Author Affiliations
Abstract
Kawasaki disease (KD), primarily affecting children but occasionally adults, is characterized by fever and acute inflammation. It can lead to serious complications like coronary artery aneurysms, heart failure, and arrhythmias, with cardiovascular issues being the primary cause of morbidity and mortality. A 48-year-old man presented with acute chest pain, sweating, and vomiting. Tests showed elevated liver enzymes, erythrocyte sedimentation rate (ESR), and troponin. Electrocardiography (ECG) indicated ST-segment elevation, and echocardiography revealed a dilated left atrium, reduced left ventricular function, and akinesis in multiple segments. Coronary angiography found total occlusion and thrombus in the mid-left anterior descending artery, with ectasia in the right coronary artery and minor ectasia in the left circumflex artery. The patient was diagnosed with ST-segment elevation myocardial infarction due to KD and was treated with dual antiplatelet therapy and statins. This case highlights the importance of diagnosing KD in adults to prevent severe complications.
Keywords
Kawasaki disease, ST elevation myocardial infarction, Coronary angiography, Coronary artery, Antiplatelet therapy.
Introduction
KD, also known as mucocutaneous lymph node syndrome, ranks among the more common forms of childhood vasculitis [1,2]. It primarily affects infants and children under five years of age and manifests as a multi-system inflammatory disorder of the blood vessels. KD can occur alongside bacterial or viral infections is crucial, and it’s significant to highlight that identifying a bacterial or viral infection doesn’t automatically exclude the potential of a KD diagnosis [3]. The disease typically unfolds in two stages: an acute phase lasting one to two weeks, followed by a chronic convalescent phase. If left untreated, KD generally resolves spontaneously within a few weeks.
The hallmark of KD is acute systemic vasculitis. Coronary artery lesions, which often manifest early during the acute phase, can sometimes develop more than four weeks after the onset of the disease [4,5]. Echocardiography, due to its high sensitivity and specificity, remains the preferred diagnostic tool for assessing coronary artery anomalies in KD. The detection of such abnormalities is crucial, particularly in cases where the full clinical criteria are not met [6].
Classic manifestations of KD include fever, rash, oral mucositis, conjunctivitis, lymphadenopathy, and skin desquamation. Diagnosis of classical KD hinges on the presence of a fever lasting at least five days and at least four of the five primary clinical features. In instances where at least four primary criteria are met, particularly when accompanied by redness and swelling of the hands and feet, a diagnosis may be confirmed with only four days of fever [7,8,9]. KD can lead to numerous complications across various body systems, with cardiac issues such as coronary artery aneurysms, heart failure, myocardial infarction, and arrhythmias being particularly severe and contributing significantly to morbidity and mortality [10]. The recurrence rate for KD is remarkably low, 1.4 to 3%, and secondary cases, though rare, typically occur among siblings [11]. In this report, we present the case of a black man who experienced an acute myocardial infarction secondary to KD.
Case Presentation
A 48-year-old Ethiopian man presented with chest pain characterized as a squeezing sensation, radiating to his left arm and shoulder, accompanied by sweating and vomiting, persisting for one day. Four days before this, he had epigastric discomfort and sought medical care at a nearby hospital,l where he received intravenous omeprazole, leading to symptom relief. He denied experiencing shortness of breath, paroxysmal nocturnal dyspnea, or orthopnea. The patient had no history of medication use, tobacco, or recreational drug consumption, and there was no family history of myocardial infarction or sudden cardiac death; both parents are alive.
His physical examination yielded unremarkable findings. ECG displayed a normal sinus rhythm with ST-segment elevation in the anteroseptal and anterolateral leads. The echocardiography report revealed a dilated left atrium and reduced global systolic function of the left ventricle with an estimated ejection fraction of 30-35%. There was akinesis noted in the basal and mid anterior, anterolateral, and anteroseptal walls, as well as in the apico-anterior and lateral walls. The right ventricular systolic function was preserved with a tricuspid annular plane systolic excursion (TAPSE) of 24mm. No intracardiac thrombus, mass, or vegetation was observed, and both the intra-atrial and intraventricular septa appeared intact.
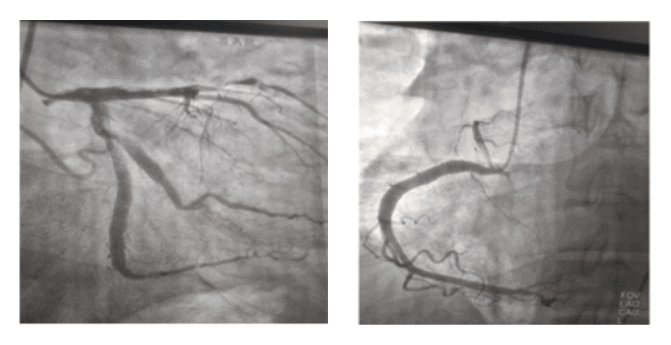
Coronary angiography identified total occlusion with a large thrombus and ectatic changes in the mid-left anterior descending coronary artery, predominant ectasia in the right coronary artery, and minor ectasia in the left circumflex coronary artery.
Initial blood tests revealed a white blood cell count of 9.4k per microliter (with 65.3% neutrophils and 26.5% lymphocytes), a hemoglobin level of 13g/dL (with a hematocrit of 44%), and a platelet count of 232k per microliter. Additionally, the results indicated creatinine at 0.7mg/dL, urea at 29mg/dL, sodium at 127mmol/L, potassium at 3.59mmol/L, and chloride at 101mmol/L. Liver enzyme levels were elevated, with alanine transaminase at 71 units per liter (twice the normal value) and aspartate transaminase at 168 units per liter (four times the normal value). High-sensitivity troponin was significantly elevated, measuring 98 times the normal level. High-density lipoprotein cholesterol and low-density lipoprotein cholesterol were recorded as 72 and 52 mg/dL, respectively.
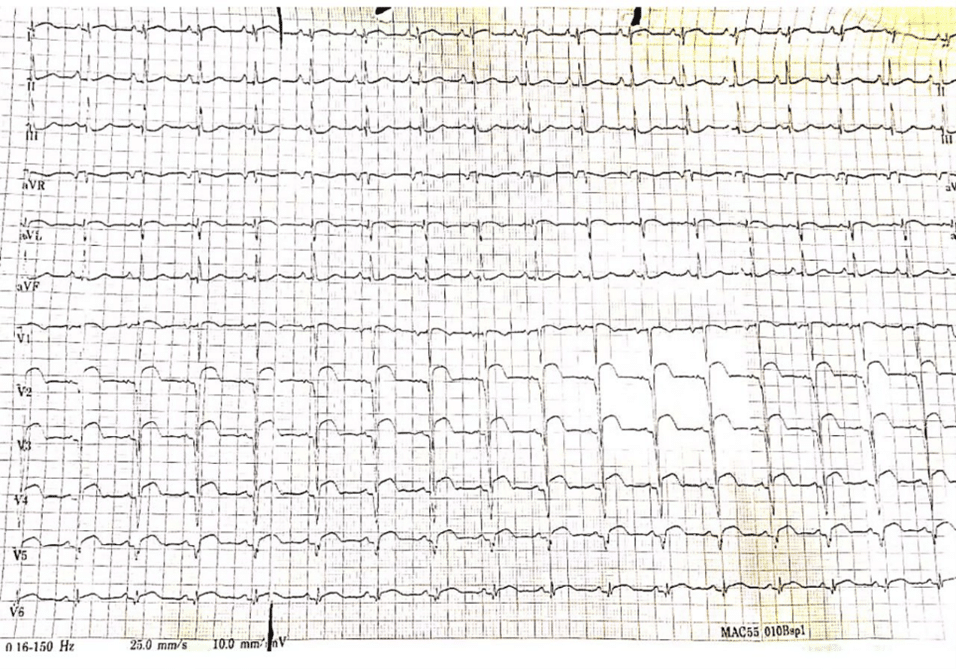
Figure 1: ECG displayed a normal sinus rhythm with ST-segment elevation in the anterio-septal leads and anterio-lateral leads
 A B
A B
Figure 2: A and B total occlusion with a large thrombus and ectatic changes in the mid left anterior descending coronary artery, predominant ectasia in the right coronary artery, and minor ectasia in the left circumflex coronary artery.
Case Management
After the procedure, the patient started a regimen of dual antiplatelet therapy consisting of 81 mg of aspirin and 75 mg of clopidogrel daily, along with an 80 mg dose of atorvastatin statin. Due to a high level of thrombus presence, intravenous heparin was administered for 48 hours post-coronary angiography, followed by oral intake of rivaroxaban at 15 mg twice daily for 21 days, subsequently transitioning to 20 mg once daily. Additionally, considering the diminished left ventricular systolic function, the patient was prescribed guideline-directed medications including metoprolol succinate, enalapril, spironolactone, and dapagliflozin. Regular outpatient follow-up has been maintained without any adverse events over the past four months while adhering to aspirin, rivaroxaban, statin, and guideline-directed medication therapy.
Discussion
Adult-onset KD is rare and often misdiagnosed or identified late due to disease progression, lack of a specific diagnostic test, and its clinical similarity to other more common infectious diseases [12]. The prevalence of KD varies significantly by region. Epidemiological studies from Hawaii and Korea indicate a higher incidence among children of Asian descent, with Korea reporting the second-highest global rates after Japan [13,14,15]. A national survey in Korea from 2015 to 2017 showed an incidence of 196.9 cases per 100,000 children under 5 years old, with most diagnoses occurring in this age group [16]. In contrast, regions such as North America, Europe, and Australia report KD incidence rates ranging from 5 to 22 cases per 100,000 children under 5 years old. While these rates have increased due to better diagnosis, they have plateaued over the last decade [17]. Japan has the highest global incidence, estimated at 264 cases per 100,000 children under 5. KD recurrence is about 3.5%, mortality rates are below 0.02%, and around 17% of cases are resistant to intravenous immunoglobulin (IVIG) treatment [18]. There is limited data on the incidence of KD in sub-Saharan Africa, but isolated cases have been reported in multiple countries across the region [19].
A comprehensive study in Asia found that the annual risk of coronary complications was 2.4% between 2000 and 2010, with acute myocardial infarction occurring in 1.52% of cases [20]. KD is characterized by systemic inflammation affecting medium-sized arteries and multiple organs and tissues, especially during the acute febrile phase. This inflammation often involves the coronary arteries, leading to coronary artery aneurysms in about 25% of untreated patients [21]. The associated vasculopathy includes acute self-limiting necrotizing arteritis, saccular aneurysms, subacute and chronic vasculitis, and luminal myofibroblastic proliferation, which can cause progressive arterial stenosis and thrombosis [8]. These events are accompanied by significant immune activation, elevated levels of circulating cytokines, CD4 and CD8 cells, polyclonal hypergammaglobulinemia, and immune complexes [22].
Symptoms of KD include fever, rash, oral mucositis, conjunctivitis, lymphadenopathy, and skin peeling. Common laboratory findings include leukocytosis, thrombocytosis, elevated ESR, positive C-reactive protein (CRP), and slightly elevated serum transaminase levels. Our patient has an elevated ESR, elevations in serum transaminases, and raised troponin. Antinuclear antibodies and rheumatoid factors are typically absent. Mild to moderate increases in serum transaminases or gamma-glutamyl transpeptidase occur in 40% to 60% of patients, with mild hyperbilirubinemia in about 10% of cases [23]. Hypoalbuminemia is common and linked to more severe and prolonged acute disease [24]. The primary complication of KD is the development of coronary artery aneurysms (CAAs), which can lead to myocardial ischemia, myocardial infarction, and sudden death. These aneurysms can cause thrombosis and progressive stenosis, potentially leading to late ischemic heart disease [25]. KD that goes undiagnosed in childhood can present as myocardial infarction in adulthood. Our patient presented with acute myocardial infarction with ST-segment elevation in the anteroseptal leads and anterolateral leads.
Echocardiography is the primary method for assessing coronary artery abnormalities in KD due to its high sensitivity and specificity [4,5]. It is crucial to conduct the initial echocardiogram promptly upon suspicion of KD, though treatment should not be delayed for this evaluation [26]. Multiple imaging planes and transducer positions are necessary for optimal visualization. Common sites for coronary artery aneurysms include the proximal left anterior descending artery (LAD), proximal Right coronary artery (RCA), left main coronary artery (LMCA), left circumflex, distal RCA, and rarely, the junction between the RCA and posterior descending coronary artery [4]. Coronary artery abnormalities are a key diagnostic indicator for KD, particularly in patients who do not meet all clinical criteria. These abnormalities differ from the minor dilation seen in other febrile illnesses [6]. During the acute phase, coronary artery abnormalities can range from dilation to aneurysms, typically starting in the proximal segments before extending distally. Distal involvement without proximal abnormalities is rare [27]. Our patient has total occlusion with a large thrombus and ectatic changes in the mid-left anterior descending coronary artery, with predominant ectasia in the right coronary artery.
The diagnosis of classic KD relies on the presence of fever lasting ≥5 days and at least four of the five primary clinical features. In cases where four primary clinical criteria are met, especially with redness and swelling of the hands and feet, the diagnosis can be made with just four days of fever [28,29,32]. Incomplete (atypical) KD poses diagnostic challenges, as the criteria for complete KD may not capture all cases. Prolonged unexplained fever in childhood, along with primary clinical features, should prompt consideration of KD. Definitive confirmation often requires echocardiography revealing coronary artery aneurysms, although such dilation may not be detectable until after the first week of illness. Laboratory findings and cardiovascular complications in incomplete and complete KD cases are similar, with certain laboratory features aiding in diagnosis. Notably, coronary artery Z scores of ≥2.5 are highly specific for diagnosis but lack sensitivity [6,30].
The main goal of treating KD is to manage acute inflammation and prevent serious cardiovascular complications, particularly coronary artery issues [29]. Treatment typically involves administering IVIG and high-dose aspirin, with IVIG effective in reducing the progression to CAA. Once CAA develops, the focus shifts to preventing coronary thrombosis. For small CAAs (2.5 ≤ Z score < 5), a regimen of low-dose aspirin is recommended, while those with giant aneurysms (Z score > 10) are advised to take a combination of aspirin and warfarin [32]. Coronary revascularization is suggested for patients with giant or multiple coronary artery aneurysms or significant stenosis [30]. Screening echocardiography is used to assess the type and severity of coronary artery lesions and establish a treatment plan [27]. Regular echocardiographic monitoring is necessary if an aneurysm is detected, with coronary angiography typically reserved for confirmed cases of coronary artery aneurysms.
Conclusion
Coronary artery lesions associated with KD are a critical concern because they have a significant impact on morbidity and mortality. The presented case of a 48-year-old male directly linked the diagnosis of ST-segment elevation myocardial infarction to KD, highlighting the enduring risks associated with the condition. This case highlights the essential role of early and accurate diagnosis, coupled with vigilant monitoring and tailored medical management. Treatments such as intravenous immunoglobulins combined with aspirin have been shown to be effective in reducing the progression to severe coronary artery abnormalities, thereby mitigating some of the disease’s most dire consequences. Continued research and clinical awareness are imperative to improve outcomes for patients with both typical and atypical presentations of KD.
References
- Agarwal S, Agrawal DK. Kawasaki disease: Etiopathogenesis and novel treatment strategies. Expert Rev Clin Immunol. 2017;13(3):247-258. doi:10.1080/1744666X.2017.1232165 PubMed | Crossref | Google Scholar
- Soni PR, Rivas MN, Arditi M. A comprehensive update on Kawasaki disease vasculitis and myocarditis. Curr Rheumatol Rep. 2020;22(6):6. doi:10.1007/s11926-020-0882-1 PubMed | Crossref | Google Scholar
- Huang SM, Huang SH, Weng KP, et al. Update on association between Kawasaki disease and infection. J Chin Med Assoc. 2019;82(3):172-174. doi:10.1097/JCMA.0000000000000025 PubMed | Crossref | Google Scholar
- Fuse S, Kobayashi T, Arakaki Y, et al. Standard method for ultrasound imaging of coronary artery in children. Pediatr Int. 2010;52(6):876-882. doi:10.1111/j.1442-200X.2010.03252.x PubMed | Crossref | Google Scholar
- Brown LM, Duffy CE, Mitchell C, Young L. A practical guide to pediatric coronary artery imaging with echocardiography. J Am Soc Echocardiogr. 2015;28(4):379-391. doi:10.1016/j.echo.2015.01.008 PubMed | Crossref | Google Scholar
- Muniz JC, Dummer K, Gauvreau K, et al. Coronary artery dimensions in febrile children without Kawasaki disease. Circulation Cardiovasc Imaging. 2013;6(2):239-244. doi:10.1161/CIRCIMAGING.112.000159
PubMed | Crossref | Google Scholar - Kanegaye JT, Van Cott E, Tremoulet AH, et al. Lymph-node-first presentation of Kawasaki disease compared with bacterial cervical adenitis and typical Kawasaki disease. J Pediatr. 2013;162(6):1259-1263.e2. doi:10.1016/j.jpeds.2012.11.064 PubMed | Crossref | Google Scholar
- Dominguez SR, Anderson MS, El-Adawy M, Glodé MP. Preventing coronary artery abnormalities: A need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012;31(12):1217-1220. doi:10.1097/INF.0b013e318266bcf9 PubMed | Crossref | Google Scholar
- Kato H, Kanematsu M, Kato Z, et al. Computed tomographic findings of Kawasaki disease with cervical lymphadenopathy. J Comput Assist Tomogr. 2012;36(1):138-142. doi:10.1097/RCT.0b013e31823b4497 PubMed | Crossref | Google Scholar
- Fukazawa R, Kobayashi J, Ayusawa M, et al. JCS/JSCS 2020 guideline on diagnosis and management of cardiovascular sequelae in Kawasaki disease. Circ J. 2020;84(8):1348-1407. doi:10.1253/circj.CJ-19-1094 PubMed | Crossref | Google Scholar
- Yang HM, Du ZD, Fu PP. Clinical features of recurrent Kawasaki disease and its risk factors. Eur J Pediatr. 2013;172(12):1641-1647. doi:10.1007/s00431-013-2101-9 PubMed | Crossref | Google Scholar
- Fraison JB, Seve P, Dauphin C, et al. Kawasaki disease in adults: Observations in France and literature review. Autoimmun Rev. 2016;15(3):242-249. doi:10.1016/j.autrev.2015.11.010 PubMed | Crossref | Google Scholar
- Holman RC, Christensen KY, Belay ED, et al. Racial/ethnic differences in the incidence of Kawasaki syndrome among children in Hawaii. Hawaii Med J. 2010;69(7):194-197. Racial/ethnic differences in the incidence of Kawasaki syndrome among children in Hawaii PubMed | Google Scholar
- Uehara R, Belay ED. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J Epidemiol. 2012;22(2):79-85. doi:10.2188/jea.JE20110131 PubMed | Crossref | Google Scholar
- Makino N, Nakamura Y, Yashiro M, et al. Nationwide epidemiologic survey of Kawasaki disease in Japan, 2015-2016. Pediatr Int. 2019;61(4):397-403. doi:10.1111/ped.13809 PubMed | Crossref | Google Scholar
- Kim GB, Eun LY, Han JW, et al. Epidemiology of Kawasaki disease in South Korea: A nationwide survey 2015-2017. Pediatr Infect Dis J. 2020;39(10):1012-1016. doi:10.1097/INF.0000000000002793 PubMed | Crossref | Google Scholar
- Lin MT, Wu MH. The global epidemiology of Kawasaki disease: Review and future perspectives. Glob Cardiol Sci Pract. 2017;2017(3):e201720. doi:10.21542/gcsp.2017.20 PubMed | Crossref | Google Scholar
- Huang WC, Huang LM, Chang IS, et al. Epidemiologic features of Kawasaki disease in Taiwan, 2003-2006. 2009;123(3):e401-e405. doi:10.1542/peds.2008-2187 PubMed | Crossref | Google Scholar
- Badoe EV, Neequaye J, Oliver-Commey JO, et al. Kawasaki disease in Ghana: Case reports from Korle Bu teaching hospital. Ghana Med J. 2011;45(1):38-42. doi:10.4314/gmj.v45i1.68922 PubMed | Crossref | Google Scholar
- Wu MH, Chen HC, Yeh SJ, et al. Prevalence and the long-term coronary risks of patients with Kawasaki disease in a general population <40 years: A national database study. Circ Cardiovasc Qual Outcomes. 2012;5(5):566-570. doi:10.1161/CIRCOUTCOMES.112.965194 PubMed | Crossref | Google Scholar
- Kato H, Sugimura T, Akagi T, et al. Long-term consequences of Kawasaki disease: A 10- to 21-year follow-up study of 594 patients. Circulation. 1996;94(6):1379-1385. doi:10.1161/01.CIR.94.6.1379 PubMed | Crossref | Google Scholar
- Butler DF, Hough DR, Friedman SJ, Davis HE. Adult Kawasaki syndrome. Arch Dermatol. 1987;123(11):1356-1361. doi:10.1001/archderm.1987.01660340118032 PubMed | Crossref | Google Scholar
- Tremoulet AH, Jain S, Chandrasekar D, et al. Evolution of laboratory values in patients with Kawasaki disease. Pediatr Infect Dis J. 2011;30(12):1022-1026. doi:10.1097/INF.0b013e31822d4f56 PubMed | Crossref | Google Scholar
- Eladawy M, Dominguez SR, Anderson MS, Glodé MP. Abnormal liver panel in acute Kawasaki disease. Pediatr Infect Dis J. 2011;30(2):141-144. doi:10.1097/INF.0b013e3181f6fe2a PubMed | Crossref | Google Scholar
- Kato H, Inoue O, Kawasaki T, et al. Adult coronary artery disease probably due to childhood Kawasaki disease. Lancet. 1992;340(8814):1127-1129. doi:10.1016/0140-6736(92)93152-D PubMed | Crossref | Google Scholar
- Capannari TE, Daniels SR, Meyer RA, Schwartz DC, Kaplan S. Sensitivity, specificity and predictive value of two-dimensional echocardiography in detecting coronary artery aneurysms in patients with Kawasaki disease. J Am Coll Cardiol. 1986;7(2):355-360. doi:10.1016/S0735-1097(86)80505-8 PubMed | Crossref | Google Scholar
- Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110(17):2747-2771. doi:10.1161/01.CIR.0000145143.19711.78 PubMed | Crossref | Google Scholar
- Ohno S, Miyajima T, Higuchi M, et al. Ocular manifestations of Kawasaki’s disease (mucocutaneous lymph node syndrome). Am J Ophthalmol. 1982;93(6):713-717. doi:10.1016/0002-9394(82)90465-2
PubMed | Crossref | Google Scholar - Kanegaye JT, Van Cott E, Tremoulet AH, et al. Lymph-node-first presentation of Kawasaki disease compared with bacterial cervical adenitis and typical Kawasaki disease. J Pediatr. 2013;162(6):1259-1263.e2. doi:10.1016/j.jpeds.2012.11.064 PubMed | Crossref | Google Scholar
- Bratincsak A, Reddy VD, Purohit PJ, et al. Coronary artery dilation in acute Kawasaki disease and acute illnesses associated with fever. Pediatr Infect Dis J. 2012;31(9):924-926. doi:10.1097/INF.0b013e31826252b3 PubMed | Crossref | Google Scholar
- Negoro N, Nariyama J, Nakagawa A, et al. Successful catheter interventional therapy for acute coronary syndrome secondary to Kawasaki disease in young adults. Circ J. 2003;67(4):362-365. doi:10.1253/circj.67.362 PubMed | Crossref | Google Scholar
- McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A scientific statement for health professionals from the American Heart Association. Circulation. 2017;135(17):e927-e999. doi:10.1161/CIR.0000000000000484 PubMed | Crossref | Google Scholar
Acknowledgments
None to declare
Funding
None to declare
Author Information
Corresponding Author:
Yadelew Jember Kassie
Department of Internal Medicine
Saint Paul Hospital Millennium Medical College, Addis Ababa, Ethiopia
Email: yadelewj@gmail.com
Co-Authors:
Zekariyas Mulugeta
Department of Internal Medicine
Saint Paul Hospital Millennium Medical College, Addis Ababa, Ethiopia
Alebachew Girum, Seifu Bacha, Henok Banti, Yonatan Debebe
Cardiology Unit, Department of Internal Medicine
Saint Paul Hospital Millennium Medical College, Addis Ababa, Ethiopia
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Informed Consent
Informed consent for publication of this report was obtained from the patient.
Conflict of Interest Statement
The authors declare that they have no conflict of interest.
Guarantor
Not reported
DOI
Cite this Article
Yadelew Jember K, Zekariyas M, Alebachew G, Seifu B, Henok B, Yonatan D. Literature Review and Case Report on ST-Segment Elevation Myocardial Infarction in Kawasaki Disease. medtigo J Med. 2024;2(3):e3062237.
doi: 10.63096/medtigo3062237 Crossref









