Author Affiliations
Abstract
Background: Takayasu arteritis (TAK) is a rare, chronic, idiopathic large-vessel vasculitis that primarily affects the aorta and its major branches. It commonly presents in young women of childbearing age with nonspecific systemic symptoms and vascular complications such as absent pulses, hypertension, and claudication. Diagnosis is challenging and relies heavily on imaging studies, as laboratory markers are nonspecific. Management typically includes high-dose corticosteroids and immunosuppressive agents, with biologic therapies and vascular interventions reserved for refractory or severe cases.
Objective: To report a rare case of a young post-partum patient diagnosed with Takayasu arteritis, complicated by heart failure, highlighting notable vascular abnormalities.
Case presentation: A 21-year-old female was admitted with symptoms of acute decompensated heart failure, initially attributed to peripartum cardiomyopathy. During hospitalization, she developed severe abdominal pain and cardiovascular instability with an absent left radial pulse. Further evaluation led to a diagnosis of Takayasu arteritis with multi-vessel involvement. Treatment with standard heart failure therapy and high-dose corticosteroids with immunosuppressive therapy resulted in improved cardiac function.
Conclusion: This case underscores the complexity of diagnosing and managing a 21-year-old female with concurrent Takayasu arteritis and acute decompensated heart failure. It highlights the critical need for early identification and comprehensive evaluation of cardiovascular conditions in post-partum patients, as timely intervention can lead to significant improvements in outcomes. The successful treatment and positive response to therapy in this case reinforce the importance of adhering to guideline-directed medical therapy.
Keywords
Takayasu arteritis, Peripartum cardiomyopathy, Vasculitis, Heart failure, Paroxysmal nocturnal dyspnea.
Introduction
TAK is a rare inflammatory condition affecting the aorta and its branches.[1] It causes inflammation, leading to artery narrowing, blockage, or weakening. This can result in various symptoms, including fever, fatigue, high blood pressure, weak pulses, claudication, visual disturbances, headaches, and heart failure.[2,3] A systematic review and meta-analysis estimated the global incidence at approximately 1.11 per million person-years (95% confidence interval (CI) 0.70–1.76), with a notably higher incidence in women, approximately 2.01 per million person-years (95% CI 1.39–2.90).[4] Diagnosis involves blood tests and imaging studies.[5] Treatment focuses on reducing inflammation with medications like corticosteroids and immunosuppressants and may include surgery for severe cases, such as angioplasty, stent placement, or aneurysm repair.[6] Early diagnosis and treatment are crucial for managing this serious condition.
We present a case of a patient with Takayasu arteritis and describe the clinical presentation, treatment, and outcomes.
Case Presentation
Our patient is a 21-year-old primiparous lady who was relatively well till 6 months into her pregnancy, at which time she was diagnosed with pregnancy-induced hypertension, which was managed at high-risk antenatal care (ANC) with methyldopa resulting in medically induced vaginal delivery at 37 weeks of gestation to affect 2.7kg male alive neonate and she received post-delivery MgSO4 for 48 hours. Her blood pressure normalized after delivery, and she was discharged from medications.
Three weeks after delivery to a healthy baby, she presented to our hospital with shortness of breath, orthopnea requiring three pillows, paroxysmal nocturnal dyspnea (PND), and bilateral lower extremity swelling for which she had transthoracic echocardiography with and an ejection fraction (EF) of 25 – 30% after which she was diagnosed with peri-partum cardiomyopathy (PPCM). Upon diagnosis, she required intensive care unit (ICU) admission and was managed for cardiogenic shock with noradrenaline drip, followed by medical ward transfer for optimization of guideline-directed medical therapy (GDMT) for heart failure and diuresis. She was discharged with Metoprolol succinate 25mg daily, spironolactone 12.5mg daily, Enalapril 5mg twice daily, dapagliflozin 10mg daily, and furosemide 40mg twice daily after 2 weeks of medical ward stay.
Two months after discharge, she was readmitted with acute decompensated heart failure (HF) precipitated by medication non-compliance of one week, after she presented with exacerbated dyspnea, with associated nonproductive cough and history of generalized body swelling progressing from lower extremities to abdomen, orthopnea of 3 pillows, intermittent PND, and loss of appetite. She denied any history of chest pain, fever, abdominal pain, night sweats, hemoptysis, joint pains, rash, hair loss, weight loss, oral or vaginal ulcers, visual changes, eye pain or redness, headaches, or syncope or contact with chronic cougher. During this admission, while responding well to diuresis and GDMT, she developed severe peri-umbilical colicky abdominal pain and left upper extremity numbness. Physical examination revealed an impalpable left radial pulse and unrecordable blood pressure on the left side, raising concerns for vasculitis and possible arterial thromboembolism. For these, she was investigated with inflammatory markers and showed an elevated erythrocyte sedimentation rate (ESR) of 44 mm/hr and C-reactive protein (CRP) of 99.3 mg/L. Her gene X-pert from 15ml of bronco alveolar lavage (BAL) was non diagnostic. She was also investigated with complete blood count, electrolytes, thyroid function test (TFT), complement level, anti-nuclear antibody (ANA), renal function test (RFT), and urine analysis (Table 1). An abdominal ultrasound with Doppler shows a dilated abdominal aorta above the superior mesenteric artery, measuring 4 cm in diameter and having an irregular wall morphology. The celiac trunk and left renal artery ostium appeared narrowed.
There was diffuse thickening of the aortic arch and descending thoracic aorta, with significant stenosis of the brachiocephalic and subclavian arteries on thoracic aortography and circumferential mural thickening and focal narrowing of the abdominal aorta, including the renal and celiac arteries on abdominal angiography (Figure 1). The echocardiogram shows that the Left-sided cardiac chambers were dilated, with global systolic dysfunction of the left ventricle, with an estimated EF of 30-35%, and moderate functional mitral regurgitation was also noted (Figure 1).
| Complete blood count | Day 1 | Day 3 | Day 7 | Laboratory reference range |
| White blood cell | 9.3 | 8.74 | 6.8 | 3.9 – 10.1 x 10 3 /mm 3 |
| Neutrophil | 78.7 | 68.6 | 61.5 | 30.4 – 74.6 % |
| Lymphocyte | 8.7 | 20.5 | 28.6 | 17.8 – 61.5 % |
| Hemoglobin | 13.3 | 12.7 | 13.7 | 10.4 – 14.7 g/dl |
| Hematocrit | 37.5 | 34.6 | 36.7 | 34.4 – 48.3 % |
| Mean cell volume | 81.7 | 79.4 | 78.8 | 74.3 – 98.3 fl |
| Mean cell hemoglobin | 28.9 | 29 | 37.2 | 25.7 – 33.6 pg |
| Platelet count | 506 | 380 | 420 | 100 – 300 x 10 3 /µL |
| ESR | 44 | – | – | <20 mm/hr |
| CRP | 99.3 | – | – | 0–10 mg/L |
| Creatinine | 0.7 | – | – | 0.5 – 0.9 mg/dl |
| Urea | 22 | – | – | 10 – 50 mg/dl |
| Sodium | 133 | – | – | 136 – 145 mmol/l |
| Potassium | 4.1 | – | – | 3.5 – 5.5 mmol/l |
| Chloride | 103 | – | – | 98 – 107mmol/l |
| Urine analysis | Unremarkable | |||
| Gene X-pert from BAL | Not detected | |||
| Complement – 3 | 1.4 | – | – | 0.9 – 1.8 g/L |
| Complement – 4 | 0.51 | – | – | 0.1 – 0.4 g/L |
| ANA | 6.5 | – | – | Reactive > 48 |
| TSH | 0.345 | – | – | 0.274 – 4.256 micIU/ml |
| Free T3 | 2.33 | – | – | 2.1 – 4.16 pg/ml |
| Free T4 | 17.9 | – | – | 8.7 – 16.5 pg/ml |
Table 1: Laboratory results
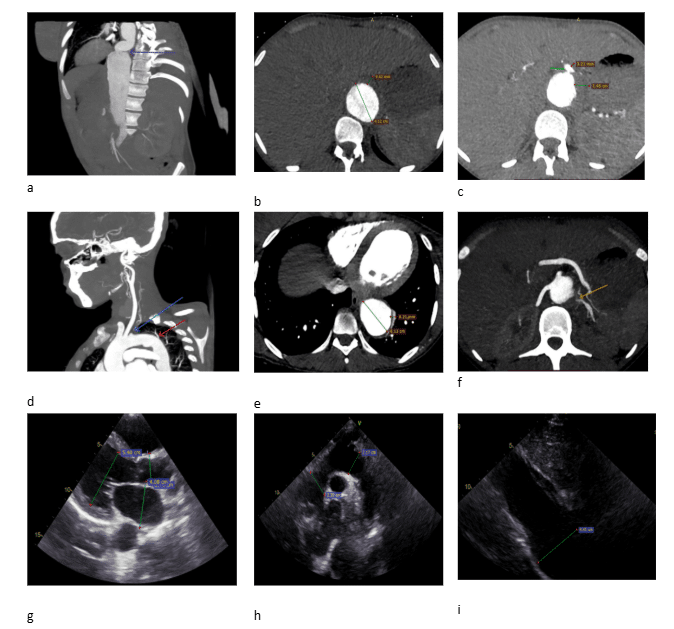
Figure a: Abdominal computed tomography (CT) angiography with sagittal section shows abdominal aorta with tortuosity (violet arrow)
Figure b: Abdominal CT angiography transverse section at kidney level, there is maximum dilatation of the abdominal aorta measuring 4.12cm with circumferential mural thickness of 3.84 mm with intimal irregularity.
Figure c: Abdominal CT angiography transverse section at celiac artery origin shows focal narrowing of celiac artery (green arrow) with circumferential thickening
Figure d: Sagittal section of CT angiography of upper extremity and aortography shows diffuse focal circumferential thickening of the thoracic aorta extending to the brachiocephalic trunk with luminal stenosis of the subclavian arteries (blue arrow) and tortuosity of the thoracic aorta (red arrow)
Figure e: Transverse section at heart level shows enlarged thoracic aorta with focal wall thickening
Figure f: Abdominal CT angiography transverse section at the level of renal artery origin shows decreased left renal artery caliber in the proximal 1/3 of the artery (yellow arrow)
Figure g: 2-dimensional (2D) Transthoracic Echo (TTE) on long axis shows dilated left atrium, left ventricle, and aorta at the aortic sinus during diastole
Figure h: 2D TTE suprasternal notch view shows normal measurement
Figure i: 2D TTE on subxiphoidal and inferior vena cava (IVC) view shows dilated abdominal aorta measuring 4.4cm
Case Management
Subsequently, after stabilization and initial investigation, she was diagnosed with TAK with multi-vessel involvement complicated with dilated cardiomyopathy. She managed GDMT of HF and started on high-dose prednisolone (40 mg/day, 1 mg/kg/day). After 2 weeks of initiation of prednisolone, methotrexate 15 mg/week, and folic acid 5 mg three times a week were added. Then, after 6 weeks of initial Echo, TTE was repeated and showed significant EF improvement of >50% with improved subjective nonspecific symptoms, and on physical examination, the pulses on the left radial artery were palpable, and blood pressure (BP) was measurable and equal to the right side. Prednisolone tapering started after she took it for a total duration of 6 weeks, with continuation of methotrexate and GDMT for HF.
She had a follow-up after 2 months of starting prednisolone tapering, at which time RFT, liver enzymes, complete blood count (CBC), acute phase reactant ESR, and CRP were done. All were in the normal range. With a plan to maintain a low-dose steroid (Prednisolone 5mg po daily) and continue methotrexate 15mg po weekly with folic acid 5mg po 3x/week, she was appointed to come after 3 months to have follow-up at both rheumatology and cardiology sides.
Discussion
TAK is a rare, chronic inflammatory disease primarily affecting large vessels such as the aorta and its branches.[1-3] It predominantly affects young females and presents with a wide spectrum of clinical manifestations.[4]
Some of the rare clinical features and presentation of TAK include heart failure with valvular involvement, persistent cough, and malignant hypertension, refractory TAK with severe coronary artery involvement, and TAK can also present with acute aortic dissection.[7-12] In some case reports, TAK can present with reversible cerebral vasoconstriction syndrome (RCVS), recurrent headaches, and transient neurological deficit, and TAK’s initial presentation could also be stroke.[13,14] In one case report, TAK complicated by peripartum cardiomyopathy after a young patient presented with signs and symptoms of HF with associated limb BP measurement difference and absent pulse, highlighting the overlapping cardiovascular burden in pregnancy and the postpartum period, requiring vigilant monitoring and multidisciplinary care.[15]
Pregnancy in women with TAK is associated with increased risks of maternal and fetal complications, particularly when the disease is active or involves critical vascular territories.[16] Some of maternal complications include hypertension, preeclampsia, and cardiovascular complication including HF, aortic regurgitation, and aneurysm formation, which may rupture during pregnancy, cerebrovascular accident including stroke, due to obstetric indications and disease-related complications, cesarean sections are more frequent in TAK patients, with rates up to 42% some of fetal complications include intrauterine growth restriction (IUGR) which reported in 15% to 40% of pregnancies in women with TAK, often associated with hypertension and vascular insufficiency, low birth weight is observed in approximately 16% of cases, correlating with maternal disease activity and hypertension, miscarriage rates range from 9% to 11%, with fetal loss reported in 1% to 12% of cases, particularly in active disease.[17-21] To minimize this complication, pre-pregnancy counseling is mandatory, and it is recommended to be pregnant during remission, and the choice of medication to control disease activity during pregnancy should be compatible with pregnancy, including corticosteroids and azathioprine.[17]
One case report presented a complex case of tuberculosis (TB) lymphadenitis with concurrent TAK in a 14-year-old girl, highlighting the diagnostic and therapeutic challenges of managing overlapping infectious and inflammatory conditions.[22] This is more important in developing nations like ours in which TB is prevalent, for our patients we try to rule out disseminated TB by doing chest CT imaging, doing gene X-pert from BAL, and doing follow-up chest CT imaging after 6 weeks of prednisolone and methotrexate therapy in these all there was no evidence of TB.
Conclusion
The reviewed case underscores the heterogeneous nature of TAK and its potential for severe complications. A multidisciplinary approach involving rheumatologists, cardiologists, radiologists, and vascular surgeons is essential for optimal outcomes. Early recognition and tailored treatment strategies remain the cornerstone of management.
References
- Jennette JC. Overview of the 2012 revised International Chapel Hill Consensus Conference nomenclature of vasculitides. Clin Exp Nephrol. 2013;17(5):603-606. doi:10.1007/s10157-013-0869-6 PubMed | Crossref | Google Scholar
- Hall S, Barr W, Lie JT, Stanson AW, Kazmier FJ, Hunder GG. Takayasu arteritis. A study of 32 North American patients. Medicine (Baltimore). 1985;64(2):89-99. Takayasu arteritis. A study of 32 North American patients
- Schmidt J, Kermani TA, Bacani AK, et al. Diagnostic features, treatment, and outcomes of Takayasu arteritis in a US cohort of 126 patients. Mayo Clin Proc. 2013;88(8):822-830. doi:10.1016/j.mayocp.2013.04.025 PubMed | Crossref | Google Scholar
- Rutter M, Bowley J, Lanyon PC, Grainge MJ, Pearce FA. A systematic review and meta-analysis of the incidence rate of Takayasu arteritis. Rheumatology (Oxford). 2021;60(11):4982-4990. doi:10.1093/rheumatology/keab406 PubMed | Crossref | Google Scholar
- Almasi S, Asadian S, Hosseini L, et al. Myocardial and Vascular Involvement in Patients with Takayasu Arteritis: A Cardiovascular MRI Study. Diagnostics (Basel). 2023;13(23):3575. doi:10.3390/diagnostics13233575 PubMed | Crossref | Google Scholar
- Maz M, Chung SA, Abril A, et al. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Giant Cell Arteritis and Takayasu Arteritis. Arthritis Rheumatol. 2021;73(8):1349-1365. doi:10.1002/art.41774 PubMed | Crossref | Google Scholar
- Serra R, Butrico L, Fugetto F, et al. Updates in Pathophysiology, Diagnosis and Management of Takayasu Arteritis. Ann Vasc Surg. 2016;35:210-225. doi:10.1016/j.avsg.2016.02.011 PubMed | Crossref | Google Scholar
- Godil S, Saqi B, Godil K, Sabzwari SRA, Rajeswaran Y. Catastrophic cardiac complications of Takayasu’s arteritis. Cureus. 2020;12(4):e9142. doi:10.7759/cureus.9142 PubMed | Crossref | Google Scholar
- Ameerah Y, Moghrabi S, Awadghanem A, Hanbali R, Maree M. An unusual case of Takayasu arteritis presenting with persistent cough in a young female patient: A case report. Radiol Case Rep. 2024;19(2):651-653. doi:10.1016/j.radcr.2023.11.009 PubMed | Crossref | Google Scholar
- Sierra LV, Binzenhöfer L, Schulze-Koops H, Lackermair K, Massberg S, Lüsebrink E. A patient with Takayasu arteritis presenting with malignant hypertension: a case report. Eur Heart J Case Rep. 2023;7(6):ytad263. doi:10.1093/ehjcr/ytad263 PubMed | Crossref | Google Scholar
- Cobilinschi CO, Grădinaru E, Săulescu I, et al. Refractory Takayasu’s Arteritis with Severe Coronary Involvement-Case Report and Literature Review. J Clin Med. 2023;12(13):4394. doi:10.3390/jcm12134394 PubMed | Crossref | Google Scholar
- Guo J, Zhang G, Tang D, Zhang J. A case report of Takayasu arteritis with aortic dissection as initial presentation. Medicine (Baltimore). 2017;96(45):e8610. doi:10.1097/MD.0000000000008610 PubMed | Crossref | Google Scholar
- Lee SH, Lee MJ, Kang B, et al. A case report of patient with Takayasu’s arteritis complicated by reversible cerebral vasoconstriction syndrome. J Rheum Dis. 2013;20(3):194-198. doi:10.4078/jrd.2013.20.3.194 Crossref | Google Scholar
- Oli P, Poudel P, Kc S, Thapa N, Kc A. Takayasu arteritis presenting as a stroke in young: a case report. Ann Med Surg (Lond). 2024;86(2):1085-1088. doi:10.1097/MS9.0000000000001582 PubMed | Crossref | Google Scholar
- Nagajothi N, Mejia JA, Raghunathan K, et al. Takayasu arteritis presenting as postpartum cardiomyopathy. Exp Clin Cardiol. 2008;13(4):189-191. Takayasu arteritis presenting as postpartum cardiomyopathy
- Gupta L, Misra DP, Ahmed S, et al. Poor obstetric outcomes in Indian women with Takayasu arteritis. Adv Rheumatol. 2020;60(1):17. doi:10.1186/s42358-020-0120-6 PubMed | Crossref | Google Scholar
- Dasari P, Gummadi HS. Pregnancy outcome in Takayasu arteritis. BMJ Case Rep CP. 2021;14(5):e238014. doi:10.1136/bcr-2020-238014 PubMed | Crossref | Google Scholar
- Velásquez Giraldo I, Torres Saavedra FA, Muñoz Vahos CH, et al. Pregnancy outcomes in patients with Takayasu’s arteritis: Case series. Rev Colomb Reumatol (Engl Ed). 2021;28(3):213-217. doi:10.1016/j.rcreue.2020.04.006 Crossref | Google Scholar
- Gudbrandsson B, Wallenius M, Garen T, Henriksen T, Molberg Ø, Palm Ø. Takayasu Arteritis and Pregnancy: A Population-Based Study on Outcomes and Mother/Child-Related Concerns. Arthritis Care Res (Hoboken). 2017;69(9):1384-1390. doi:10.1002/acr.23146 PubMed | Crossref | Google Scholar
- Comarmond C, Saadoun D, Nizard J, Cacoub P. Pregnancy issues in Takayasu arteritis. Semin Arthritis Rheum. 2020;50(5):911-914. doi:10.1016/j.semarthrit.2020.08.001 PubMed | Crossref | Google Scholar
- Abisror N, Mekinian A, Hachulla E, et al. Analysis of risk factors for complications and adverse obstetrical outcomes in women with Takayasu arteritis: a French retrospective study and literature review. Clin Rheumatol. 2020;39(9):2707-2713. doi:10.1007/s10067-020-05024-4 PubMed | Crossref | Google Scholar
- Tadesse YG, Mulisa MD, Beyene ET, Adugna BA. Complex Case of Tuberculosis Lymphadenitis with Concurrent Takayasu Arteritis in a 14-Year-Old Girl from Ethiopia. Open Access Rheumatol. 2023;15:231-236. doi:10.2147/OARRR.S438427 PubMed | Crossref | Google Scholar
Acknowledgments
We would like to express our gratitude to the patient for giving us consent to publish this case report.
Funding
There is no source of funding for this manuscript.
Author Information
Corresponding Author:
Fitsum Negusse Assefa
Department of Internal Medicine
St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
Email: fitsum0604270@gmail.com
Co-Authors:
Ayto Addisu Negash
Department of Emergency Medicine and Critical Care Division
St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
Selamawit Tilahun Raru
Department of Internal Medicine, Pulmonology and Critical Care Division
St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
Alebachew Girum, Abel Mureja Argaw
Department of Internal Medicine and Cardiology Division
St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
Birhanu Desyibelew Demelash
Department of Internal Medicine and Rheumatology Division
College of Health Sciences Addis Ababa University, Addis Ababa, Ethiopia
Authors Contributions
All authors contributed equally to conceiving the idea, designing the methodology, acquiring the data, writing the manuscript, and reviewing the literature.
Ethical Approval
Ethical clearance was obtained from the institutional review board of St. Paul’s Hospital Millennium Medical College and is available to the editors upon request.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report.
Conflict of Interest Statement
The authors declare no conflict of interest.
DOI
Cite this Article
Fitsum NA, Ayto AN, Selamawit TR, Alebachew G, Abel MA, Birhanu DD. Multivessel Involvement in Postpartum Takayasu Arteritis: Vascular Complications and Dilated Cardiomyopathy: A Case Report and Review of Literature. medtigo J Med. 2025;3(2):e3062328. doi:10.63096/medtigo3062328 Crossref









