Author Affiliations
Abstract
Phytonutrients, also referred to as phytochemicals, are plant-derived compounds with effective health benefits, which include anti-inflammatory, anti-cancer, and antioxidant properties. However, their therapeutic efficacy and potency are frequently limited by low bioavailability, poor solubility, and rapid metabolism. The study, creation, and marketing of nutraceuticals and functional bioactive ingredients have attracted considerable attention on a global scale. The consumption of plant-derived bioactive components has increased over the past 30 years due to the connection between nutrition, health, and disease. Fruits, vegetables, cereals, and other plant-derived meals contain secondary metabolites known as phytochemicals, which are bioactive nutritional plant-derived molecules that may offer significant health advantages beyond basic sustenance. In addition to their cholesterol-lowering, antithrombotic, and antifungal properties, the phytochemicals have antimicrobial, antifungal, and antioxidant properties. They may also reduce the risk of major chronic diseases like cancer, genetic disorders, diabetes mellitus, high blood pressure, osteoporosis, and psychotic disorders. Nano-encapsulation is a cutting-edge technology that addresses these challenges by enhancing the delivery, stability, and bioactivity of phytonutrients. Therefore, this review explores the methods of nano-encapsulation, which include liposome encapsulation, polymeric nanoparticles, solid lipid nanoparticles, nanostructured lipid carriers, nanoemulsions, and micelles, their principles, methods, and their application in optimizing the therapeutic potential of phytonutrients. Furthermore, it highlights recent advancements, current challenges, and future directions for integrating nano-encapsulation technologies into clinical and nutraceutical settings.
Keywords
Phytonutrients, Bioactive compounds, Nanotechnology, Nanoencapsulation, Therapeutic potential.
Introduction
Encapsulation, which forms an outer membrane or coating of one material over another, is a common approach for protecting and preserving bioactive, volatile, and easily degradable compounds from biochemical and thermal destruction. It can also be used to cover up offensive smells and flavors. The technique of encapsulation was initially created about 60 years ago to wrap solids, liquids, and gaseous substances. Under certain conditions, the coating enables the controlled release of chemicals at a specific targeted site.[1,2] The encapsulated components, also known as the coated material, might be pure substances or a combination.[3]
Phytonutrients, also known as phytochemicals, are bioactive secondary metabolites that are naturally found in plants, which provide significant health benefits beyond basic nutrition. These secondary metabolites or compounds, such as flavonoids, carotenoids, alkaloids, and polyphenols, possess significant capabilities for their preventive and therapeutic roles, including antioxidant, anti-cancer activities, and anti-inflammatory effects in chronic diseases. Despite all the significant potential of phytonutrients, the clinical application of phytonutrients is often hampered by their low or poor water solubility, which results in limited or low absorption, instability in the gastrointestinal environment, low bioavailability, high rate of systemic elimination, and susceptibility to environmental contamination and degradation on exposure to sunlight, oxygen, and heat.[4] These challenges faced by phytonutrients that possess significant therapeutic value reduce their efficacy and potency, particularly in targeted therapeutic contexts.
Nano-encapsulation has enhanced the effective distribution of therapeutic phytochemicals to target locations, which has become a game-changing approach nowadays. The use of the following nanoscale carriers —liposomes, polymeric nanoparticles (PNPs), and solid lipid nanoparticles (SLNs) — is used to improve absorption in the gastrointestinal tract, prevent degradation, and release of regulated phytonutrients.[5] The utilization of nano-encapsulation paves the way to concentrate the medicinal benefits of phytonutrients at the target site while reducing systemic adverse side effects and also enhancing their possibility of targeted administration.
Due to the recent Innovations and advancements in nanotechnology, there is a significant improvement in the development of reliable and efficient delivery systems for phytonutrients to a specific target site. Research by Karthikeyan et al.[6] has reported that curcumin-loaded Poly(lactic-co-glycolic acid) (PLGA) nanoparticles have been shown to enhance bioavailability and exhibit superior cytotoxic effects against cancer cells compared to free curcumin. Research by Himanath et al.[7] revealed that nano emulsions containing carotenoids like lycopene have demonstrated improved antioxidant activity and bioaccessibility in clinical and preclinical studies. These reports have shown the transformative potential and efficiency of advances in nano-encapsulation technologies, which increase the therapeutic effects and benefits of phytonutrients in the therapies of some chronic diseases. This review article aimed at providing an in-depth exploration of nano-encapsulation as an advanced strategy for enhancing the therapeutic effect and potential of phytonutrients, dives and highlight the principles and methods of nano-encapsulation, it also explores the potential of nanotechnology to address bioavailability and stability challenges of phytonutrients in a living system, it examines the current limitations of nano-encapsulation technologies which includes scale-up and safety concerns. Finally, it outlines future directions for integrating these recent advancements into clinical and nutraceutical applications.
Methodology
Research design: This review uses a methodical strategy to find, assess, and compile pertinent research.
Data sources: A systematic review of peer-reviewed articles, chapters, books, patents, and conference proceedings was conducted to identify research published between 2017 and 2025 in scholarly databases such as Google Scholar, Web of Science & Scopus, ScienceDirect, NCBI, PsycINFO, and PubMed. The following keywords, such as “Phytonutrients, Bioactive compounds, Nanotechnology, Nanoencapsulation, Therapeutic potential, and Nano-encapsulation techniques specifically for phytonutrients,” were used for the search.
Inclusion and exclusion criteria
Inclusion: Empirical studies, review articles, and theoretical papers that explicitly link nanoencapsulation and phytonutrients.
Exclusion: Studies focusing solely on nano-encapsulation or unrelated to nanotechnology or phytonutrient applications, and articles with inconclusive or non-peer-reviewed findings.
Data extraction: The key information extracted during the search was study objectives, methodology, key findings, and conclusions. A total of 15 research papers met the criteria for this review after the eligibility assessment.
Nano-Encapsulation: Mechanism and Methods
Mechanism of Nano-Encapsulation:
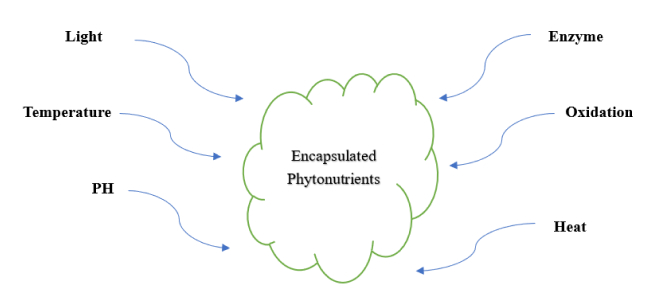
Figure 1: Encapsulated phytonutrient containing bioactive compound protected from external damage
The term nano-encapsulation refers to the enclosing of a phytonutrient, which is an active compound, within nanometer-scale carriers to enhance their stability and delivery to a specific target site. This advanced strategy enhances the therapeutic efficacy of bioactive compounds present in phytonutrients by addressing challenges like low solubility, poor stability, and rapid degradation. The encapsulation of a phytonutrient protects the active compound from environmental degradation by either oxidation, enzymes, light,/or heat. It improves solubility and dispersion in aqueous media of the active compounds and facilitates controlled, targeted, and sustained release at the desired site of action. This section gives a detailed exploration of the mechanism underlying nano-encapsulation and describes various methods used to develop nanoscale delivery systems for bioactive compounds present in phytonutrients. The primary mechanisms involved in nano-encapsulation include:
- Protection from environmental degradation: Environmental elements like temperature, oxygen, light, pH, and enzymatic activity that can reduce the bioactivity of active compounds present in phytonutrients are protected by nano-encapsulation of the phytonutrients with suitable nanocarriers. There is a report on curcumin’s quick oxidation and degradation in aqueous conditions, which can be stopped by encapsulating it in SLNs.[8]
- Enhanced solubility and absorption: Low water solubility of bioactive compounds present in phytonutrients and other food substances results in lower absorption and bioavailability of beneficial bioactive compounds in the living system, which is also a common characteristic of phytonutrients such as flavonoids and carotenoids, in which low solubility restricts their absorption in the digestive system. These challenges can be overcome by distributing hydrophobic substances in aqueous fluids using nanoscale carriers, such as nano emulsions and nanoencapsulation, which improves solubility.[7]
- Regulated and extended release: The therapeutic effects of bioactive compounds present in phytonutrients can be enhanced, regulated, and extended by the utilization of nanocarriers, which can be designed to release them at a specific location. A study showed that active chemicals are delivered to acidic tumor settings using PNPs made for pH-responsive release.[6]
- Targeted distribution: Disease conditions often produce receptors that can be targeted for therapeutic purposes. Targeting receptors or tissues can be achieved with functionalized nanocarriers that possess ligands or surface modifications that can easily bind with low binding energy to the surface of receptors or tissues, thereby improving treatment efficacy and potency and reducing off-target effects in a system. A cancer study shows nanoparticles coated with resveratrol that target folate have demonstrated increased absorption in cancer cells.[9]
Methods of Nano-Encapsulation:
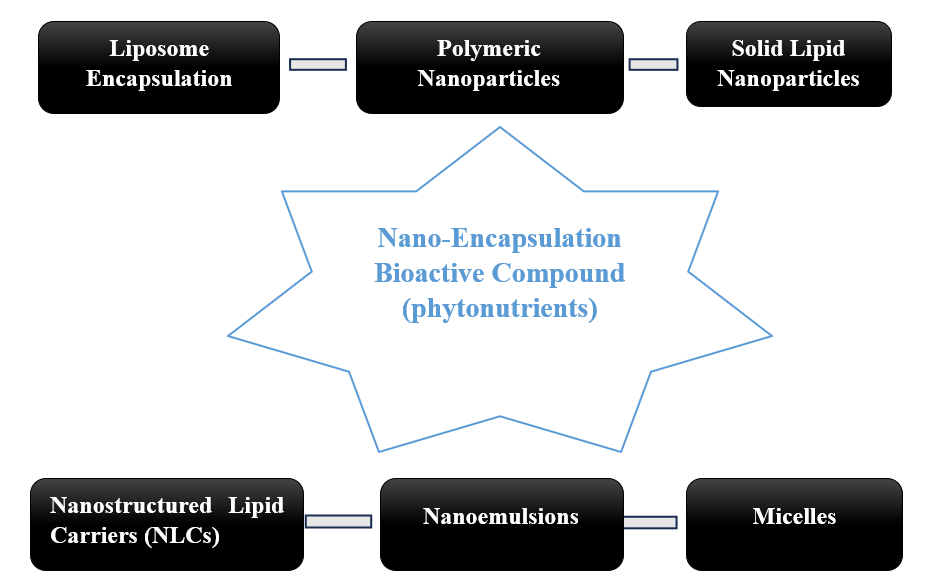
Figure 2: Different methods of nano-encapsulation of bioactive compounds
The process of enclosing bioactive substances such as phytonutrients in a particular nanometer-sized container is known as nano-encapsulation. These nanometer-carriers enable a specific, targeted distribution and regulated release, thereby enhancing the solubility, bioavailability, and protection of bioactive compounds from environmental deterioration by other substances. The bioactive compounds present in the phytonutrients, the preferred distribution mode, and the intended target site are all factors that influence the encapsulating method selection. The stability and bioavailability challenges related to phytonutrient delivery to a specific target site can be addressed in a variety of ways by using advanced nano-encapsulation techniques. In nanoencapsulation, the application and intended goal determine which approach is best, as each has distinct benefits and drawbacks. The therapeutic potential of bioactive compounds present in phytonutrients can be increased by ongoing advancements in encapsulation techniques, which allow their successful application in clinical and nutraceutical contexts. The most popular techniques for nano-encapsulation are examined in detail below, together with their benefits, drawbacks, and uses, which are backed by current research.
Liposome encapsulation: Liposomes are spherical vesicles with one or more phospholipid bilayers encircling an aqueous center. Both hydrophilic (in the aqueous core) and hydrophobic (in the lipid bilayer) substances can be encapsulated by them. Liposomes are widely recognized as a significant and advanced drug delivery technology at the nanoscale, offering appealing qualities such as a distinctive bilayer shape that mimics the cellular membrane, ease of preparation, and good biocompatibility. Over the past few decades, considerable effort has been invested in developing drug delivery systems based on liposomes. Liposome-encapsulated medication candidates have been studied for their longer duration of therapeutic efficacy and lower toxicity. Various academic and corporate research groups have established liposomal encapsulation of hydrophilic and hydrophobic small molecule therapies, as well as other large molecules. As of now, there is a growing number of liposomal-based treatments that have received Food and Drug Administration (FDA) approval and are now undergoing clinical studies. These treatments have a variety of uses in antiviral, antibacterial, and anticancer treatments.[10]
It has been reported that quercetin-loaded liposomes showed enhanced stability and bioavailability, with significant anti-inflammatory effects.[4] Liposome encapsulation offers several advantages, including biocompatibility, biodegradability, and versatility in encapsulating a wide range of compounds. However, it also has limitations, such as limited storage stability due to lipid oxidation, and leakage of encapsulated compounds may occur during storage.
Methods of preparation:
- Thin-film hydration: This involves the use of an aqueous solution containing the phytonutrient to hydrate a phospholipid film, which serves as a nonfouling surface.
- Reverse-phase evaporation: It is carried out by organic solvents, which dissolve lipids and combine them with an aqueous phase. The solvent in the medium is then removed, generating vesicles.
- Microfluidic techniques: This involves the use of microfluidic devices to provide precise and accurate control over the size and composition of liposomes used.
Polymeric nanoparticles (PNPs): PNPs are colloidal systems composed of natural or synthetic polymers such as chitosan, PLGA, or polycaprolactone (PCL), and represent another cutting-edge technique for nanoencapsulation. PNPs can be created by directly polymerizing monomers (emulsion polymerization, surfactant-free emulsion polymerization, mini-emulsion polymerization, micro-emulsion polymerization, and microbial polymerization) or by dispersing preformed polymers (nanoprecipitation, emulsification solvent evaporation, emulsification solvent diffusion, and salting-out). The desired characteristics of the particles and their intended applications dictate the manufacturing process for nanoparticles.[11] They can include both hydrophilic and hydrophobic phytonutrients, making them incredibly adaptable.
The primary purpose of encapsulation processes employing PNPs is to enhance the bioactive compounds found in phytonutrient solubility, stability, bioavailability, and therapeutic potential. PNPs provide regulated or targeted release and protect sensitive materials from environmental degradation by some external and internal factors. These nanoparticles are helpful in a range of applications, such as cancer therapy, anti-inflammatory treatments, and nutraceutical delivery.
Method of preparation:
PNPs are made using a variety of methods, each of which is customized to the unique characteristics of the polymer and the encapsulated component. These methods include:
- Emulsion-Solvent evaporation: This process involves dissolving the polymer and the bioactive component in an organic solvent, emulsifying them in an aqueous phase with a surfactant, and then evaporating the organic solvent. This technique is widely applied for encapsulating hydrophobic phytonutrients like curcumin, as curcumin-loaded PLGA nanoparticles prepared by this method showed enhanced and effective bioavailability and anti-cancer properties.[6]
- Ionic gelation: It is also one of the mild techniques that involves the interaction between a positively charged polymer like chitosan and a negatively charged cross-linker like tripolyphosphate to form nanoparticles. This technique is suitable for encapsulating heat-sensitive and hydrophilic bioactive compounds present in phytonutrients. It was reported that quercetin-loaded chitosan nanoparticles exhibited improved stability and controlled release.[4]
- Nanoprecipitation: It is another method of PNPs in the encapsulation process, in which polymer and bioactive compounds in phytonutrients are dissolved in a solvent, which is then added to a non-solvent. Upon stirring, the polymer will be precipitated and form nanoparticles. This method can be used for both hydrophilic and hydrophobic compounds, like polyphenol-loaded nanoparticles prepared via nanoprecipitation, which showed enhanced antioxidant activity.[12]
- Spray-drying: This process creates nanoparticles by atomizing a solution of the polymer and bioactive component found in phytonutrients into tiny droplets, which are subsequently quickly dried in a hot air stream. This method is particularly well-suited for the large-scale production of encapsulated bioactive compounds in phytonutrients. It was reported that spray-dried lutein nanoparticles demonstrated improved stability in gastrointestinal environments, which leads to stability and bioavailability of the encapsulated bioactive compound present in phytonutrients.[13]
- Double emulsion-solvent evaporation: This method involves creating a water-in-oil-in-water emulsion. This process allows encapsulation of hydrophilic compounds within a polymeric matrix. It is commonly used for hydrophilic drugs or phytonutrient bioactive compounds, such as encapsulation of water-soluble vitamins using PLGA nanoparticles for controlled release.
Solid lipid nanoparticles (SLNs): SLNs are advanced nanocarriers derived from solid lipids that remain solid at room and body temperatures and are presented as a means of delivering both corrective dynamic therapy and water-soluble medication. Nanoparticles are mostly colloidal particles with an approximate size range of 10 to 1000 nm. They are designed to improve drug delivery and reduce lethality and are derived from produced distinctive polymers. They have evolved as a flexible alternative to liposomes for the delivery of medications. They are made from synthetic or unique polymers and are best suited to enhance the delivery of sedatives, bioactive compounds present in phytonutrients, and reduce lethality.[14] An essential tool in medicines, nutraceuticals, and cosmetics is the utilization of SLNs, which are known for their biocompatibility, biodegradability, and capacity to improve the stability and bioavailability of encapsulated bioactive substances.
Preparation methods:
- Hot homogenization: In this process, the lipid content in the nanoparticles is melted, mixed with the bioactive compound, and finally, the entire content is emulsified under high pressure.
- Cold homogenization: This process involves solidification, milling, and homogenizing of the lipid with a cold aqueous surfactant solution.
- Solvent emulsification-evaporation: This involves dissolving lipids in an organic solvent, emulsifying in water, and finally, the nanoparticles are formed as the solvent evaporates.
- Microemulsion technique: Lipids, surfactants, and water are mixed to form a microemulsion, which solidifies upon cooling.
The advantages of SLNs include controlled and targeted release that provides sustained delivery, improving therapeutic efficacy, and reducing dosing frequency and effect. It enhances stability, which protects sensitive bioactive compounds in phytonutrients from oxidation, hydrolysis, and light degradation. It also improved bioavailability by overcoming solubility limitations of hydrophobic bioactive compounds.
SLNs have been reported to possess several applications, such as in cancer therapy, where resveratrol-loaded SLNs have been shown to have enhanced bioavailability and significant anti-cancer activity.[8] SLNs’ encapsulation technique is also applied in anti-inflammatory therapies; a report showed that SLNs encapsulating curcumin using the solid lipid nanoparticles technique exhibited the highest anti-inflammatory effects and controlled drug release.[15] SLNs’ encapsulation technique is applied in nutraceuticals and cosmetics. It was reported that Lycopene-loaded SLNs demonstrated improved stability and antioxidant capacity, making them suitable for functional foods. Also, SLNs are used in skincare products to deliver vitamins and antioxidants, providing enhanced stability and prolonged activity.[9]
NLCs: SLNs synthesis served as the foundation for the investigation of a novel lipid nanoparticulate drug delivery method. One interesting method of drug delivery using lipid nanocarriers is the incorporation of the drug into different biocompatible lipids that are synthesized at the nanoscale. Lipid nanocarriers are being developed as a substitute for emulsions, liposomes, and PNPs. Additionally, the second-generation lipid carriers known as nanostructured lipid nanocarriers were created to address issues with solid lipid nanoparticles and are employed in several treatment modalities. They offer higher encapsulation efficiency and stability compared to SLNs. Although their usefulness for hydrophilic medicines is now well established, NLCs were initially thought to be best suited for the delivery of lipophilic pharmaceuticals. Lipids’ biocompatibility is what led to their development as a potentially effective medication delivery system. It was discovered to have better qualities than other lipid compositions.[16] The preparation methods of NLCs are very similar to SLNs, but with the inclusion of liquid lipids in the formulation to prevent lipid crystallization. NLCs possess the advantages of enhanced encapsulation capacity and improved stability and drug release profiles. It has been reported that EGCG-loaded NLCs demonstrated enhanced anti-inflammatory and antioxidant properties.[5]
Nano emulsions: Combinations of water, oil, and surfactants that create droplets with a size of nanometers are known as nano emulsions. They work very well to distribute bioactive compounds present in hydrophobic phytonutrients. A family of emulsions known as nanoemulsions has droplets that range in size from 10 to 500 nm. Nano emulsions have small droplet sizes that provide remarkable features, including durable stability and customizable rheology. They are used in a wide variety of biomedical and clinical applications. Pharmaceutical formulations for topical, ocular, intravenous, and other administration methods are frequently developed using nanoemulsions. Nanoemulsions can also be used as a template to create hydrophobic bioactive medicinal component nanocrystals. They are also used to create biomaterials like encapsulated nanodroplets and biomimetic microparticles because of their customizable composition and characteristics.[17]
Preparation methods:
Nano emulsions are prepared using a variety of techniques, which can be broadly divided into high-energy and low-energy procedures.
- High-Energy methods: These methods of nanoemulsion encapsulation use mechanical energy to break down oil droplets into nanoscale sizes. These methods include High-Pressure Homogenization (HPH). In this process of nanoemulsion encapsulation, a coarse emulsion is forced through a narrow nozzle under high pressure of 100–1500 bar. The turbulence and shear forces reduce droplet sizes to the nanoscale. This method is suitable for heat-sensitive compounds and is mostly scalable for industrial production. A report shows that nanoemulsions loaded with curcumin have been prepared using high-pressure homogenization (HPH), which results in enhancing the solubility and anti-inflammatory properties of curcumin.[18] Ultrasonication is also a high-energy method of nanoemulsions encapsulation, which involves the use of high-frequency sound waves ranging from 20 to 50 kHz. The high-frequency sound waves are applied to a coarse emulsion, causing cavitation, which reduces droplet size. It was reported that quercetin nanoemulsions prepared by ultrasonication exhibited improved antioxidant and anti-inflammatory effects.[19] It was also reported that nanoemulsions encapsulating vitamin E demonstrated prolonged release and enhanced bioavailability.[20]
- Low-Energy methods: These methods utilize the physicochemical properties and changes such as temperature or composition of the entire system to form nano emulsions without the use of high mechanical energy as in high-energy method and these methods include Phase Inversion Temperature (PIT) method; in this method the system is heated to a particular temperature where the surfactant’s affinity for oil and water changes eventually causing a phase inversion. Upon cooling, nanoemulsions are formed due to the low interfacial tension. The advantage of this method is that it is energy-efficient and suitable for heat-stable compounds. It was reported that nanoemulsions of essential oils prepared by the PIT method showed enhanced antimicrobial activity, and lycopene nanoemulsions improved antioxidant capacity and bioavailability in vivo.[7,21] Spontaneous Emulsification (SE) is also a low-energy method of nanoemulsion encapsulation in which a water-immiscible phase containing oil and a surfactant is added to an aqueous phase under mild stirring. The spontaneous diffusion of surfactant molecules forms nano-sized droplets. This method is simple, cost-effective, and does not require heat or high energy. A report shows nanoemulsions of resveratrol prepared using SE showed enhanced antioxidant properties.[22] Another low-energy method of nanoemulsion encapsulation is the Solvent Evaporation Method. In this process, the oil phase containing the active compound and surfactant is dissolved in an organic solvent and emulsified in an aqueous phase. The organic solvent is evaporated, leaving behind nano-sized droplets. This is ideal for hydrophobic compounds. It was reported that nanoemulsions encapsulating carotenoids prepared by this method demonstrated improved bioavailability.[15]
Micelles: Micelles are self-assembled structures formed by amphiphilic molecules. They are mostly used to encapsulate hydrophobic compounds in their core, enhancing solubility and stability. Polymeric micelles are one of the promising nanocarrier systems, especially if used in the encapsulation process for drug and gene delivery. The efficacy of several micellar preparations of anticancer drugs has been shown through extensive preclinical and clinical research. Compared to long-circulating liposomes, polymeric micelles may have several advantages, such as tissue penetration, controlled drug release, and reduced toxicity from hypersensitivity reactions and hand-foot syndrome. Important features of the polymeric micelles as drug carriers, including particle size, stability, loading capacity, and drug release kinetics, can be altered using the topologies and physicochemical properties of the constituent block copolymers.[24]
The preparation methods of micelles involve solvent evaporation, in which amphiphilic molecules and bioactive compounds present in phytonutrients are dissolved in a solvent, and the mixture is finally evaporated to form micelles. Micelles have the advantage of high solubilization efficiency and the ability to cross biological barriers such as the blood-brain barrier. It was reported that micellar formulations of curcumin demonstrated improved bioavailability and systemic anti-inflammatory effects.[17] But this method of nanoencapsulation has limited stability under certain environmental conditions.
Applications of nano-encapsulation in phytonutrient delivery:
- Anti-inflammatory applications: Phytonutrients in nano capsules are more effective at suppressing inflammatory pathways. The bioavailability of phytochemicals has been impacted by the creation of nano-phytomedicines, the attachment of phytomedicines to polymers, modifications to their surface and permeability characteristics. New formulations that are packed with phytomedicines have been developed and have shown incredible outcomes. For instance, curcumin nanoparticles decreased the generation of cytokines in inflammatory disorders by blocking the NF-κB pathway.[25]
- Anti-cancer applications: Nano-encapsulation facilitates targeted delivery to tumor cells, increases the bioavailability of anti-cancer phytonutrients, and minimizes systemic toxicity. The bioactive compounds present in phytochemicals of interest can be encapsulated, embedded, or adsorbed on these nano-scale carriers. Additionally, compared to non-nano formulations of the same anti-cancer phytochemical, the nano-size facilitates simple infiltration to cancer cells at a greater rate, lowering the dosage needed to achieve an effective anti-cancer impact. These nano-delivery vehicles’ ability to function on numerous surfaces can guarantee ligand decorating, which can result in tailored phytochemical delivery to cancer cells and reduced cytotoxicity to healthy tissues. It has been reported that nano-encapsulation of resveratrol increased its cytotoxicity against breast cancer cells while reducing off-target effects.[9]
- Neuroprotective effects: Some of the factors that contribute to neurodegenerative diseases (NDs) include protein deposition, oxidative stress, hereditary variables, and viral infections, which lead to disorders of the neurons that cause gradual damage and malfunction. Neuronal loss and damage can impact the central and peripheral nervous systems and result in movement impairments and cognitive failure. Neuronal degeneration can affect a patient’s quality of life, family social network, and other aspects of their existence. Alzheimer’s disease (AD), Parkinson’s disease (PD), and their concomitant disorders, such as Huntington’s disease (HD), Multiple Sclerosis (MS), and Amyotrophic Lateral Sclerosis (ALS), are among the most prevalent NDs.[26] Novel therapeutic agents must be developed immediately to avoid neurodegeneration, as most currently available therapy modalities are aimed at symptomatic relief. The transport of bioactive compounds in phytonutrients across the blood-brain barrier has been made possible via nanotechnology by a nano-encapsulation process, which creates new opportunities for the treatment of neurodegenerative diseases. It was reported that EGCG-loaded nanoparticles demonstrated neuroprotective effects in Parkinson’s disease models.[27]
- Cardiovascular benefits: To protect bioactive substances such as polyphenols, essential oils, peptides, oligonucleotides, and antioxidants from oxidation or lysis by external factors like pH, temperature changes, and light exposure, nano-encapsulation has been used extensively in the development of new pharmaceuticals and as a high-tech food ingredient. This prolongs the half-lives of the active components, which have a major positive impact on cardiovascular health and guarantee the stability of these compounds. Astaxanthin and other carotenoids were enhanced by nano-encapsulation, which also decreased oxidative stress in cardiovascular disorders.[28]
Challenges in nano-encapsulation:
- Challenge in scaling-up and manufacturing: This arises because of substantial financial and technological barriers in converting laboratory-scale nano-encapsulation methods to industrial production, as it involves significant cost and technical hurdles.
- Toxicity issues: One of the major advantages of nanocarriers is that they are biocompatible, but a thorough assessment of their long-term safety is necessary to ensure safety.
- Regulatory obstacles: This issue involves the government authorities that are involved in nutraceutical and pharmaceutical regulations. The lack of standardized guidelines for nanotechnology in nutraceuticals and pharmaceuticals poses challenges for market approval.
- Stability problems: Transitions of produced nano-encapsulated phytonutrients are a major concern. Maintaining the stability of nano-encapsulated phytonutrients during storage and transport poses a serious challenge, as some environmental factors can affect their integrity.
Future directions: The creation of stimuli-responsive or smart nanocarriers that release phytonutrients in response to temperature, pH, or specific enzymes, or the integration of these factors, is strongly encouraged. Secondly, the development of hybrid systems that integrate multiple nanocarriers, including lipid-based and polymeric systems, to enhance delivery and therapeutic efficacy. By using nano-encapsulation to give phytonutrients to patients according to their unique genetic and metabolic profiles, personalized treatment is also greatly encouraged. To ensure sustainability, there is a need to create nanocarriers using environmentally benign materials and green synthesis techniques.
Conclusion
Encapsulation is a tried-and-true method of preserving bioactive substances. Nano-encapsulation offers several advantages, including higher bioavailability of bioactive substances, enhanced stability, a sustained release profile, and improved protection. A paradigm shift in bioactive compounds, particularly in phytonutrient delivery, is achieved through nano-encapsulation, which tackles the major problems of stability, targeted action, and bioavailability. Phytonutrients can attain improved therapeutic potential by utilizing nanotechnology, which makes them promising options for the treatment of several chronic illnesses that are very effective with low side effects. Despite current obstacles, developments in bioengineering, biotechnology, nanotechnology, and material science offer hope for the broad use of nano-encapsulation in the nutraceutical and healthcare sectors. Future studies should investigate new uses for this game-changing technology while addressing scalability and safety concerns.
References
- Desai KG, Park H. Recent developments in microencapsulation of food ingredients. Drying Technol. 2005;23(7):1361-1394. doi:10.1081/DRT-200063478
Crossref | Google Scholar - Shishir MRI. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci Technol. 2018;78:34-60. doi:10.1016/j.tifs.2018.05.018
Crossref | Google Scholar - Saifullah M, Ferdowsi R, Rahman MR, Vuong Q. Micro and nano encapsulation, retention and controlled release of flavor and aroma compounds: a critical review. Trends Food Sci Technol. 2019;91:72-85. doi:10.1016/j.tifs.2019.02.030
Crossref | Google Scholar - Patra S, Pradhan B, Nayak R, et al. Dietary polyphenols in chemoprevention and synergistic effect in cancer: Clinical evidences and molecular mechanisms of action. Phytomedicine. 2021;90:153554. doi:10.1016/j.phymed.2021.153554
PubMed | Crossref | Google Scholar - Chen Z, Farag MA, Zhong Z, et al. Multifaceted role of phyto-derived polyphenols in nanodrug delivery systems. Adv Drug Deliv Rev. 2021;176:113870. doi:10.1016/j.addr.2021.113870
PubMed | Crossref | Google Scholar - Karthikeyan A, Senthil N, Min T. Nanocurcumin: A Promising Candidate for Therapeutic Applications. Front Pharmacol. 2020;11:487. doi:10.3389/fphar.2020.00487
PubMed | Crossref | Google Scholar - Himanath G, Ramesh S, Preetha R, Valiathan S. Nanoemulsion with coconut oil and soy lecithin as a stable delivery system for lycopene and its incorporation into yogurt to enhance antioxidant properties and maintain quality. ACS Food Sci Technol. 2021;1(9):1654-1664. doi:10.1021/acsfoodscitech.1c00117
Crossref | Google Scholar - Paolino D, Mancuso A, Cristiano MC, et al. Nanonutraceuticals: The New Frontier of Supplementary Food. Nanomaterials (Basel). 2021;11(3):792. doi:10.3390/nano11030792
PubMed | Crossref | Google Scholar - Senthil Kumar C, Thangam R, Mary SA, Kannan PR, Arun G, Madhan B. Targeted delivery and apoptosis induction of trans-resveratrol-ferulic acid loaded chitosan coated folic acid conjugate solid lipid nanoparticles in colon cancer cells. Carbohydr Polym. 2020;231:115682. doi:10.1016/j.carbpol.2019.115682
PubMed | Crossref | Google Scholar - Filipczak N, Pan J, Yalamarty SSK, Torchilin VP. Recent advancements in liposome technology. Adv Drug Deliv Rev. 2020;156:4-22. doi:10.1016/j.addr.2020.06.022
PubMed | Crossref | Google Scholar - Pulingam T, Foroozandeh P, Chuah JA, Sudesh K. Exploring Various Techniques for the Chemical and Biological Synthesis of Polymeric Nanoparticles. Nanomaterials (Basel). 2022;12(3):576. doi:10.3390/nano12030576
PubMed | Crossref | Google Scholar - Enaru B, Socaci S, Farcas A, et al. Novel Delivery Systems of Polyphenols and Their Potential Health Benefits. Pharmaceuticals (Basel). 2021;14(10):946. doi:10.3390/ph14100946
PubMed | Crossref | Google Scholar - Shi L, Zhou J, Guo J, Gladden I, Kong L. Starch inclusion complex for the encapsulation and controlled release of bioactive guest compounds. Carbohydr Polym. 2021;274:118596. doi:10.1016/j.carbpol.2021.118596
PubMed | Crossref | Google Scholar - Lingayat VJ, Zarekar NS, Shendge RS. Solid lipid nanoparticles: a review. Nanosci Nanotechnol Res. 2017;4(2):67-72. doi:10.12691/nnr-4-2-5
Crossref | Google Scholar - Rostamabadi H, Falsafi SR, Jafari SM. Nanoencapsulation of carotenoids within lipid-based nanocarriers. J Control Release. 2019;298:38-67. doi:10.1016/j.jconrel.2019.02.005
PubMed | Crossref | Google Scholar - Salvi VR, Pawar P. Nanostructured lipid carriers (NLC) system: a novel drug targeting carrier. J Drug Deliv Sci Technol. 2019;51:255-267. doi:10.1016/j.jddst.2019.02.017
Crossref | Google Scholar - Gupta A. Nanoemulsions. In: Nanoparticles for Biomedical Applications. Elsevier; 2020:371-384. doi:10.1016/B978-0-12-816662-8.00021-7
Crossref | Google Scholar - Divyashree S, Sharath J, Janhavi P, Deepashree S, Muthukumar SP. Curcumin and its derivatives as nutraceuticals: an update. In: Studies in Natural Products Chemistry. Vol 77. Elsevier; 2023:135-162. doi:10.1016/B978-0-323-91294-5.00005-1
Crossref | Google Scholar - Song R, Lin Y, Li Z. Ultrasonic-assisted preparation of eucalyptus oil nanoemulsion: Process optimization, in vitro digestive stability, and anti-Escherichia coli activity. Ultrason Sonochem. 2022;82:105904. doi:10.1016/j.ultsonch.2021.105904
PubMed | Crossref | Google Scholar - A N, Kovooru L, Behera AK, Kumar KPP, Srivastava P. A critical review of synthesis procedures, applications, and future potential of nanoemulsions. Adv Colloid Interface Sci. 2021;287:102318. doi:10.1016/j.cis.2020.102318
PubMed | Crossref | Google Scholar - Ren G, Sun Z, Wang Z, Zheng X, Xu Z, Sun D. Nanoemulsion formation by the phase inversion temperature method using polyoxypropylene surfactants. J Colloid Interface Sci. 2019;540:177-184. doi:10.1016/j.jcis.2019.01.018
PubMed | Crossref | Google Scholar - Bayón-Cordero L, Alkorta I, Arana L. Application of Solid Lipid Nanoparticles to Improve the Efficiency of Anticancer Drugs. Nanomaterials (Basel). 2019;9(3):474. doi:10.3390/nano9030474
PubMed | Crossref | Google Scholar - Ordoubadi M, Gregson FKA, Wang H, et al. On the particle formation of leucine in spray drying of inhalable microparticles. Int J Pharm. 2021;592:120102. doi:10.1016/j.ijpharm.2020.120102
PubMed | Crossref - Kesharwani SS, Kaur S, Tummala H, Sangamwar AT. Multifunctional approaches utilizing polymeric micelles to circumvent multidrug resistant tumors. Colloids Surf B Biointerfaces. 2019;173:581-590. doi:10.1016/j.colsurfb.2018.10.022
PubMed | Crossref | Google Scholar - Sharma N, Behl T, Singh S, et al. Targeting Nanotechnology and Nutraceuticals in Obesity: An Updated Approach. Curr Pharm Des. 2022;28(40):3269-3288. doi:10.2174/1381612828666221003105619
PubMed | Crossref | Google Scholar - Paramanick D, Singh VD, Singh VK. Neuroprotective effect of phytoconstituents via nanotechnology for treatment of Alzheimer disease. J Control Release. 2022;351:638-655. doi:10.1016/j.jconrel.2022.09.058
PubMed | Crossref | Google Scholar - González LF, Bevilacqua LE, Naves R. Nanotechnology-Based Drug Delivery Strategies to Repair the Mitochondrial Function in Neuroinflammatory and Neurodegenerative Diseases. Pharmaceutics. 2021;13(12):2055. doi:10.3390/pharmaceutics13122055
PubMed | Crossref | Google Scholar - Nematollahi HR, Hosseini R, Bijani A, et al. Interleukin 10, lipid profile, vitamin D, selenium, metabolic syndrome, and serum antioxidant capacity in elderly people with and without cardiovascular disease: Amirkola health and ageing project cohort-based study. ARYA Atheroscler. 2019;15(5):233-240. doi:10.22122/arya.v15i5.1623
PubMed | Crossref | Google Scholar
Acknowledgments
The author wishes to acknowledge the entire research team for working tirelessly in preparing this paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Information
Corresponding Author:
Yakubu Magaji Yuguda
Department of Science Laboratory Technology
Federal Polytechnic, Kaltungo, Gombe State, Nigeria
Email: magajiyakubu@gmail.com
Co-Authors:
Hauwa Mohammad Galadima, Abubakar Adamu Jauro, Mohammad Adamu Jauro
College of Medical Sciences, Gombe State University, Gombe State, Nigeria
Amina Mohammad Yushau
Department of Biochemistry
Bayero University, Kano, Nigeria
Authors Contributions
Yakubu Magaji Yuguda contributed to the abstract, methodology, mechanism of nanoencapsulation, application of nanoencapsulation in phytonutrient delivery, and conclusion, as well as reviewing, editing, and approving the manuscript. Hauwa Mohammad Galadima was responsible for the introduction, challenges, and future direction sections. Abubakar Adamu Jauro wrote on the method of nanoencapsulation, specifically liposome encapsulation, polymeric nanoparticles, and solid lipid nanoparticles. Mohammad Adamu Jauro contributed to the method of nanoencapsulation, focusing on nanostructured lipid carriers and nano emulsions. Amina Mohammad Yushau wrote on the method of nanoencapsulation, covering spray drying and micelles.
Ethical Approval
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Conflict of Interest Statement
The authors declare no conflicts of interest.
Guarantor
None
DOI
Cite this Article
Yuguda YM, Galadima HM, Jauro AA, Jauro MA, Yushau AM. Nano-Encapsulation of Phytonutrients for Enhanced Therapeutic Potential: A Literature Review. medtigo J Pharmacol. 2025;2(3):e30612314. doi:10.63096/medtigo30612314 Crossref









