Author Affiliations
Abstract
Background: Elevated intracranial pressure (ICP) is a critical factor contributing to morbidity and mortality in stroke patients, emphasizing the need for timely and accurate monitoring. Conventional methods for measuring ICP are invasive and carry significant risks, underscoring the importance of finding reliable non-invasive alternatives.
Purpose: This study investigates the efficacy of using magnetic resonance imaging (MRI) to measure the optic nerve sheath diameter (ONSD) as a non-invasive indicator of elevated ICP among stroke patients in the Southwestern region of Nigeria.
Materials and Methods: The research involved 150 stroke patients aged 18 and above who underwent brain MRI scans. High-resolution T2-weighted images were used to measure the ONSD 3 mm behind the globe, and the results were analyzed for correlations with ICP levels.
Results: The study found a positive correlation (r = 0.85, p < 0.001) between MRI-measured ONSD and invasively measured ICP. The findings also showed that the mean ONSD was slightly higher in males (5.800 mm) than in females (5.720 mm), with minimal variation, indicating the method’s reliability across genders. The results support using MRI-based ONSD measurement as a dependable non-invasive method for detecting elevated ICP in stroke patients.
Conclusion: These findings suggest that ONSD measurement could serve as a valuable clinical tool for ICP monitoring, warranting further research to confirm its potential integration into routine practice.
Keywords
Intracranial pressure, Optic nerve sheath diameter, Magnetic resonance imaging, Stroke, Non-invasive monitoring.
Introduction
The optic nerve, also known as the second cranial nerve (CN II), plays a crucial role in transmitting vital visual information. It begins with the retinal ganglion cells within the retina.[1,2] The optic nerves from both eyes come together at a point called the optic chiasm. At this point, roughly half of the nerve strands from each eye proceed on the same side of the brain, whereas the other half cross over to connect filaments from the other eye.[3] The optic nerve is secured by the same meningeal layers that secure the brain: the dura mater, arachnoid mater, and pia mater. The subarachnoid space around the nerve contains cerebrospinal liquid.[4] The optic nerve can be separated into four parts: intraocular, infraorbital, intracanalicular, and intracranial.[2] The optic chiasm is found over the pituitary organ and underneath the hypothalamus. The continuation of the optic nerve strands past the chiasm shapes the optic tracts, which at that point extend to the lateral geniculate body of the thalamus and other visual centres within the brain.[4] The optic nerve sheath is a protective covering that encompasses the optic nerve, giving a basic bolster and carrying cerebrospinal liquid (CSF).[5] It is composed of three meningeal layers, comparable to those encompassing the brain and spinal cord. The dura mater is the outermost layer of the optic nerve sheath; it is intense and sinewy, giving vigorous security.[6] The dura mater is continuous with the sclera at the optic circle. The arachnoid mater is the center layer, which is more slender and more sensitive than the dura mater. It features a web-like appearance and makes a difference to pad the optic nerve.[7] The pia mater is the innermost significant layer, closely attached to the surface of the optic nerve. The pia mater takes after the shapes of the optic nerve filaments.[3]
The ONSD has become a non-invasive marker for indirectly estimating ICP, and because the optic nerve sheath is directly connected to the intracranial subarachnoid space, alterations in intracranial pressure can impact its diameter.[8] Several studies have shown that there is a positive correlation between ONSD and ICP. When ICP increases, the diameter of the optic nerve sheath tends to increase because of the pressure transmitted from the subarachnoid space surrounding the optic nerve. The ONSD measurement can help in the early detection of increased ICP, which guides treatment decisions and interventions.[9]
A stroke, also known as a cerebrovascular accident (CVA), happens when there is an interruption or reduction of blood supply to the brain. This leads to cell death and subsequent neurological deficits. There are two main types of strokes: ischemic stroke, caused by the blockage of blood vessels supplying the brain, and hemorrhagic stroke, caused by bleeding into or around the brain.[5,10] This interruption in blood flow leads to the rapid death of brain cells, usually within minutes. When brain cells perish or are impaired because of a stroke, various symptoms manifest in the parts of the body controlled by these affected brain cells. In individuals who have suffered a stroke, it is crucial to closely monitor ICP as heightened ICP can result in secondary brain injury and have a detrimental impact on neurological outcomes. Traditional methods for assessing ICP, such as invasive intraventricular or intraparenchymal monitoring, present inherent risks and may not always be viable in various clinical settings.[11] In contrast, non-invasive measurement of the ONSD through MRI has emerged as a promising alternative for indirectly evaluating ICP.[10,11] MRI is now commonly used in patients with suspected brain lesions. The anatomical structures and borders of the optic nerve and nerve sheaths are well delineated in MRI scans. Consequently, measuring the ONSD using MRI can offer dependable insights into ICP levels without the need for additional procedures.[12] The timely identification of elevated ICP can exert a substantial impact on patient care, potentially averting further neurological damage and enhancing patient outcomes.[11] While the ONSD has emerged as a potential indicator of increased ICP in various neurological conditions, there is a lack of research specifically investigating the utility of ONSD measurement in predicting increased ICP in stroke patients. Current methods for monitoring ICP are invasive and carry inherent risks. Therefore, there is a critical need for non-invasive techniques to detect elevated ICP in Stroke patients. This study aimed to assess the feasibility and effectiveness of using MRI measurement of ONSD as a non-invasive method for determining increased ICP in stroke patients.
Methodology
This prospective longitudinal research was conducted over a period of three months (April to June 2024) at a Multisystem Hospital in the Southwestern region of Nigeria. Established in 2018, the hospital features 450 inpatient beds and a fully equipped radio-diagnostic unit with two operational 1.5 Tesla MRI machines. These machines serve patients from across Southwestern Nigeria, excluding Lagos, performing over a thousand MRI scans annually. Moreover, the 1.5 Tesla machines are the most advanced available in West Africa during this study. This study followed the ethical standards outlined in the 1964 declaration of Helsinki.[13] Ethical approval was granted by the ethics and research committee of the institution. Additionally, permission and support were obtained from the radiology department’s managing consultants. Informed written consent was obtained from all subjects before the examination, where the reason for the study, benefits, possible effects, and stages of examination were explained to the subjects.
One hundred and forty-nine (149) consenting adults from southwestern nigeria, who underwent brain MRI examination and were diagnosed with stroke by clinical examination and imaging studies, were included in this study. Adults (≥18 years) diagnosed with either ischemic or hemorrhagic stroke were included in the study. Patients with pre-existing optic nerve or orbital disease, contraindications to MRI (e.g., pacemakers, claustrophobia), and previous neurosurgical interventions were excluded from the research.
All MRI examinations were conducted on 149 adults using the 1.5 Tesla neurovascular technology (NT) MRI (GE) system, the most advanced model available in West Africa. Routine MRI studies were carried out for pre-contrast and contrast studies, and slice thickness was 3 mm, field of view 230, matrix 320×320. Before the commencement of the imaging procedure, the patient’s clinical history was taken, and heights and weights were taken using a Leica mechanical height and weight scale model 15 R. Values were then recorded into an Excel sheet for calculation of the body mass index (BMI) as kg/m2. A T2-weighted MRI sequence was obtained by the patient lying supine on the MRI couch with their head covered with the head coil and legs outside. Measurement of ONSD was taken 3 mm behind the globe in both eyes. Average measurements from both eyes were recorded for analysis. Measurements were made electronically on the monitor of the MRI machine using the Advantage workstation Software (Version 2019r) by consensus of two radiologists. Measurements were then compared for variability due to age, gender, and BMI.
The data obtained from the study was analyzed using the statistical package for social sciences (SPSS) software version 23 (SPSS Inc., Chicago, Illinois, USA) and microsoft excel 2016. A normality test was first carried out to determine the distribution of data. Descriptive analysis was performed to determine mean, median, range, and standard deviation, while parametric tools were used to carry out inferential statistics to establish relationships and comparisons. Charts and Tables were used to visualize the results.
Results
A total of 149 adults were recruited for this study. The survey data indicates that 66.4% (99) of the population consists of females, while 33.6% (50) are males. Figure 1 (Chart A-D) shows the normal distribution of data for ONSD, BMI, Age, and ICP, respectively. The correlation was tested using Pearson’s correlation statistics between ONSD, ICP, and BMI. There was a statistically significant strong positive relationship between ONSD and ICP (r = 0.8, p = 0.00) as shown in Fig. 2. However, no significant relationship seems to exist between ONSD and BMI (r = – 0.005, p = 0.95) as shown in Fig. 3. The study compared the average values for ONSD, ICP, and BMI between male and female populations using a paired sample T-test, with a significance level (p-value) set at 0.05. The findings revealed that there were no statistically significant differences between the average values for males and females for ONSD (p-value = 0.57), ICP (p-value = 0.23), and BMI (p-value = 0.78).
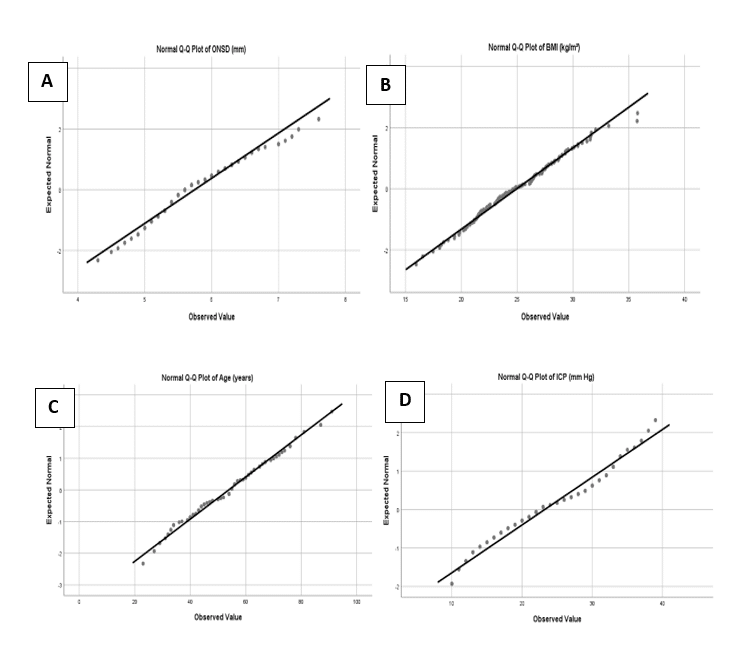
Figure 1: Kolmogorov-Smirnov test for normality
| Parameter | Mean | ||
| Male (N = 50) | Female (N = 99) | Overall (N = 149) | |
| Age | 58 | 52 | 54 |
| BMI | 25.68 ± 3.46 | 25.47 ± 4.20 | 24.97 ± 3.75 |
| ICP | 24.08 ± 7.5 | 22.32 ± 7.7 | 23.23 ± 8.04 |
| ONSD | 5.8± 0.74 | 5.72 ± 0.59 | 5.75 ± 0.67 |
Table 1: Summary of descriptive statistics
As shown in Table 1, the average age of the study population was 54 years, with the average age for males (58 years) being higher than that of females (52 years). Similarly, the mean ICP and ONSD were 23.23 mmHg and 5.75 mm, respectively, with the average for males being slightly higher than that of the females. BMI was calculated using the recorded height and weight of the study population, the result shows a <1% difference between the male and female mean BMI.
| Parameters | r-value | p-value |
| ICP | 0.807 | 0.000 |
| BMI | -0.005 | 0.95 |
Table 2: Correlation between ONSD and other parameters (ICP and BMI)
Table 2 shows the correlation between ONSD and two other parameters: ICP and BMI. The r-value of 0.807 indicates a strong positive correlation between ONSD and ICP. This means that as ICP increases, ONSD also tends to increase. The p-value of 0.000 (typically considered significant if < 0.05) indicates that this correlation is statistically significant. Therefore, the relationship between ONSD and ICP is highly likely to be real and not due to random chance. The r-value of -0.005 indicates no significant correlation between ONSD and BMI. This suggests that changes in BMI do not have any meaningful impact on ONSD. The p-value of 0.95, which is much higher than the conventional threshold for statistical significance (0.05), supports the conclusion that there is no significant relationship between ONSD and BMI.
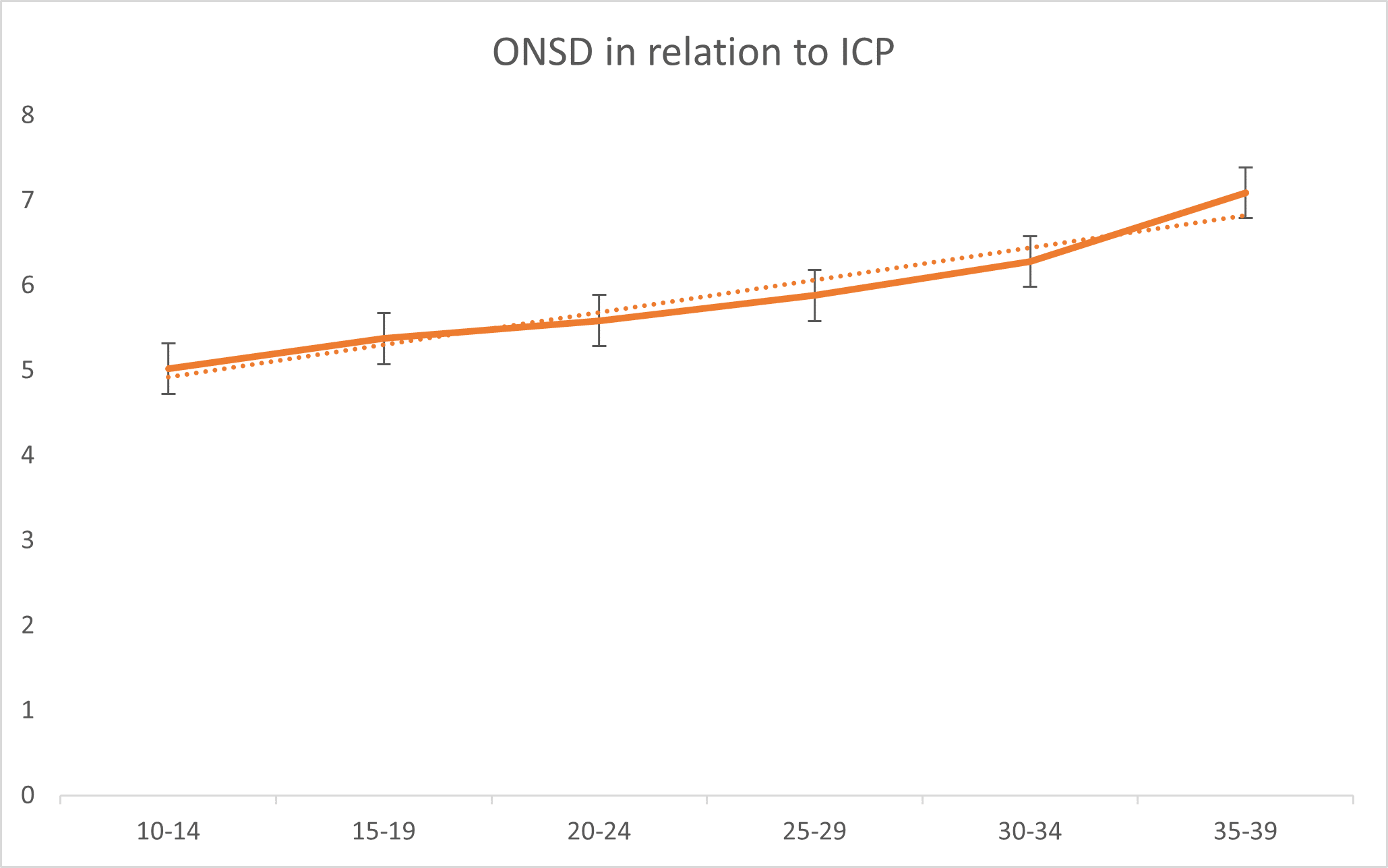
Figure 2: Relationship between ONSD and ICP
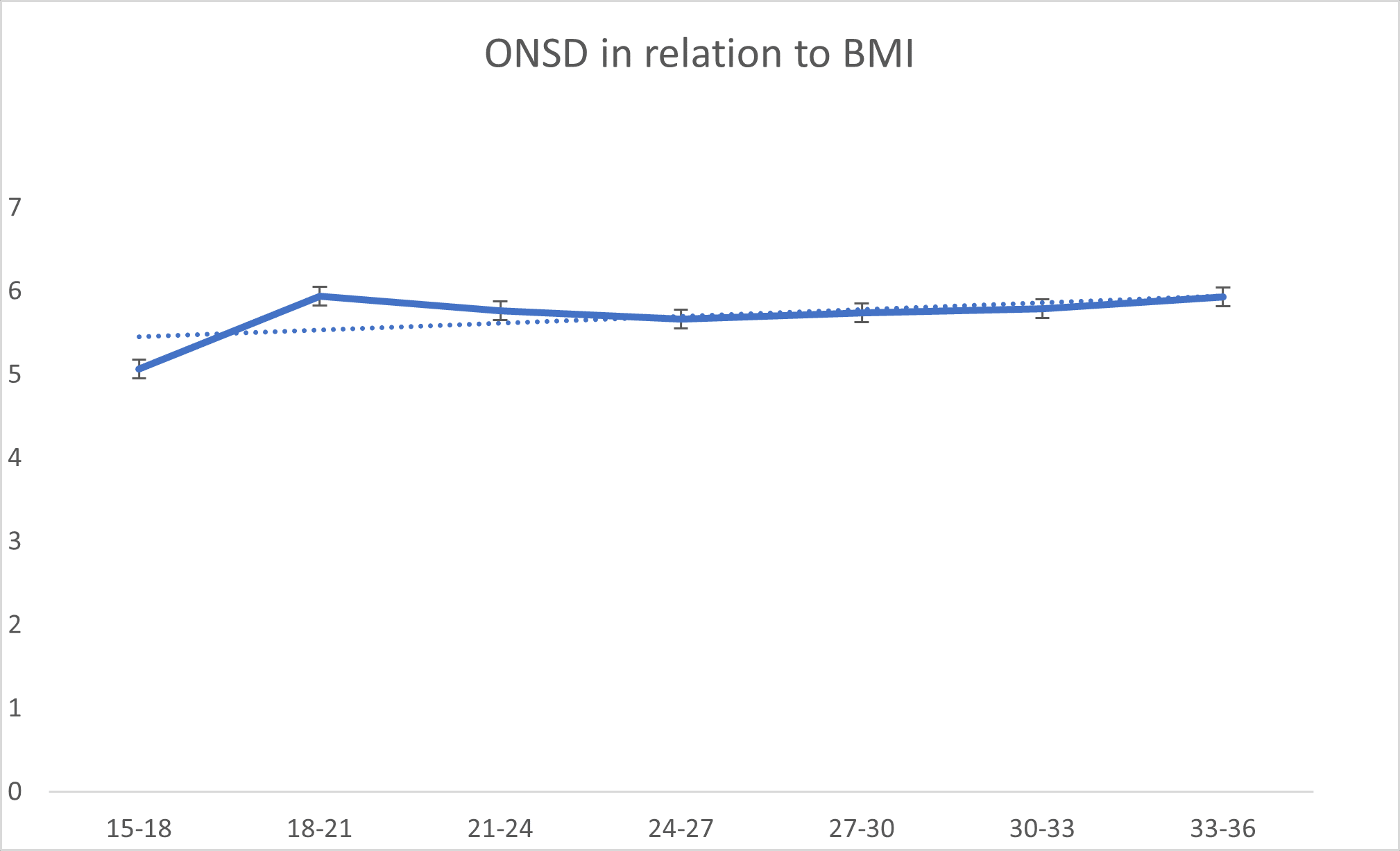
Figure 3: Relationship between ONSD and BMI
| Parameters | Mean ± SD | p-value | |
| Male | Female | ||
| ONSD | 5.8 ± 0.74 | 5.72 ± 0.59 | 0.578 |
| ICP | 24.08 ± 7.5 | 22.32 ± 7.7 | 0.236 |
| BMI | 25.68 ± 3.46 | 25.47 ± 4.20 | 0.781 |
Table 3: Summary of paired sample t-test of the difference between male and female parameters
Table 3 above shows no statistically significant differences between average means of ONSD, BMI, and ICP measurements for males and females.
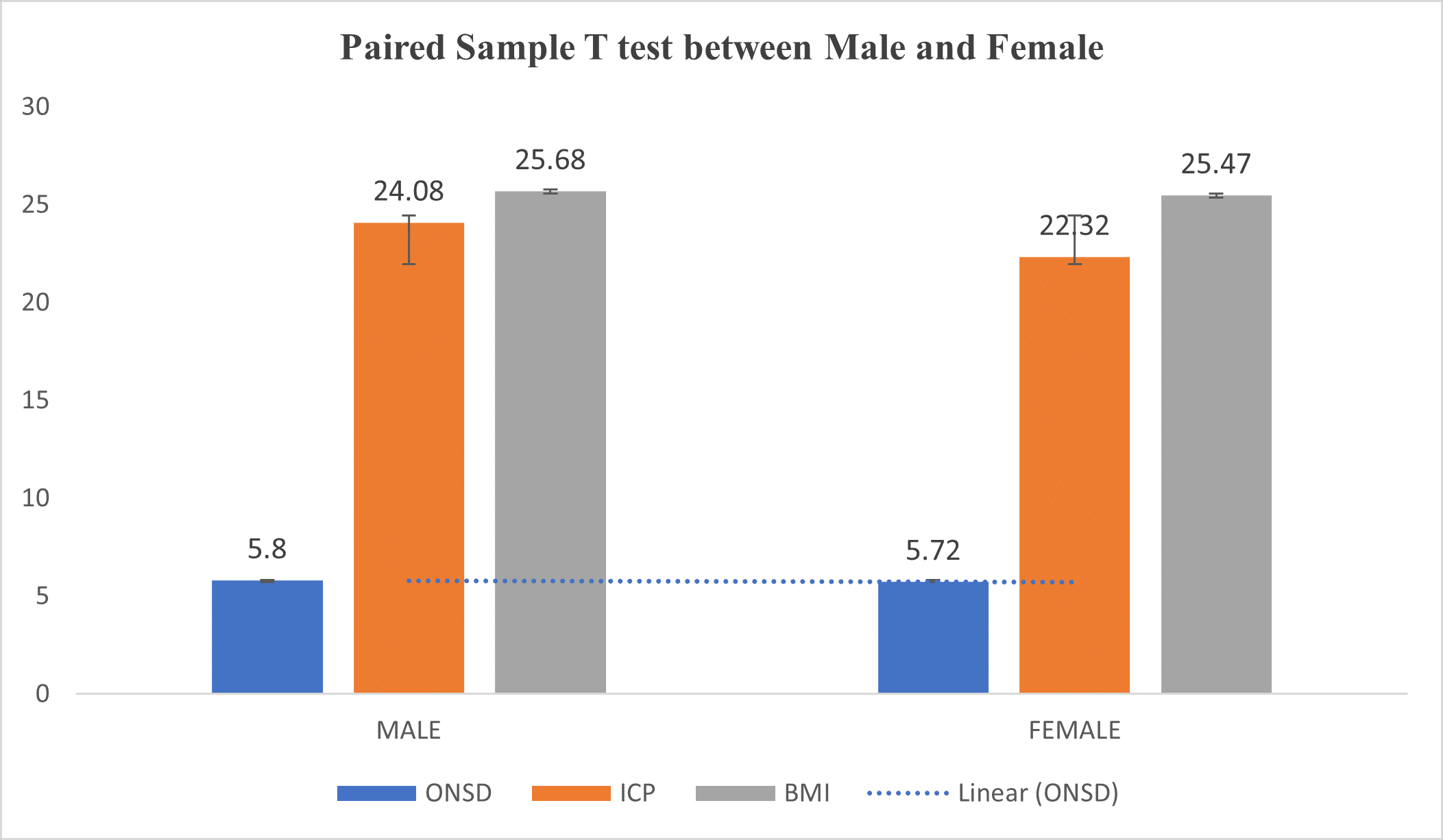
Figure 4: Mean comparison using a paired sample T-test between males and females
Discussion
Average ONSD measurement: Results from this study demonstrated that the average mean ONSD measurements for men and women are 5.8 ± 0.74 and 5.72 ± 0.59, respectively, among 149 stroke patients with a mean age of 57.76 and 51.89 years. Research conducted by Dubourg et al.[8] on two hundred and thirty-one adult patients (older than 18 years) with traumatic brain injury, demonstrated that ONSD is a reliable non-invasive marker for raised ICP. Their study reported a strong correlation between ONSD and ICP, with an r value of 0.74, which aligns closely with the strong correlation (r=0.807) observed in this study. This similarity reinforces the validity of using ONSD as an indicator of ICP. Yic et al.[14] conducted a cross-sectional observational study in which they included fifty-six adult patients, over 18 years of age who required sedation, mechanical ventilation, and invasive ICP monitoring as a result of a severe acute neurologic injury (traumatic or non-traumatic) and had a glasgow coma score (GCS) equal to or less than 8 on admission to the Intensive care unit(ICU). Of this total, 16 (28.6%) were female, with a mean age of 42.1 (range 18–73) years. The mean diameter of the optic nerve for the total number of patients studied was 5.4 mm (range 3.8–7.3) with a standard deviation (SD) of 0.7 mm.
Blaivas et al.[15] performed a prospective, blinded observational study on emergency department (ED) patients with a suspicion of Elevated Intracranial pressure (EICP) due to possible focal intracranial pathology on thirty-five patients were enrolled; 14 had CT results consistent with EICP. The mean ONSD for the 14 patients with CT evidence of EICP was 6.27 mm (95% confidence interval (CI) = 5.6 to 6.89); the mean ONSD for the others was 4.42 mm (95% CI = 4.15 to 4.72). The study suggests that bedside ultrasonography may be useful in the diagnosis of elevated intracranial pressure using ONSD as a marker. It can be postulated, therefore, that there is a strong positive relationship between ONSD and ICP.
Geeraerts et al.[16] prospectively studied the relationship between ONSD and ICP and investigated whether increased ONSD at patient admission is associated with raised ICP in the first 48 hours. He enrolled thirty-one adult patients with severe traumatic brain injury requiring sedation and ICP monitoring, and thirty-one control patients without brain injury requiring sedation. He measured ONSD with a 7.5-MHz linear ultrasound probe in this study. He observed that the largest ONSD value (the highest value for the right and left eye) was significantly higher in high ICP patients with a 6.3mm value. He found out that there was a significant relationship between the largest ONSD and ICP at admission (r=0.68) and concluded that ONSD was a suitable predictor of high ICP (area under the receiver operating characteristic (ROC) curve 0.96).
A study by Wang et al.[17] used an Ultrasound machine to measure ONSD and correlated it with ICP. They recruited three hundred and sixteen neurology patients (mean age, 41.03 ± 15.00 years; range, 18 to 80 years; 174 males; 122 patients with elevated ICP) in the study. ONSD was measured using ultrasonography before lumbar puncture. The study found a strong positive correlation between ONSD measured via Ultrasound and ICP, with a correlation coefficient of r = 0.758. This finding is very similar to the strong correlation found in the current study (r = 0.807), suggesting that both MRI and ultrasonography methods are reliable for assessing ICP through ONSD measurements. The consistency across different imaging modalities strengthens the argument for using ONSD as a non-invasive indicator of ICP.
Chen et al.[11]found that the median ONSD value in healthy adults was 5.1mm, with the 95th percentile at 5.9mm. However, there are notable differences between Chen’s study and ours. Firstly, Chen’s participants were healthy, while our study included individuals with normal or elevated ICP. Secondly, Chen measured ONSD in the horizontal plane and calculated the mean value. In contrast, we measured both sagittal and horizontal sections twice, and the final ONSD value for each patient was the average of two values from both eyes assessed by two observers to minimize variability. Other studies have also examined ONSD in healthy individuals. Goeres et al.[18] reported a mean ONSD of 3.68mm (95% CI = 2.85–4.40) in healthy Canadians. Soldatos et al.[19] found a median ONSD of 3.6 ± 0.6 in healthy Greek participants. Consequently, there is no clear consensus on the median ONSD in healthy adults. Additionally, the reported cut-off value for elevated ICP has varied from 4.6–5 mm, indicating that diagnostic criteria for elevated ICP based on ultrasonography ONSD have not been established.
Although ultrasound measurement of ONSD is increasingly used to detect elevated ICP, there is no consensus on a definitive threshold for this condition. Most studies suggest a cut-off point of 5mm, but this only allows for a qualitative diagnosis of increased ICP without providing quantitative values. Quantitative measurements of ICP seem to be more valuable for assessing disease severity and prognosis. To our knowledge, no previous study has quantified ICP noninvasively using MRI in Stroke patients. Therefore, we developed a mathematical function to achieve this. We calculated an intraclass correlation coefficient (ICC) (95% CIs) of 0.86 (0.79–0.90) for noninvasive ICP assessment using our prediction model, indicating strong agreement between observed and estimated ICP values. Thus, we propose that MRI measurement of ONSD is a promising tool for the quantitative and noninvasive evaluation of ICP in stroke patients.
Gender differences in ONSD and ICP: A study by Geeraerts et al.[9] explored gender differences in ONSD and ICP. They found that while males generally exhibited higher ONSD and ICP values, the differences were not statistically significant, mirroring the current study’s findings. This suggests that gender may not significantly impact the relationship between ONSD and ICP. A study by Liu et al.[20] focused on the factors influencing ONSD measurements using MRI in healthy Chinese adults. The study found that while age and sex did not significantly impact ONSD measurements, there were some variations due to individual anatomical differences. The findings support the current study’s results, showing no significant differences in ONSD between males and females and no correlation with BMI. Consistent findings across studies indicate no significant gender differences in ONSD measurements, supporting the robustness of ONSD as a non-invasive measure irrespective of gender.
BMI and ONSD correlation: A study by Soldatos et al.[19] examined the correlation between BMI and ONSD, finding no significant association. The present study’s results (r = -0.005, p = 0.95) are consistent with these findings, suggesting that BMI does not significantly influence ONSD. A study by Rajajee et al.[21] investigated the relationship between BMI and ICP in neurocritical care patients using ultrasonographic measurements of ONSD. They found no significant association between BMI and ICP, suggesting that BMI does not directly influence intracranial pressure. This is consistent with the current study’s findings, where BMI was not significantly correlated with ONSD, a proxy for ICP. A study by Sekhon et al.[22] investigated the relationship between BMI and ONSD in pediatric populations using MRI. They found no significant correlation between BMI and ONSD, suggesting that BMI does not influence ONSD in children, similar to findings in adults. The current study and previous research demonstrate no significant correlation between BMI and ONSD. This suggests that BMI does not directly impact ONSD measurements, supporting the robustness of ONSD as a measure for ICP independent of BMI. Studies consistently show no significant association between BMI and ICP, indicating that BMI does not directly influence intracranial pressure. These comparisons reinforce the validity of using ONSD as a non-invasive method for monitoring ICP, independent of BMI. Future research should continue to explore these relationships in larger and more diverse populations to confirm these findings and further investigate the potential clinical applications of ONSD measurements for assessing ICP.
Conclusion
In conclusion, this study aimed to evaluate the feasibility and effectiveness of MRI measurements of the ONSD as a non-invasive method for assessing increased ICP in stroke patients. The results indicated a strong positive correlation between ONSD and ICP, reinforcing the reliability of ONSD as a proxy for ICP. There were no significant differences in ONSD measurements across gender or BMI, supporting the robustness of ONSD as a consistent marker irrespective of these variables.
References
- Smith AM, Czyz CN. Neuroanatomy, Cranial Nerve 2 (Optic). StatPearls Publishing; 2022. Neuroanatomy, Cranial Nerve 2
- Gamm DM, Albert DM. Optic nerve. Encyclopedia Britannica Publishing; 2024. Optic nerve
- Mahabadi N, Al Khalili Y. Neuroanatomy, Retina. StatPearls Publishing; 2023. Neuroanatomy, Retina
- François P, Lescanne E, Velut S. The dural sheath of the optic nerve: descriptive anatomy and surgical applications. Adv Tech Stand Neurosurg. 2011;36:187-198. doi:10.1007/978-3-7091-0179-7_7 PubMed | Crossref | Google Scholar
- Tadi P, Lui F. Acute stroke. StatPearls Publishing; 2023. Acute stroke
- Chugh C. Acute ischemic stroke: management approach. Indian J Crit Care Med. 2019;23(Suppl 2):S140-S146. doi:10.5005/jp-journals-10071-23192 PubMed | Crossref | Google Scholar
- Maher M, Schweizer TA, Macdonald RL. Treatment of spontaneous subarachnoid hemorrhage: guidelines and gaps. Stroke. 2020;51(4):1326-1332. doi:10.1161/STROKEAHA.119.025997 PubMed | Crossref | Google Scholar
- Dubourg J, Javouhey E, Geeraerts T, Messerer M, Kassai B. Ultrasonography of optic nerve sheath diameter for detection of raised intracranial pressure: a systematic review and meta-analysis. Intensive Care Med. 2011;37(7):1059-1068. doi:10.1007/s00134-011-2224-2 Crossref | Google Scholar
- Geeraerts T, Merceron S, Benhamou D, Vigué B, Duranteau J. Non-invasive assessment of intracranial pressure using ocular sonography in neurocritical care patients. Intensive Care Med. 2008;34(11):2062-2067. doi:10.1007/s00134-008-1149-x PubMed | Crossref | Google Scholar
- Katan M, Luft A. Global burden of stroke. Semin Neurol. 2018;38(2):208-211. doi:10.1055/s-0038-1649503
PubMed | Crossref | Google Scholar - Chen LM, Wang LJ, Hu Y, Jiang XH, Wang YZ, Xing YQ. Ultrasonic measurement of optic nerve sheath diameter: a non-invasive surrogate approach for dynamic, real-time evaluation of intracranial pressure. Br J Ophthalmol. 2019;103(4):437-441. doi:10.1136/bjophthalmol-2018-312934 PubMed | Crossref | Google Scholar
- Kim DH, Jun JS, Kim R. Measurement of the optic nerve sheath diameter with magnetic resonance imaging and its association with eyeball diameter in healthy adults. J Clin Neurol. 2018;14(3):345-350. doi:10.3988/jcn.2018.14(3):345-350 PubMed | Crossref | Google Scholar
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191-2194. doi:10.1001/jama.2013.281053
PubMed | Crossref | Google Scholar - Yic CD, Pontet J, Mercado M, Muñoz M, Biestro A. Ultrasonographic measurement of the optic nerve sheath diameter to detect intracranial hypertension: an observational study. Ultrasound J. 2023;15(1):4. doi:10.1186/s13089-022-00304-3
PubMed | Crossref | Google Scholar - Blaivas M, Theodoro D, Sierzenski PR. Elevated intracranial pressure detected by bedside emergency ultrasonography of the optic nerve sheath. Acad Emerg Med. 2003;10(4):376-381. doi:10.1111/j.1553-2712.2003.tb01352.x
PubMed | Google Scholar - Geeraerts T, Launey Y, Martin L, et al. Ultrasonography of the optic nerve sheath may be useful for detecting raised intracranial pressure after severe brain injury. Intensive Care Med. 2007;33(10):1704-1711. doi:10.1007/s00134-007-0797-6 PubMed | Crossref | Google Scholar
- Wang LJ, Zhang Y, Li C, et al. Ultrasonographic optic nerve sheath diameter as a noninvasive marker for intracranial hypotension. Ther Adv Neurol Disord. 2022;15:17562864211069744. doi:10.1177/17562864211069744
PubMed | Crossref | Google Scholar - Goeres P, Zeiler FA, Unger B, et al. Ultrasound assessment of optic nerve sheath diameter in healthy volunteers. J Crit Care. 2016;31(1):168-171. doi:10.1016/j.jcrc.2015.10.009 PubMed | Crossref | Google Scholar
- Soldatos T, Karakitsos D, Chatzimichail K, et al. Optic nerve sonography in the diagnostic evaluation of adult brain injury. Crit Care. 2008;12(3):R67. doi:10.1186/cc6897 PubMed | Crossref | Google Scholar
- Liu C, Ji HX, Hu SH, Gong GP, Wei Q, Tian YH. Optic nerve sheath diameter measured using magnetic resonance imaging and factors that influence results in healthy Chinese adults: a cross-sectional study. Chin Med J (Engl). 2021;134(18):2263-2265. doi:10.1097/CM9.0000000000001353 PubMed | Crossref | Google Scholar
- Rajajee V, Vanaman M, Fletcher JJ, Jacobs TL. Optic nerve ultrasound for the detection of raised intracranial pressure. Neurocrit Care. 2011;15(3):506-515. doi:10.1007/s12028-011-9606-8 PubMed | Crossref | Google Scholar
- Sekhon MS, Griesdale DE, Robba C, et al. Optic nerve sheath diameter on computed tomography is correlated with simultaneously measured intracranial pressure in patients with severe traumatic brain injury. Intensive Care Med. 2014;40(9):1267-1274. doi:10.1007/s00134-014-3392-7 PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Information
Corresponding Author:
Aminu Muzammil
Department of Radiology
Afe Babalola Multisystem Hospital, Ekiti State, Nigeria
Email: muxamm99@gmail.com
Co-Authors:
Linus Anderson Enye
Department of Human Anatomy
Afe Babalola University, Nigeria
Nicholas Kayode Irurhe
Department of Radiology
Lagos University Teaching Hospital, Nigeria
Abdullahi Alfa, Oluwaseun Oyelayo
Department of Radiology
Afe Babalola Multisystem Hospital, Nigeria
Ikechukwu Emmanuel Onwunta, Ikenna Caleb Onyemauwa
Department of Radiography and Radiological Science
University of Nigeria, Nigeria
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
This study followed the ethical standards outlined in the 1964 declaration of Helsinki. Ethical approval, identified by the number ABUADHREC/26/06/2024/489, was granted by the ethics and research committee of Afe Babalola University (ABUAD). Additionally, permission and support were obtained from the radiology department’s managing consultants. Informed written consent was obtained from all subjects before the examination, where the reason for the study, benefits, possible effects, and stages of examination were explained to the subjects.
Conflict of Interest Statement
Not applicable
Guarantor
None
DOI
Cite this Article
Muzammil A, Enye LA, Irurhe NK, et al. Measurement of Optic Nerve Sheath Diameter as a Determinant of Increased Intracranial Pressure among Stroke Patients in Southwestern Nigeria. medtigo J Med. 2025;3(1):e3062317. doi:10.63096/medtigo3062317 Crossref









