhancing awareness at triage centers and ensuring consistent monitoring of home-isolated COVID-19 patients can significantly reduce morbidity and mortality, ultimately saving lives.
References
- WHO. Archived: WHO Timeline – COVID-19. 2020.
Archived: WHO Timeline – COVID-19 - WHO. Coronavirus Disease (COVID-19) Situation Reports. 2025.
Coronavirus Disease (COVID-19) Situation Reports - Andrews MA, Areekal B, Rajesh KR, et al. First confirmed case of COVID-19 infection in India: A case report. Indian J Med Res. 2020;151(5):490-492. doi:10.4103/ijmr.IJMR_2131_20
PubMed | Crossref | Google Scholar - Covid19india.org. Coronavirus in India: Latest map and case count.
Coronavirus in India: Latest map and case count - Johns Hopkins University & Medicine. Mortality Analyses. 2023.
Mortality Analyses - Dhillon P, Kundu S, Shekhar C, Ram U, Dwivedi L, Dwivedi S, Yadav S, Unisa S. Case-Fatality Ratio and Recovery Rate of COVID-19: Scenario of Most Affected Countries and Indian States. ResearchGate. 2020. doi:10.13140/RG.2.2.25447.68000
Case-Fatality Ratio and Recovery Rate of COVID-19: Scenario of Most Affected Countries and Indian States - Fang Y, Nie Y, Penny M. Transmission dynamics of the COVID-19 outbreak and effectiveness of government interventions: A data-driven analysis. J Med Virol. 2020;92(6):645-659. doi:10.1002/jmv.25750
PubMed | Crossref | Google Scholar - Soares RCM, Mattos LR, Raposo LM. Risk Factors for Hospitalization and Mortality Due to COVID-19 in Espírito Santo State, Brazil. Am J Trop Med Hyg. 2020;103(3):1184-1190. doi:10.4269/ajtmh.20-0483
PubMed | Crossref | Google Scholar - Albitar O, Ballouze R, Ooi JP, Sheikh Ghadzi SM. Risk factors for mortality among COVID-19 patients. Diabetes Res Clin Pract. 2020;166:108293. doi:10.1016/j.diabres.2020.108293
PubMed | Crossref | Google Scholar - Pijls BG, Jolani S, Atherley A, et al. Demographic risk factors for COVID-19 infection, severity, ICU admission, and death: a meta-analysis of 59 studies. BMJ Open. 2021;11(1):e044640. doi:10.1136/bmjopen-2020-044640
PubMed | Crossref | Google Scholar - Asirvatham ES, Sarman CJ, Saravanamurthy SP, Mahalingam P, Maduraipandian S, Lakshmanan J. Who is dying from COVID-19 and when? An Analysis of Fatalities in Tamil Nadu, India. Clin Epidemiol Glob Health. 2021;9:275-279. doi:10.1016/j.cegh.2020.09.010
PubMed | Crossref | Google Scholar - Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239-1242. doi:10.1001/jama.2020.2648
PubMed | Crossref | Google Scholar - Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020;323:e206775. https://doi.org/10.1001/jama.2020.6775
PubMed | Crossref - Global health 5050. The Sex, Gender, and COVID-19 Project. 2022.
The Sex, Gender, and COVID-19 Project: the COVID-10 sex-disaggregated data tracker - Joe W, Kumar A, Rajpal S, Mishra U, Subramanian SV. Equal risk, unequal burden? Gender differentials in COVID-19 mortality in India. J Glob Health Sci. 2020;2(1):e17. doi:10.35500/jghs.2020.2.e17
Crossref | Google Scholar - Bwire GM. Coronavirus: Why Men are More Vulnerable to Covid-19 Than Women? SN Compr Clin Med. 2020;2(7):874-876. doi:10.1007/s42399-020-00341-w
PubMed | Crossref | Google Scholar - Du Y, Tu L, Zhu P, et al. Clinical Features of 85 Fatal Cases of COVID-19 from Wuhan. A Retrospective Observational Study. Am J Respir Crit Care Med. 2020;201(11):1372-1379. doi:10.1164/rccm.202003-0543OC
PubMed | Crossref | Google Scholar - Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. (online ahead of print) 2020. doi:10.1001/jama.2020.1585
Crossref | Google Scholar - Zhu L, She ZG, Cheng X, et al. Association of Blood Glucose Control and Outcomes in Patients with COVID-19 and Pre-existing Type 2 Diabetes. Cell Metab. 2020;31(6):1068-1077.e3. doi:10.1016/j.cmet.2020.04.021
PubMed | Crossref | Google Scholar - Fuso L, Pitocco D, Antonelli-Incalzi R. Diabetic lung, an underrated complication from restrictive functional pattern to pulmonary hypertension. Diabetes Metab Res Rev. 2019;35(6):e3159. doi:10.1002/dmrr.3159
PubMed | Crossref | Google Scholar - Kulcsar KA, Coleman CM, Beck SE, Frieman MB. Comorbid diabetes results in immune dysregulation and enhanced disease severity following MERS-CoV infection. JCI Insight. 2019;4(20):e131774. doi:10.1172/jci.insight.131774
PubMed | Crossref | Google Scholar - Gao C, Cai Y, Zhang K, et al. Association of hypertension and antihypertensive treatment with COVID-19 mortality: a retrospective observational study. Eur Heart J. 2020;41(22):2058-2066. doi:10.1093/eurheartj/ehaa433
PubMed | Google Scholar - Wang D, Yin Y, Hu C, et al. Clinical course and outcome of 107 patients infected with the novel coronavirus, SARS-CoV-2, discharged from two hospitals in Wuhan, China. Crit Care. 2020;24(1):188. doi:10.1186/s13054-020-02895-6
PubMed | Crossref | Google Scholar - Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395(10239):1763-1770. doi:10.1016/S0140-6736(20)31189-2
PubMed | Crossref | Google Scholar - Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846-848. doi:10.1007/s00134-020-05991-x
PubMed | Crossref | Google Scholar - Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. doi:10.1016/S0140-6736(20)30566-3
PubMed | Crossref | Google Scholar - Peng YD, Meng K, Guan HQ, et al. Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCo. Zhonghua Xin Xue Guan Bing Za Zhi. 2020;48(6):450-455. doi:10.3760/cma.j.cn112148-20200220-00105
PubMed | Crossref | Google Scholar - Inciardi RM, Adamo M, Lupi L, et al. Characteristics and outcomes of patients hospitalized for COVID-19 and cardiac disease in Northern Italy. Eur Heart J. 2020;41(19):1821-1829. doi:10.1093/eurheartj/ehaa388
PubMed | Crossref | Google Scholar - Aparisi Á, Amat-Santos IJ, Serrador A, Rodríguez-Gabella T, Arnold R, San Román JA. Current clinical outcomes of tricuspid regurgitation and initial experience with the TricValve system in Spain. Rev Esp Cardiol (Engl Ed). 2020;73(10):853-854. doi:10.1016/j.rec.2020.03.001
PubMed | Crossref | Google Scholar - Srivastava K. Association between COVID-19 and cardiovascular disease. Int J Cardiol Heart Vasc. 2020;29:100583. doi:10.1016/j.ijcha.2020.100583
PubMed | Crossref | Google Scholar - Zhao Q, Meng M, Kumar R, et al. The impact of COPD and smoking history on the severity of COVID-19: A systemic review and meta-analysis. J Med Virol. 2020;92(10):1915-1921. doi:10.1002/jmv.25889
PubMed | Crossref | Google Scholar - Sanyaolu A, Okorie C, Marinkovic A, et al. Comorbidity and its Impact on Patients with COVID-19. SN Compr Clin Med. 2020;2(8):1069-1076. doi:10.1007/s42399-020-00363-4
PubMed | Crossref | Google Scholar - Fang L, Gao P, Bao H, et al. Chronic obstructive pulmonary disease in China: a nationwide prevalence study. Lancet Respir Med. 2018;6(6):421-430. doi:10.1016/S2213-2600(18)30103-6
PubMed | Crossref | Google Scholar - Halbert RJ, Natoli JL, Gano A, Badamgarav E, Buist AS, Mannino DM. Global burden of COPD: systematic review and meta-analysis. Eur Respir J. 2006;28(3):523-532. doi:10.1183/09031936.06.00124605
PubMed | Crossref | Google Scholar - Henry BM, Lippi G. Chronic kidney disease is associated with severe coronavirus disease 2019 (COVID-19) infection. Int Urol Nephrol. 2020;52(6):1193-1194. doi:10.1007/s11255-020-02451-9
PubMed | Crossref | Google Scholar - Kompaniyets L, Goodman AB, Belay B, et al. Body Mass Index and Risk for COVID-19-Related Hospitalization, Intensive Care Unit Admission, Invasive Mechanical Ventilation, and Death – United States, March-December 2020. MMWR Morb Mortal Wkly Rep. 2021;70(10):355-361. doi:10.15585/mmwr.mm7010e4
PubMed | Crossref | Google Scholar
Acknowledgments
We would like to thank Rajiv Gandhi University of Health Sciences, Karnataka, for supporting our study. We would like to express our gratitude to Dr. Praveen Kumar, the head of the Department of Community Medicine, and Dr. O.S. Siddappa, the director of the Shimoga Institute of Medical Sciences. We are indebted to the contribution of G R Manoj, Melisha Rodrigues, Nagavani, Praveen R, Sagar, Shabarish Maddodi, Shamanth H S Acharya, Shabrez Khan, Shravani Edamadaka, Sowmyashree P N, and Sushmitha S.
Funding
This study was funded by the Rajiv Gandhi Institute of Medical Sciences.
Author Information
Corresponding Author:
Sakshi Kumari
Department of Community Medicine
Shimoga Institute of Medical Sciences, Karnataka, India
Email: sakshiupadhyay0912@gmail.com
Co-Authors:
Raghavendraswamy Koppad, Sijin Wilson
Department of Community Medicine
Shimoga Institute of Medical Sciences, Karnataka, India
Sandeep Kumar Maurya
Department of Pharmaceutical Sciences
Dr. Harisingh Gour Central University, Sagar, Madhya Pradesh, India
Authors Contributions
Dr. Sakshi Kumari was responsible for conceptualization, data curation, formal analysis, investigation, project administration, resource management, methodology, data interpretation, manuscript drafting, and contributions to the content, text, and visualization. She also reviewed and revised the manuscript and served as the corresponding author.
Dr. Raghavendra Swamy Koppad performed data analysis, interpreted the results, and supervised the project.
Dr. Harisingh Gour Vishwavidyalaya contributed to the text and content of the manuscript.
Dr. Sijin Wilson also contributed to the content and text of the manuscript.
Ethical Approval
Ethical approval was obtained from the IEC of Shimoga Institute of Medical Sciences, which is recognized by the CDSCO (Registration No. ECR/952/Inst/KA/2017/RR-20) and registered with NECRBHR-DHR (File No. EC/NEW/INST/2020/922).
Conflict of Interest Statement
Not reported
Guarantor
Sakshi Kumari, MBBS, Shimoga Institute of Medical Sciences, India.
DOI
Cite this Article
Sakshi K, Raghavendraswamy K, Sijin W, Sandeep KM. Outcomes of Home Isolated COVID-19 Patients and Risk Factors Associated with the Adverse Outcomes: Longitudinal Retrospective Study in Shimoga, Karnataka. Medtigo J Med. 2025;3(2):e3062323. doi:10.63096/medtigo3062323 Crossref









