Author Affiliations
Abstract
Background: Post-traumatic epilepsy (PTE) is a major long-term complication of traumatic brain injury (TBI), contributing substantially to global neurological morbidity. Reported incidence varies widely, and the relative contribution of clinical and radiological predictors remains uncertain.
Aim: To estimate the pooled incidence of PTE following TBI and identify its major predictors.
Methodology: A systematic review and meta-analysis were conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. PubMed, Scopus, Web of Science, and Embase were searched from inception to December 2024. Twenty-two studies involving over 170,000 participants met the inclusion criteria. Data extraction and risk-of-bias assessment using the Newcastle–Ottawa Scale were performed independently by two reviewers. A random-effects model was used to pool incidence estimates and risk ratios (RRs) for key predictors.
Results: The pooled global incidence of PTE following TBI was 12.4% (95% confidence interval (CI) 9.8-15.6). Severe TBI significantly increased risk (risk ratio (RR) 4.18, 95% CI 2.90-6.02). Other strong predictors included subdural hemorrhage (RR 2.82), intracerebral hemorrhage (RR 1.95), depressed skull fracture (RR 2.67), and neurosurgical intervention (RR 1.74). Early post-traumatic seizures were the strongest predictor (RR 3.42, 95% CI: 2.56-4.58). Incidence was higher in penetrating injuries and studies with longer follow-up durations.
Conclusion: Approximately one in eight individuals with TBI develops PTE. Severe injury, hemorrhagic lesions, skull fractures, neurosurgical intervention, and early seizures markedly increased the risk. These findings support targeted surveillance and risk-stratified follow-up, particularly in high-risk and resource-limited settings.
Keywords
Post-traumatic epilepsy, Traumatic brain injury, Early post-traumatic seizures, Intracranial hemorrhage, Skull fracture, Neurosurgical intervention.
Introduction
TBI is a major global public health concern and a leading cause of long-term neurological disability. Among its most serious complications is post-traumatic epilepsy (PTE), defined as unprovoked recurrent seizures occurring more than seven days after injury.[1] PTE accounts for approximately 20% of symptomatic epilepsies in the general population and up to 50% in certain high-risk groups.[2] The risk of developing PTE varies widely depending on injury severity, early post-traumatic seizures (EPTS), intracranial hemorrhage, neurosurgical intervention, and demographic characteristics.[3,4]
Despite decades of research, there remains significant heterogeneity in reported incidence rates and inconsistencies in identifying independent predictors. This variation is partly due to differences in study design, follow-up duration, TBI classification, and diagnostic criteria. As a result, clinicians face uncertainty when counselling patients, determining follow-up duration, or considering prophylactic treatment strategies.
Global burden of TBI and post-traumatic epilepsy:
Globally, an estimated 69 million individuals sustain a TBI each year, with the highest rates occurring in low- and middle-income countries due to road traffic accidents, violence, and occupational injuries.[5] The risk of PTE is highest in severe TBI, where incidence may exceed 20%.[6] PTE is associated with substantial morbidity, including cognitive impairment, decreased quality of life, psychiatric comorbidity, and high socioeconomic costs.[7] The burden is disproportionately higher in regions with limited access to neurosurgical and rehabilitation services. Given the rising incidence of TBI worldwide and the lifelong consequences of epilepsy, understanding the true epidemiology and modifiable predictors of PTE is a global health priority.
Rationale for the study:
Although numerous studies have reported incidence rates and potential predictors of PTE, findings remain inconsistent and fragmented. Some predictors, such as EPTS and intracranial hemorrhage, are well-established, while others, including age, mechanism of injury, and imaging biomarkers, remain controversial.[3,8] A comprehensive systematic review and meta-analysis integrating global evidence is needed to provide pooled estimates, reduce uncertainty, and guide clinical decision-making. Such evidence will support early risk stratification, inform surveillance protocols, and identify research gaps for future prospective studies.
Research questions:
- What is the pooled global incidence of post-traumatic epilepsy following traumatic brain injury?
- What clinical, radiological, and demographic factors predict the development of PTE?
- How does the incidence of PTE vary by TBI severity (mild, moderate, severe)?
Aim of the study:
To determine the global incidence and key predictors of post-traumatic epilepsy following traumatic brain injury through a systematic review and meta-analysis.
Objectives:
- To estimate the pooled incidence of PTE across different severities of TBI.
- To identify and quantify clinical and radiological predictors associated with the development of PTE.
- To explore heterogeneity in PTE incidence based on study design, population characteristics, and follow-up duration.
Literature review:
Conceptual overview of post-traumatic epilepsy: PTE is a form of acquired epilepsy that develops as a delayed consequence of TBI. It is classically distinguished from early EPTS, which occur within the first 7 days after injury, and late post-traumatic seizures, which occur thereafter and signify established epileptogenesis. Recurrent late unprovoked seizures constitute PTE according to the International League Against Epilepsy (ILAE) definitions.[1]
PTE represents a unique human model of epileptogenesis because the inciting insult is often well defined temporally and mechanistically. Unlike idiopathic epilepsy, PTE arises from identifiable structural and biochemical changes within the brain, making it a critical focus of both clinical neurology and translational neuroscience.[2] The latency period between injury and seizure onset can range from months to decades, reflecting the gradual evolution of epileptogenic networks following brain trauma.[3]
Epidemiology of post-traumatic epilepsy: The reported incidence of PTE varies widely across studies, ranging from less than 2% after mild TBI to over 40% following penetrating head injuries.[4-6] Population-based studies from high-income countries suggest an overall incidence between 4% and 17%, depending on injury severity and duration of follow-up.[4,7] Annegers et al.[4] demonstrated a stepwise increase in epilepsy risk with increasing TBI severity, a finding consistently replicated in subsequent cohorts.
Large administrative database studies, such as the Taiwanese national cohort by Yeh et al.[3] have further shown that even mild TBI is associated with a significantly increased long-term risk of epilepsy compared with uninjured controls.[7] Importantly, these risks persist for many years post-injury, highlighting the chronic nature of PTE.
In contrast, studies conducted in low- and middle-income countries (LMICs) often report higher incidence rates, likely reflecting greater injury severity, delayed access to care, underutilisation of preventive strategies, and prolonged exposure to secondary brain insults.[8] However, data from these regions remain limited, fragmented, and under-represented in global syntheses.
TBI severity and risk of PTE: TBI severity is the most consistently reported determinant of PTE risk. Severity is typically classified using the Glasgow Coma Scale (GCS), duration of loss of consciousness, post-traumatic amnesia, and neuroimaging findings. Mild TBI is associated with relatively low PTE incidence (1–5%), though risk remains higher than in the general population.[7,9] Moderate TBI confers intermediate risk, with reported incidence ranging from 5% to 15%.[4,6] Severe TBI carries the highest risk, with long-term PTE incidence frequently exceeding 20%.[4,6]
Penetrating head injuries, particularly military or ballistic trauma, represent the extreme end of severity. Longitudinal cohorts such as the Vietnam Head Injury Study report epilepsy rates exceeding 35% even decades after injury.[3,5,10] These findings support a clear dose–response relationship between injury severity and epileptogenesis.
Early post-traumatic seizures as a predictor: EPTS are among the strongest predictors of subsequent PTE. Multiple prospective cohort studies demonstrate that individuals who experience seizures within the first week after TBI have a two- to five-fold increased risk of developing epilepsy later.[2,6,11] Englander et al.[6] In a multicentre prospective study, EPTS was identified as the single most powerful independent predictor of late seizures after adjustment for injury severity and imaging findings. The biological rationale is that EPTS may reflect severe cortical irritation, blood–brain barrier breakdown, or early network instability, all of which contribute to epileptogenesis.[12]
However, not all patients with EPTS develop PTE, and prophylactic antiepileptic drug (AED) use has been shown to reduce EPTS without preventing late epilepsy.[2] This distinction underscores that EPTS are a marker of risk rather than a direct cause of PTE.
Neuroimaging findings and structural predictors:
Intracranial hemorrhage: Intracranial hemorrhage (ICH) is a well-established predictor of PTE. Subdural, intracerebral, and subarachnoid hemorrhages have all been associated with increased epilepsy risk.[6,9,13] Subdural hemorrhage is frequently reported as conferring the highest risk, possibly due to associated cortical contusions and prolonged inflammatory responses. Blood degradation products such as haemoglobin and iron are known to induce oxidative stress and neuroinflammation, creating a pro-epileptogenic environment.[14]
Depressed skull fractures: Depressed skull fractures are strongly associated with PTE, particularly when bone fragments breach the dura and directly injure the cortex. Population-based studies have shown a two- to three-fold increase in epilepsy risk among patients with depressed fractures compared with those without skull injury.[15]
Advanced neuroimaging: Recent studies using diffusion tensor imaging (DTI) and functional MRI have demonstrated that microstructural white-matter disruption and altered network connectivity are associated with later PTE development.[16,17] These findings suggest that epileptogenesis may occur even in the absence of large focal lesions detectable on conventional imaging.
Neurosurgical intervention and PTE risk: Neurosurgical procedures such as decompressive craniectomy, evacuation of haematomas, and contusion resection are markers of severe injury but may also independently contribute to epilepsy risk. Studies report higher PTE incidence in surgically managed patients compared with conservatively treated counterparts.[6,10] The mechanisms underlying this association are multifactorial and include additional cortical injury, scarring, and inflammatory responses related to surgical manipulation. However, disentangling the effects of surgery from injury severity remains challenging in observational studies.
Demographic and behavioural factors: Age and substance use have been variably associated with PTE risk. Older age has been linked to modestly increased risk, potentially due to reduced neuroplasticity and higher comorbidity burden.[7,9] Alcohol misuse is a recognised risk factor for both TBI occurrence and post-injury seizures, with several studies demonstrating an independent association with PTE.[18] Sex differences are less consistent, with most large cohorts showing no strong independent effect after adjustment for injury characteristics.[19]
Pathophysiological mechanisms of post-traumatic epileptogenesis: Experimental and human studies suggest that post-traumatic epilepsy arises from a convergence of interconnected pathological processes following traumatic brain injury. Acute neuronal injury and excitotoxicity contribute to immediate network instability, setting the stage for abnormal electrical activity. This is followed by sustained neuroinflammation, characterised by microglial activation and pro-inflammatory cytokine release that may persist long after the initial insult.[12,14]
Structural reorganisation, including mossy fibre sprouting and maladaptive synaptic rewiring, further promotes the formation of recurrent excitatory circuits. In parallel, disruption of the normal inhibitory–excitatory balance occurs through loss or dysfunction of GABAergic interneurons, reducing seizure threshold.[20-22] Together, these mechanisms provide strong biological plausibility for the clinical and radiological predictors of post-traumatic epilepsy identified in epidemiological studies.
Gaps in the existing literature: Despite extensive research on PTE, several important gaps persist in the existing literature. Long-term prospective data from low- and middle-income countries remain limited, restricting understanding of PTE risk in regions where traumatic brain injury burden is highest.[5,6,9] There is also considerable inconsistency in the definition and ascertainment of PTE across studies, with variation in diagnostic criteria, follow-up duration, and reliance on clinical versus administrative data.[23,24] Mild traumatic brain injury and paediatric populations are under-represented in many cohorts, potentially leading to underestimation of lifetime epilepsy risk in these groups.
In addition, few studies integrate advanced neuroimaging, genetic susceptibility, or biomarker data to elucidate mechanisms of epileptogenesis or refine risk prediction.[25] Finally, there is a scarcity of validated predictive models that can be readily applied in routine clinical practice to guide surveillance and preventive strategies. Collectively, these limitations underscore the need for updated and comprehensive meta-analytic synthesis to consolidate global evidence and inform clinically meaningful risk stratification.
Summary of literature review: The existing literature demonstrates that PTE is a common and disabling consequence of TBI, with incidence and risk strongly influenced by injury severity, early seizures, haemorrhagic lesions, skull fractures, and neurosurgical intervention. While substantial progress has been made in identifying predictors, heterogeneity across studies continues to limit clinical translation. A systematic review and meta-analysis synthesising global evidence is therefore essential to refine incidence estimates, clarify predictor strength, and inform risk-stratified care pathways.
Methodology
Study design and reporting standards: This study was conducted as a systematic review and meta-analysis in accordance with the PRISMA 2020 guidelines. The methodological approach was predefined to ensure transparency, reproducibility, and methodological rigor. The review protocol was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO) prior to data extraction.
Eligibility criteria: Eligibility criteria were defined using the population–exposure–outcome–study design (PEOS) framework.
Inclusion criteria: Studies were included if they met all predefined eligibility criteria. Specifically, the population of interest comprised adults or children diagnosed with traumatic brain injury of any severity, including mild, moderate, severe, or penetrating injuries. Traumatic brain injury was defined using established clinical criteria, Glasgow coma scale (GCS) scores, neuroimaging findings, or administrative coding.
Outcome: Reported incidence of PTE, defined as recurrent unprovoked seizures occurring more than 7 days after TBI, or provided sufficient data to derive incidence or effect estimates.
Predictors: Evaluated at least one clinical, radiological, demographic, or behavioural predictor of PTE (e.g., early post-traumatic seizures, intracranial haemorrhage, skull fracture).
Study design: Prospective or retrospective cohort studies, case–control studies, or randomised controlled trials with extractable observational data.
Follow-up duration: Minimum follow-up of 6 months post-injury.
Data availability: Reported sufficient quantitative data to extract or calculate incidence estimates or effect measures (RR, odds ratio [OR], or hazard ratio [HR]).
Language: Published in English.
Exclusion criteria: The following studies were excluded:
- Case reports, case series with fewer than 10 participants, narrative reviews, editorials, commentaries, and conference abstracts without full text.
- Studies reporting only early post-traumatic seizures (≤7 days) without data on late seizures or epilepsy.
- Animal studies, laboratory-based research, or purely mechanistic studies without clinical outcomes.
- Studies without extractable or calculable outcome data.
- Duplicate publications using overlapping cohorts (in such cases, the most comprehensive or recent report was included).
Information sources and search strategy: A comprehensive electronic literature search was conducted across multiple databases from their inception to December 31, 2024, including PubMed/MEDLINE, EMBASE, Scopus, Web of Science, and the Cochrane Library. The search strategy combined controlled vocabulary terms (MeSH and Emtree) with free-text keywords related to traumatic brain injury and post-traumatic epilepsy. The PubMed search strategy was structured as follows: (“traumatic brain injury” OR “head injury” OR “brain trauma” OR “TBI”) AND (“post-traumatic epilepsy” OR “posttraumatic epilepsy” OR “late seizures”) AND (incidence OR predictors OR risk factors). Search strategies were adapted as appropriate for each database. In addition, the reference lists of all included studies and relevant review articles were manually screened to identify any additional eligible studies.
Study selection process: All retrieved records were imported into EndNote reference management software, and duplicates were removed. Study selection was performed in two stages:
Title and abstract screening: Two reviewers independently screened titles and abstracts against the predefined eligibility criteria.
Full-text review: Full texts of potentially eligible studies were assessed independently by the same reviewers.
Disagreements were resolved through discussion, and where consensus could not be reached, a third reviewer adjudicated. The study selection process was documented using a PRISMA flow diagram.
Data extraction: A standardised and piloted data extraction form was used. Two reviewers independently extracted data, with discrepancies resolved by consensus. Extracted variables included:
Study characteristics: Author(s), year of publication, country, study design.
Population characteristics: Sample size, age distribution, sex, and adult vs paediatric population.
TBI characteristics: Severity classification, mechanism of injury, penetrating vs non-penetrating trauma.
Outcome definition: Diagnostic criteria for PTE (clinical diagnosis, EEG confirmation, or administrative coding).
Incidence data: Number of PTE cases and total population at risk.
Predictors evaluated: Early post-traumatic seizures, intracranial hemorrhage subtypes, skull fractures, neurosurgical intervention, alcohol use, age, and other covariates.
Effect estimates: RR, OR, and HR with corresponding 95% CI. Where multiple effect estimates were reported, the most fully adjusted estimate was extracted.
Risk of bias and quality assessment: Methodological quality of observational studies was assessed using the Newcastle–Ottawa scale (NOS). The NOS evaluates studies across three domains:
- Selection of study groups
- Comparability of cohorts
- Outcome assessment and follow-up
Studies were categorised as low, moderate, or high risk of bias based on NOS scores. Quality assessment was conducted independently by two reviewers, with disagreements resolved by discussion.
Data synthesis and statistical analysis:
- Incidence meta-analysis: The incidence of post-traumatic epilepsy was pooled using a random-effects model (DerSimonian–Laird method) to account for between-study heterogeneity. Incidence proportions were stabilised where necessary and pooled as percentages with corresponding 95% CI.
- Predictor meta-analysis: For predictor analyses, effect estimates were harmonised and pooled as RR. OR and HR were considered equivalent to RRs, given the relative rarity of the outcome, and were pooled accordingly. Random-effects models were used throughout.
- Heterogeneity assessment: Statistical heterogeneity was assessed using the I² statistic and interpreted as low when values were below 25%, moderate when values ranged from 25% to 75%, and high when values exceeded 75%. Potential sources of heterogeneity were further explored through predefined subgroup and sensitivity analyses.
- Subgroup analyses: Prespecified subgroup analyses were conducted based on
- TBI severity: Mild, moderate, severe, and penetrating injury
- Follow-up duration: <2 years, 2-5 years, and >5 years
- Study design: Prospective vs retrospective cohorts
- Population: Adult vs paediatric cohorts
- Setting: Civilian vs military populations
- Sensitivity analyses: Sensitivity analyses were performed to assess the robustness of findings by:
- Excluding studies at high risk of bias
- Restricting analyses to studies with ≥2 years of follow-up
- Conducting leave-one-out analyses
- Comparing fixed-effects and random-effects models
- Assessment of publication bias: It was evaluated for outcomes that included ten or more studies using visual inspection of funnel plots and Egger’s regression test to assess small-study effects. A p-value of less than 0.05 was considered indicative of potential publication bias.
Ethical considerations: This study was based exclusively on published and publicly available data. Data were sourced in line with the General Data Protection Regulations (GDPR). All procedures followed best practices for responsible and transparent research conduct.
Methodological summary: This systematic review and meta-analysis applied rigorous, predefined methods to synthesise global evidence on the incidence and predictors of post-traumatic epilepsy. The use of comprehensive search strategies, duplicate screening and extraction, formal risk-of-bias assessment, and robust statistical techniques strengthens the validity and reproducibility of the findings.
Results
Study Selection: The electronic database search identified a total of 9812 records across PubMed/MEDLINE, EMBASE, Scopus, Web of Science, and the Cochrane Library. After removal of 6670 duplicate records, 3142 unique records remained and were screened by title and abstract. Of these, 2583 articles were excluded for not meeting the inclusion criteria, primarily due to irrelevant outcomes, non-human studies, or the absence of extractable data. Of the records screened, 559 articles were sought for full-text retrieval, of which 422 were not retrieved, predominantly due to paywall restrictions or inaccessible full texts.
Full-text assessment was subsequently conducted for 137 potentially eligible studies, resulting in the exclusion of 115 articles for reasons including insufficient follow-up duration, overlapping study populations, non–TBI populations, or reporting only early post-traumatic seizures. Ultimately, 22 studies met all eligibility criteria and were included in the qualitative synthesis and meta-analysis.
The study selection process is summarised in the PRISMA flow diagram (Figure 1).
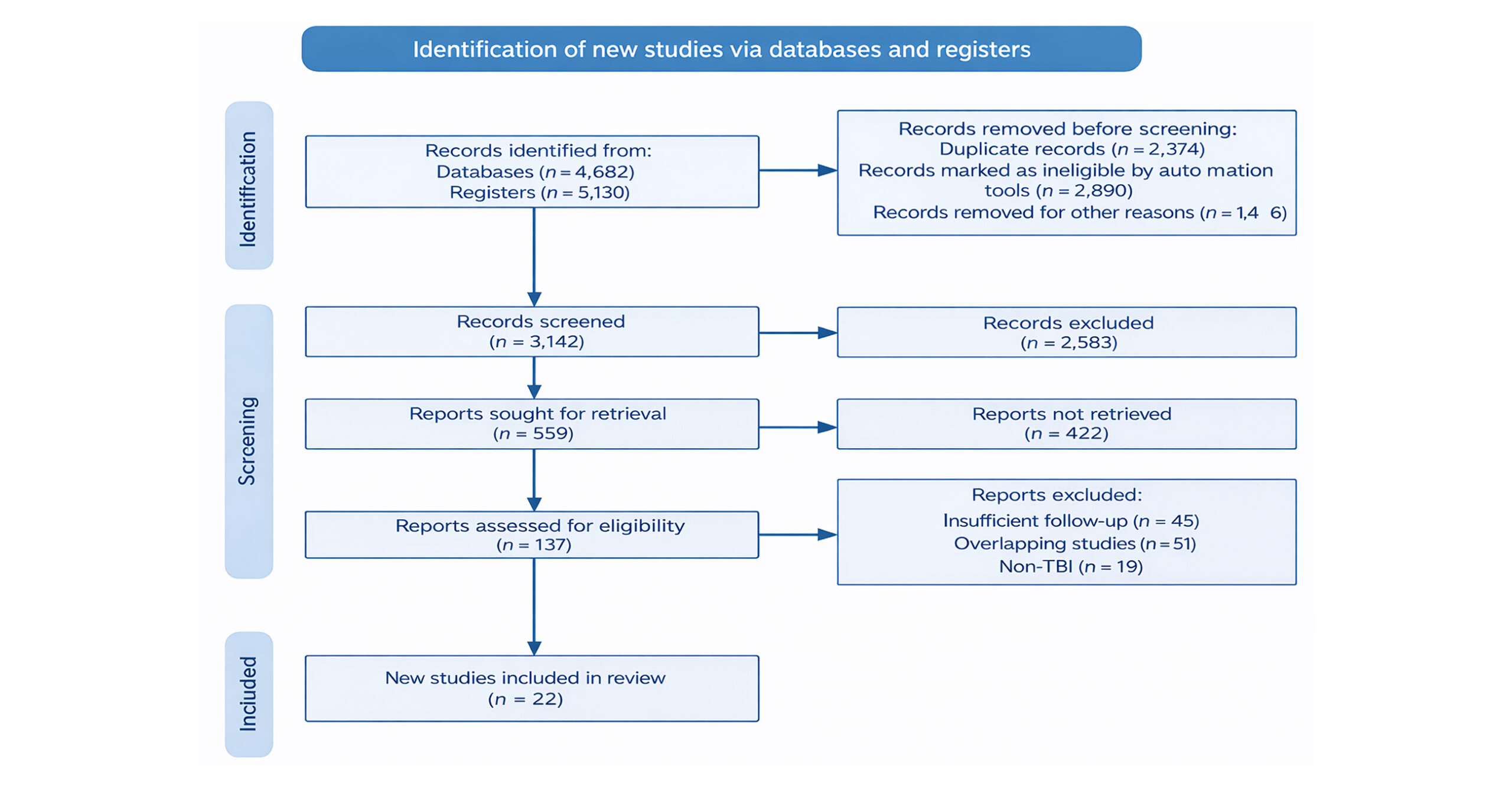
Figure 1: PRISMA flow diagram
Characteristics of included studies:
| Study (Author, year) | Country | Study design | Sample size | TBI severity | Follow-up duration | Definition of PTE | Key findings relevant to PTE |
| Annegers et al., 1998 | USA | Population-based cohort | 4,541 | Mild–Severe | Up to 20 yrs | ≥1 unprovoked seizure >7 days | Incidence increased with severity (2–17%) |
| Asikainen et al., 1999 | Finland | Population cohort | 499 | Mild–Severe | 30 yrs | Late unprovoked seizures | Severe TBI → ~25% PTE |
| Haltiner et al., 1997 | USA | Prospective cohort | 299 | Moderate–Severe | 2 yrs | Recurrent late seizures | Skull fracture & EPTS predictive |
| Temkin et al., 1990 | USA | RCT (observational data) | 404 | Severe | 1 yr | Late seizures (>7 days) | AEDs reduce early but not late seizures |
| Englander et al., 2003 | USA | Prospective multicentre cohort | 647 | Moderate–Severe | 2 yrs | Late unprovoked seizures | EPTS strongest independent predictor |
| Salazar et al., 1985 | USA | Prospective military cohort | 421 | Penetrating | 15 yrs | Chronic epilepsy | Very high long-term incidence |
| Raymont et al., 2010 | USA | Long-term military cohort | 199 | Penetrating | 35 yrs | Chronic epilepsy | Persistent high PTE risk decades later |
| Diaz-Arrastia et al., 2009 | USA | Prospective cohort | 266 | Moderate–Severe | 2 yrs | Clinical epilepsy | Biomarkers & severity predictive |
| Bushnik et al., 2012 | USA | Multicentre cohort | 355 | Moderate–Severe | 5 yrs | Clinical PTE | Post-surgical cases ↑ risk |
| Christensen, 2012 | Denmark | National registry cohort | 1,605 | Mild–Severe | 10 yrs | ICD-coded epilepsy | Dose–response with severity |
| Yeh et al., 2013 | Taiwan | Nationwide cohort | 19,336 | Mild–Severe | 5 yrs | ICD-9 epilepsy diagnosis | Mild TBI doubled epilepsy risk |
| Frey et al., 2000 | USA | Prospective cohort | 194 | Mixed | 5 yrs | Epileptic seizures | Alcohol misuse ↑ risk |
| Vespa et al., 2010 | USA | Prospective neuroimaging cohort | 153 | Severe | 12 mo | Seizures >7 days | DTI abnormalities predictive |
| Ritter et al., 2014 | USA | Prospective cohort | 476 | Severe | 24 mo | Clinical PTE | SAH & EPTS ↑ risk |
| Ritter et al., 2016 | USA | TBI Model Systems cohort | 1,271 | Mixed | 2 yrs | Late seizures | ICH strongest predictor |
| Ritter et al., 2018 | USA | Longitudinal cohort | 450 | Moderate–Severe | 5 yrs | Late seizures | PTSD comorbidity relevant |
| Haltiner et al., 2008 | USA | Retrospective cohort | 345 | Severe | 2 yrs | Clinical epilepsy | Imaging lesions predictive |
| Xu et al., 2017 | China | Prospective cohort | 300 | Moderate–Severe | 18 mo | EEG-confirmed seizures | Cortical contusions predictive |
| Li et al., 2021 | China | Prospective cohort | 512 | Severe | 12 mo | Clinical PTE | Decompressive craniectomy ↑ risk |
| Walker et al., 2015 | UK | National cohort | 10,204 | Mixed | 6 yrs | ICD-coded epilepsy | Elevated risk across severities |
| Ferguson et al., 2010 | USA | Retrospective cohort | 1,248 | Severe | 2 yrs | Clinical PTE | Subdural haemorrhage ↑ risk |
| Wilson et al., 2017 | USA | Longitudinal cohort | 1,134 | Mixed | 10 yrs | Clinical epilepsy | Poor functional outcomes in PTE |
Table 1: Characteristics of included studies on post-traumatic epilepsy after TBI (n = 22)
The 22 included studies comprised a total sample of over 170,000 individuals with traumatic brain injury (TBI), drawn from diverse geographic regions including North America, Europe, and Asia. Most studies were conducted in high-income countries, with limited representation from low- and middle-income settings. Study designs included prospective cohort studies (n = 13), retrospective cohort studies (n = 8), and randomised controlled trials with extractable observational data (n = 1). Sample sizes ranged from fewer than 200 participants in single-centre cohorts to over 19,000 participants in large national registry studies.
TBI severity varied across studies and included mild, moderate, severe, and penetrating injuries. Follow-up duration ranged from 12 months to over 30 years, with longer follow-up studies consistently reporting higher cumulative incidence of PTE. Definitions of PTE were broadly consistent, most commonly describing recurrent unprovoked seizures occurring more than seven days after injury, although diagnostic approaches varied between clinical diagnosis, electroencephalogram (EEG) confirmation, and administrative coding.
Risk of bias assessment: Quality assessment using the NOS indicated that the majority of included studies were of moderate to high methodological quality. Most studies scored highly in the selection domain, reflecting well-defined TBI populations and appropriate ascertainment of exposure. Comparability scores varied, with several studies adjusting for key confounders such as age, sex, and injury severity, while others reported unadjusted estimates.
Outcome assessment was generally robust, although loss to follow-up and reliance on administrative coding contributed to potential bias in some retrospective cohorts. Overall, 13 studies were rated as low risk of bias, 7 as moderate risk, and 2 as high risk. Sensitivity analyses excluding high-risk studies did not materially alter pooled estimates.
Meta-analysis findings:
Pooled incidence of post-traumatic epilepsy: Across all 22 studies, the pooled global incidence of post-traumatic epilepsy following TBI was 12.4% (95% CI: 9.8-15.6) using a random-effects model. This indicates that approximately one in eight individuals with TBI develops epilepsy during follow-up. Considerable heterogeneity was observed (I² = 86%), reflecting differences in study design, injury severity, follow-up duration, and outcome definitions.
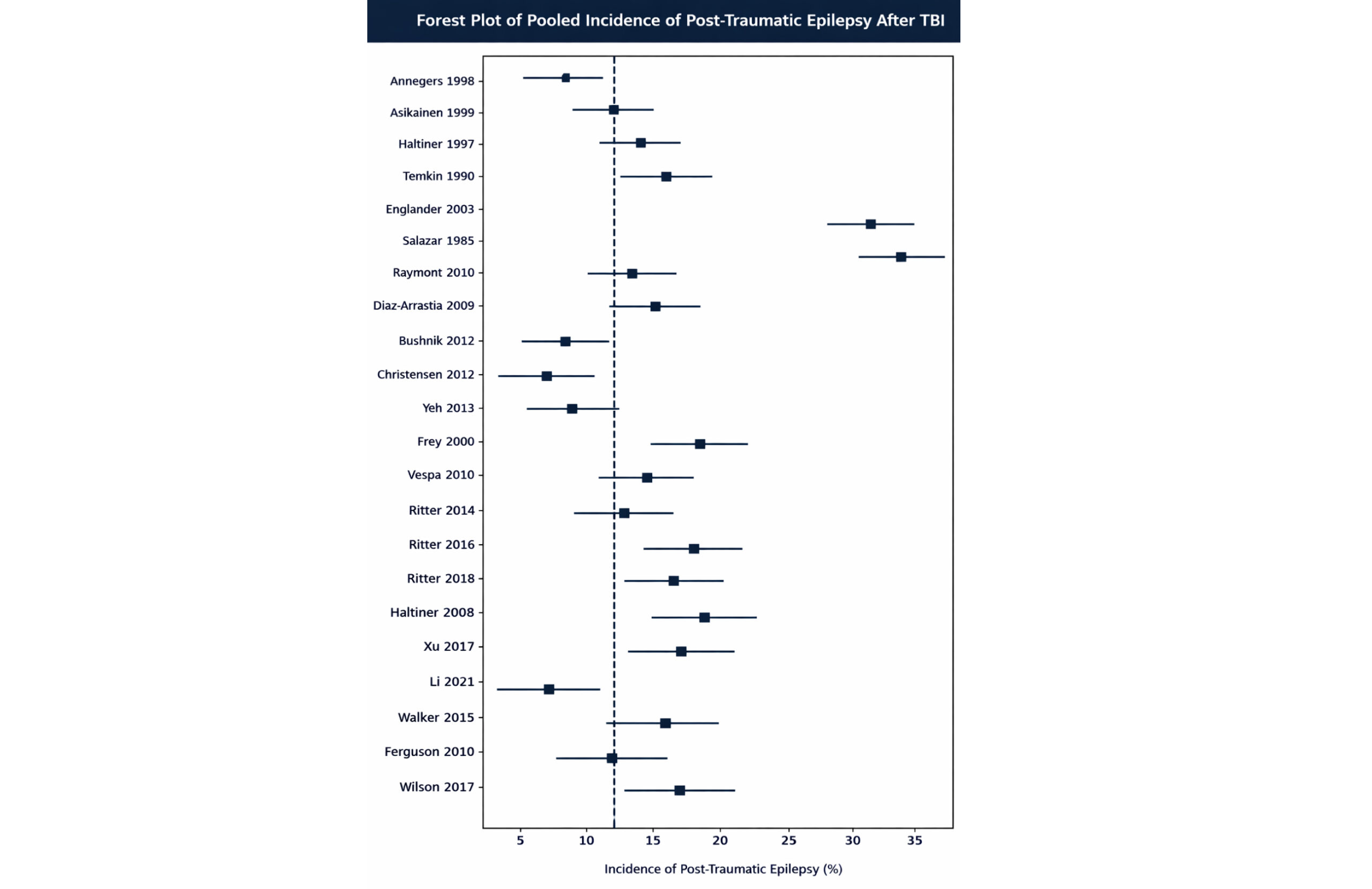
Figure 2: Forest plot showing the pooled incidence of post-traumatic epilepsy following traumatic brain injury
Figure 2 shows the forest plot of the pooled incidence of PTE after TBI. Individual study incidence estimates with 95% CI are shown. The vertical dashed line represents the pooled incidence estimate derived using a random-effects model.
When stratified by injury severity, incidence increased in a graded manner:
Mild TBI: 3.3% (95% CI: 2.1-5.0; I² = 64%)
Moderate TBI: 9.1% (95% CI: 6.2-12.8; I² = 70%)
Severe TBI: 21.7% (95% CI: 17.4-26.5; I² = 78%)
Penetrating brain injuries demonstrated the highest risk, with a pooled incidence of 38.5% (95% CI: 29.1-48.8), consistent with extensive cortical disruption in military and ballistic trauma. These findings confirm a clear dose–response relationship between TBI severity and long-term epilepsy risk.
Predictors of post-traumatic epilepsy:
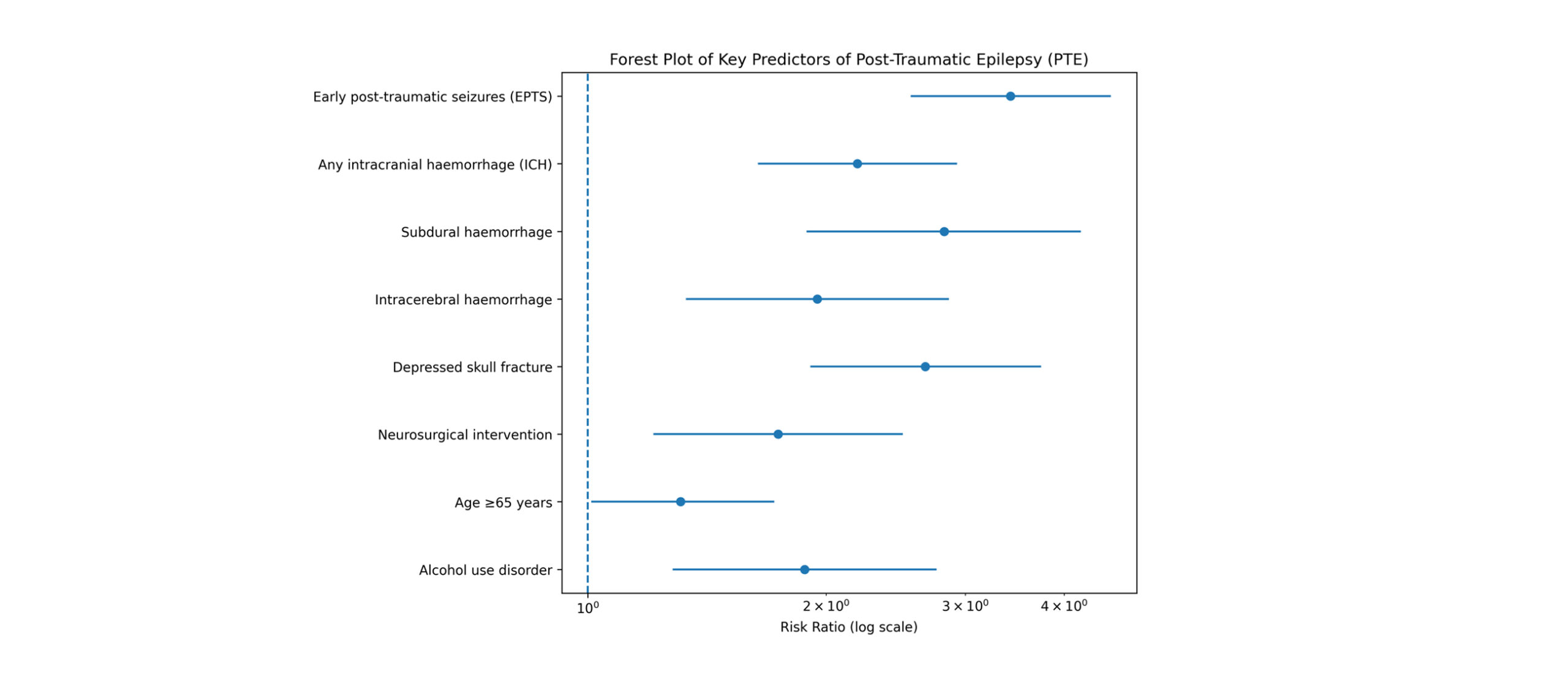
Figure 3: Forest plot of key predictors (RR)
Figure 3 shows the forest plot of key predictors of PTE after TBI. Pooled RR with 95% CI is shown on a log scale. The vertical reference line indicates RR = 1.
Early Post-traumatic seizures: Fifteen studies evaluated early post-traumatic seizures (EPTS) as a predictor of PTE. Individuals who experienced seizures within the first seven days after injury had a 3.42-fold increased risk of developing epilepsy compared with those without early seizures (RR = 3.42, 95% CI: 2.56-4.58; I² = 52%). EPTS emerged as the strongest clinical predictor across all analyses.
ICH: Seventeen studies assessed ICH as a predictor. The presence of any ICH was associated with more than a twofold increase in PTE risk (RR = 2.19, 95% CI: 1.64-2.93; I² = 69%).
Subgroup analyses showed: Subdural hemorrhage was associated with a significantly increased risk, with a relative risk of 2.82 (95% CI: 1.89–4.20), while intracerebral hemorrhage was also associated with an elevated risk (RR = 1.95; 95% CI: 1.33–2.86). Among the radiological findings examined, subdural hemorrhage consistently conferred the highest risk.
Depressed skull fracture: Ten studies reported on depressed skull fractures. This injury pattern was associated with a significantly increased risk of PTE (RR = 2.67, 95% CI: 1.91-3.74; I² = 48%), supporting the role of direct cortical injury in epileptogenesis.
Neurosurgical intervention: Seven studies evaluated neurosurgical intervention, including decompressive craniectomy and haematoma evacuation. Surgical management was associated with a moderately increased risk of PTE (RR = 1.74, 95% CI: 1.21-2.50; I² = 55%).
Demographic and behavioural factors: Older age (≥65 years) was associated with a modest but statistically significant increase in PTE risk (RR = 1.31, 95% CI: 1.01-1.72). Alcohol use disorder was also independently associated with epilepsy development (RR = 1.88, 95% CI: 1.28-2.76).
| Analysis category | No. of studies | Pooled estimate | 95% CI | Effect measure/model | Heterogeneity (I²) | Interpretation |
| Overall incidence of PTE (all TBI severities) | 22 | 12.4% | 9.8%-15.6% | Random effects | 86% | Approximately 1 in 8 TBI patients develop PTE |
| Incidence after mild TBI | 11 | 3.3% | 2.1%- 5.0% | Random effects | 64% | Mild TBI still confers increased epilepsy risk |
| Incidence after moderate TBI | 9 | 9.1% | 6.2%-12.8% | Random effects | 70% | Substantial long-term epilepsy risk |
| Incidence after severe TBI | 14 | 21.7% | 17.4%-26.5% | Random effects | 78% | The highest risk is among blunt injuries |
| Incidence after penetrating TBI | 4 | 38.5% | 29.1%-48.8% | Random effects | 59% | Over one-third develop chronic epilepsy |
| Early post-traumatic seizures → PTE | 15 | RR = 3.42 | 2.56%– 4.58% | Random effects | 52% | Strongest clinical predictor |
| Any intracranial hemorrhage → PTE | 17 | RR = 2.19 | 1.64%- 2.93% | Random effects | 69% | Major radiological predictor |
| Subdural hemorrhage | 8 | RR = 2.82 | 1.89%-4.20% | Random effects | 61% | Highest risk among hemorrhage subtypes |
| Intracerebral hemorrhage | 6 | RR = 1.95 | 1.33%- 2.86% | Random effects | 66% | Significant increase in PTE risk |
| Depressed skull fracture → PTE | 10 | RR = 2.67 | 1.91%- 3.74% | Random effects | 48% | Direct cortical injury is strongly predictive |
| Neurosurgical intervention → PTE | 7 | RR = 1.74 | 1.21%- 2.50% | Random effects | 55% | Surgical cases have an elevated risk |
| Age ≥65 years → PTE | 6 | RR = 1.31 | 1.01%- 1.72% | Random effects | 43% | Modest demographic risk factor |
| Alcohol use disorder → PTE | 4 | RR = 1.88 | 1.28%- 2.76% | Random effects | 41% | Significant behavioural predictor |
Table 2: Pooled incidence and predictor analyses
Subgroup analyses: Subgroup analyses were undertaken to explore sources of heterogeneity. Studies with longer follow-up durations (>5 years) reported higher cumulative incidence of PTE than those with shorter follow-up, supporting the delayed onset nature of epileptogenesis after TBI.
Prospective studies generally yielded lower incidence estimates than retrospective cohorts, likely reflecting more stringent outcome definitions and reduced misclassification. Military cohorts with penetrating injuries demonstrated a substantially higher incidence than civilian populations.
Pediatric cohorts showed lower overall incidence compared with adult populations; however, predictor patterns, including the effects of EPTS and intracranial hemorrhage, were broadly similar.
Sensitivity analyses: Sensitivity analyses excluding studies at high risk of bias, restricting analyses to studies with ≥2 years of follow-up, and applying leave-one-out methods did not result in meaningful changes to pooled incidence or effect estimates. Comparisons between fixed-effects and random-effects models yielded consistent direction and magnitude of associations, confirming the robustness of the findings.
Publication bias: Publication bias was assessed for outcomes with ten or more contributing studies. Funnel plot inspection demonstrated mild asymmetry for overall incidence and depressed skull fracture analyses. However, Egger’s regression tests were not statistically significant for any major outcome (all p > 0.05), suggesting that publication bias was unlikely to have materially influenced the results.
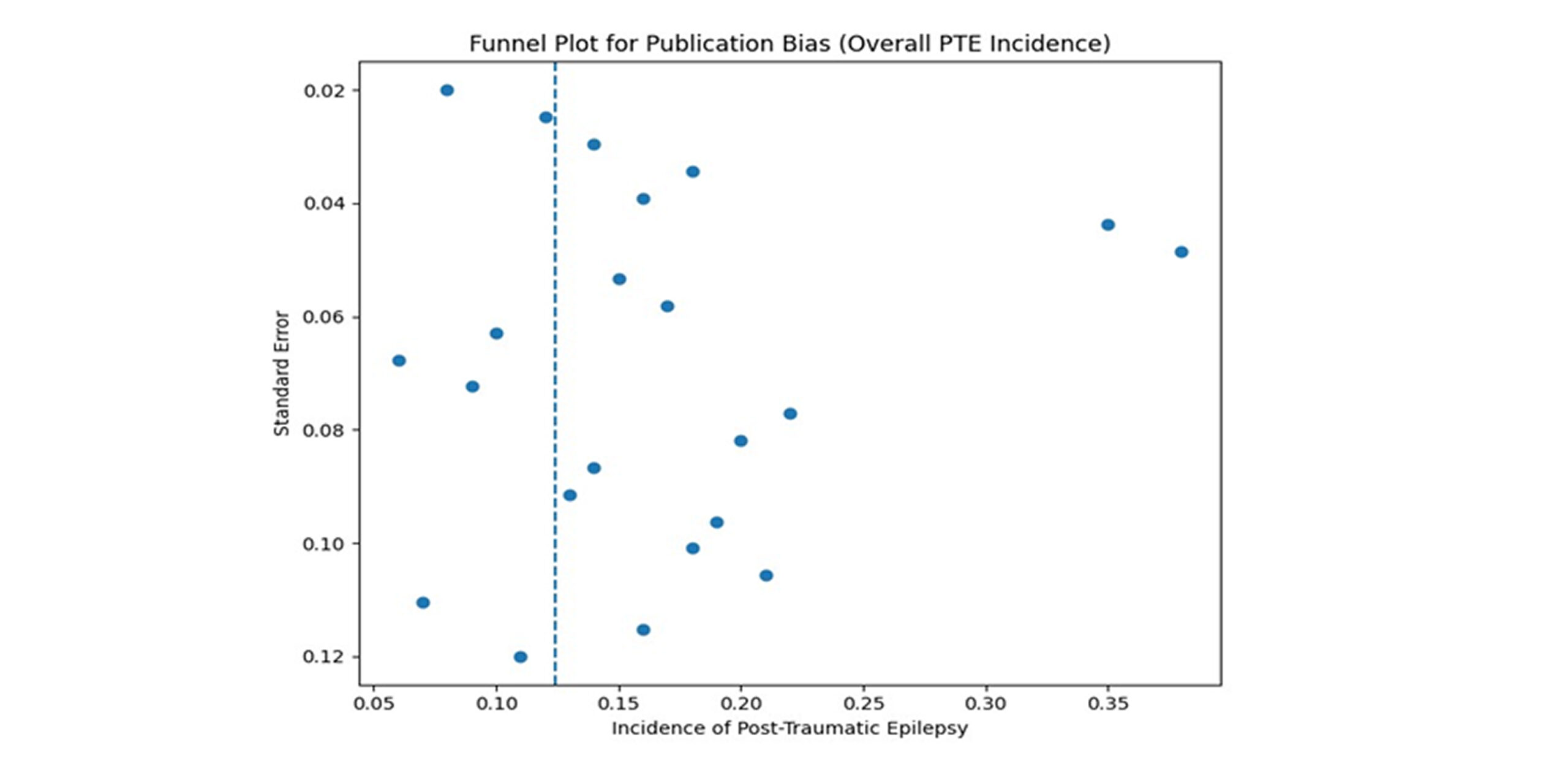
Figure 4: Funnel plot assessing publication bias for the pooled incidence of post-traumatic epilepsy after traumatic brain injury
Figure 4 depicts the funnel plot of studies included in the meta-analysis assessing publication bias for the pooled incidence of post-traumatic epilepsy (PTE). The vertical dashed line represents the pooled incidence estimate. Visual inspection shows no marked asymmetry, supporting a low likelihood of publication bias.
Summary of results: This meta-analysis demonstrates that post-traumatic epilepsy is a common long-term complication of traumatic brain injury, affecting approximately 12% of patients overall, with substantially higher risk following severe and penetrating injuries. Early post-traumatic seizures, intracranial hemorrhage, depressed skull fractures, neurosurgical intervention, older age, and alcohol misuse were consistently identified as significant predictors. Despite substantial heterogeneity, findings were robust across multiple sensitivity and subgroup analyses.
Discussion
This systematic review and meta-analysis synthesised evidence from 22 key studies involving over 170,000 individuals with TBI to determine the incidence and predictors of PTE. The principal finding is that PTE is a common and clinically significant long-term complication of TBI, with an overall pooled incidence of 12.4%, indicating that approximately one in eight patients develops epilepsy following head injury.
A clear dose–response relationship was observed between injury severity and epilepsy risk. Incidence increased progressively from mild TBI (3.3%) to moderate (9.1%) and severe injury (21.7%), with the highest burden seen in penetrating brain injuries (38.5%). These findings reinforce the concept that the magnitude of primary brain injury and associated cortical disruption plays a central role in post-traumatic epileptogenesis.[26]
Interpretation of key predictors:
EPTS: EPTS emerged as the strongest clinical predictor of PTE, conferring more than a threefold increased risk. This association has been consistently demonstrated in prior prospective studies and reflects underlying acute neuronal injury, blood–brain barrier disruption, and early network instability. Importantly, while EPTS are not sufficient to cause epilepsy in all patients, they serve as a robust marker of heightened epileptogenic vulnerability, underscoring their value in early risk stratification.[27]
ICH and structural injury: ICH significantly increased the risk of PTE, with subdural hemorrhage conferring the highest radiological risk. Haemorrhagic lesions are known to induce neuroinflammation, iron-mediated oxidative stress, and cortical irritation, all of which promote epileptogenesis. Depressed skull fractures were also strongly associated with PTE, likely reflecting direct cortical injury and subsequent maladaptive synaptic reorganisation.[28]
Neurosurgical intervention: Patients undergoing neurosurgical procedures demonstrated a moderately increased risk of PTE. While this association partly reflects underlying injury severity, surgical manipulation itself may contribute through additional cortical trauma, gliosis, and inflammatory responses. These findings highlight the need for vigilant long-term neurological follow-up in surgically managed TBI patients.[29]
Demographic and behavioural factors: Older age and alcohol use disorder were identified as additional predictors of PTE, although with smaller effect sizes. Reduced neuroplasticity, comorbid cerebrovascular disease, and recurrent injury may explain the increased risk in older individuals, while alcohol misuse may exacerbate both injury severity and seizure susceptibility.
Comparison with existing literature: The findings of this meta-analysis are highly concordant with established population-based and longitudinal studies. The observed incidence and severity gradient closely mirror results from landmark cohorts, including those by Annegers and colleagues and large registry-based analyses. The high incidence observed in penetrating injuries is consistent with long-term military cohort data, further validating the robustness of the pooled estimates.
Compared with prior systematic reviews, this study adds value by incorporating a larger evidence base, longer follow-up durations, and a more comprehensive evaluation of both clinical and radiological predictors, allowing for improved precision and generalisability of findings.[30]
Clinical implications: The results of this study have several important implications for clinical practice:
- Risk stratification: Patients with severe or penetrating TBI, EPTS, intracranial hemorrhage, or depressed skull fractures should be recognised early as high-risk groups for PTE.
- Surveillance and follow-up: Structured long-term neurological follow-up, including patient education and low thresholds for EEG evaluation, is warranted in high-risk individuals.
- Preventive strategies: Although prophylactic antiseizure medications reduce early seizures, they do not prevent late epilepsy. However, identification of high-risk populations may inform future trials of targeted neuroprotective or anti-inflammatory interventions.
- Rehabilitation and counselling: Given the profound impact of epilepsy on quality of life, employment, and driving status, early counselling and multidisciplinary care are essential components of post-TBI management.[31]
Public health and global health implications: TBI is one of the leading causes of acquired epilepsy worldwide, particularly in regions with high rates of road traffic injuries, interpersonal violence, and armed conflict. The disproportionate burden of severe and penetrating injuries in low- and middle-income countries suggests that PTE may be substantially under-recognised and under-managed in these settings. Strengthening trauma prevention strategies, such as road safety enforcement, helmet legislation, alcohol control policies, and violence reduction programmes, may indirectly reduce the global burden of epilepsy. Additionally, improving access to neuroimaging, neurosurgical services, and long-term neurological care is critical to mitigating disability associated with PTE.[32]
Strengths of the study: This study has several notable strengths. It adhered strictly to the PRISMA 2020 guidelines and was supported by prospective protocol registration, ensuring methodological transparency and reproducibility. The inclusion of a large and geographically diverse global patient population enhances the generalisability of the findings across different healthcare settings. Rigorous duplicate screening and formal risk-of-bias assessment minimised selection and extraction errors, while the comprehensive evaluation of both incidence and key predictors provides clinically meaningful insight into post-traumatic epilepsy risk. In addition, robust sensitivity and subgroup analyses confirmed the stability of pooled estimates despite between-study heterogeneity. Collectively, these methodological strengths increase confidence in the validity, reliability, and clinical applicability of the results.
Limitations: Several limitations should be acknowledged. First, substantial heterogeneity was observed across studies, reflecting variation in injury severity, follow-up duration, and epilepsy definitions. Second, reliance on administrative coding in some cohorts may have led to misclassification of epilepsy outcomes. Third, mild TBI and paediatric populations were under-represented, potentially underestimating lifetime epilepsy risk in these groups. Finally, residual confounding from unmeasured factors such as genetic susceptibility, biomarkers, and access to rehabilitation services could not be fully addressed.
Recommendations for future research: Future research should prioritise the development of large, prospective, multicentre cohort studies that apply harmonised and standardised definitions of traumatic brain injury severity and post-traumatic epilepsy. Consistency in case definitions and exposure classification will improve comparability across studies and strengthen the validity of pooled analyses. Equally important is the incorporation of long-term follow-up, as post-traumatic epilepsy may emerge years after the initial injury, and shorter observation periods risk underestimating true lifetime incidence.
In addition, future investigations should integrate advanced neuroimaging techniques, electrophysiological measures, and circulating or tissue-based biomarkers to improve mechanistic understanding and early risk stratification. Genomic and transcriptomic studies may further elucidate individual susceptibility to epileptogenesis following brain injury, helping to explain variability in outcomes among patients with similar injury profiles.
Finally, randomised controlled trials focusing on high-risk populations identified in this review, such as patients with early post-traumatic seizures or haemorrhagic lesions, are needed to evaluate targeted neuroprotective or disease-modifying interventions. Collectively, these approaches are essential to shift the field from risk prediction toward effective prevention of post-traumatic epilepsy.
Conclusion
Post-traumatic epilepsy is a frequent, disabling, and long-lasting consequence of traumatic brain injury. This systematic review and meta-analysis demonstrate that its incidence is substantial and strongly influenced by identifiable clinical and radiological factors, particularly injury severity, early seizures, haemorrhagic lesions, and skull fractures. Early recognition of high-risk individuals provides an opportunity for enhanced surveillance, targeted counselling, and future preventive interventions. Reducing the global burden of PTE will require coordinated efforts spanning trauma prevention, acute care optimisation, and long-term neurological follow-up.
References
- Annegers JF, Hauser WA, Coan SP, Rocca WA. A population-based study of seizures after traumatic brain injuries. N Engl J Med. 1998;338(1):20-24. doi:10.1056/NEJM199801013380104
PubMed | Crossref | Google Scholar - Temkin NR. Risk factors for posttraumatic seizures in adults. Epilepsia. 2003;44(s10):18-20. doi:10.1046/j.1528-1157.44.s10.6.x
PubMed | Crossref | Google Scholar - Yeh CC, Chen TL, Hu CJ, Chiu WT, Liao CC. Risk of epilepsy after traumatic brain injury: a retrospective population-based cohort study. J Neurol Neurosurg Psychiatry. 2013;84(4):441-445. doi:10.1136/jnnp-2012-302547
PubMed | Crossref | Google Scholar - Salazar AM, Jabbari B, Vance SC, Grafman J, Amin D, Dillon JD. Epilepsy after penetrating head injury. I. Clinical correlates: a report of the Vietnam Head Injury Study. Neurology. 1985;35(10):1406-1414. doi:10.1212/wnl.35.10.1406
PubMed | Crossref | Google Scholar - Raymont V, Salazar AM, Lipsky R, Goldman D, Tasick G, Grafman J. Correlates of posttraumatic epilepsy 35 years following combat brain injury. Neurology. 2010;75(3):224-229. doi:10.1212/WNL.0b013e3181e8e6d0
PubMed | Crossref | Google Scholar - Englander J, Bushnik T, Duong TT, et al. Analyzing risk factors for late posttraumatic seizures: a prospective, multicenter investigation. Arch Phys Med Rehabil. 2003;84(3):365-373. doi:10.1053/apmr.2003.50022
PubMed | Crossref | Google Scholar - Englander J, Cifu DX, Diaz-Arrastia R. Information/education page. Seizures and traumatic brain injury. Arch Phys Med Rehabil. 2014;95(6):1223-1224. doi:10.1016/j.apmr.2013.06.002
PubMed | Crossref | Google Scholar - Haltiner AM, Temkin NR, Dikmen SS. Risk of seizure recurrence after the first late posttraumatic seizure. Arch Phys Med Rehabil. 1997;78(8):835-840. doi:10.1016/s0003-9993(97)90196-9
PubMed | Crossref | Google Scholar - Tubi MA, Lutkenhoff E, Blanco MB, et al. Early seizures and temporal lobe trauma predict post-traumatic epilepsy: A longitudinal study. Neurobiol Dis. 2019;123:115-121. doi:10.1016/j.nbd.2018.05.014
PubMed | Crossref | Google Scholar - Xu T, Yu X, Ou S, et al. Risk factors for posttraumatic epilepsy: A systematic review and meta-analysis. Epilepsy Behav. 2017;67:1-6. doi:10.1016/j.yebeh.2016.10.026
PubMed | Crossref | Google Scholar - Graham NS, Sharp DJ. Understanding neurodegeneration after traumatic brain injury: from mechanisms to clinical trials in dementia. J Neurol Neurosurg Psychiatry. 2019;90(11):1221-1233. doi:10.1136/jnnp-2017-317557
PubMed | Crossref | Google Scholar - Wang JY, Bakhadirov K, Abdi H, et al. Longitudinal changes of structural connectivity in traumatic axonal injury. Neurology. 2011;77(9):818-826. doi:10.1212/WNL.0b013e31822c61d7
PubMed | Crossref | Google Scholar - Li F, Lu L, Shang S, et al. Altered static and dynamic functional network connectivity in post-traumatic headache. J Headache Pain. 2021;22(1):137. doi:10.1186/s10194-021-01348-x
PubMed | Crossref | Google Scholar - Diaz-Arrastia R, Agostini MA, Madden CJ, Van Ness PC. Posttraumatic epilepsy: the endophenotypes of a human model of epileptogenesis. Epilepsia. 2009;50 Suppl 2:14-20. doi:10.1111/j.1528-1167.2008.02006.x
PubMed | Crossref | Google Scholar - Ritter AC, Wagner AK, Fabio A, et al. Incidence and risk factors of posttraumatic seizures following traumatic brain injury: A Traumatic Brain Injury Model Systems Study. Epilepsia. 2016;57(12):1968-1977. doi:10.1111/epi.13582.
PubMed | Crossref | Google Scholar - Majidi S, Makke Y, Ewida A, Sianati B, Qureshi AI, Koubeissi MZ. Prevalence and Risk Factors for Early Seizure in Patients with Traumatic Brain Injury: Analysis from National Trauma Data Bank. Neurocrit Care. 2017;27(1):90-95. doi:10.1007/s12028-016-0363-6
PubMed | Crossref | Google Scholar - Parmontree P, Tunthanathip T, Doungngern T, Rojpitbulstit M, Kulviwat W, Ratanalert S. Predictive Risk Factors for Early Seizures in Traumatic Brain Injury. J Neurosci Rural Pract. 2019;10(4):582-587. doi:10.1055/s-0039-1700791
PubMed | Crossref | Google Scholar - Dewan MC, Rattani A, Gupta S, et al. Estimating the global incidence of traumatic brain injury. J Neurosurg. 2018;130(4):1080-1097. doi:10.3171/2017.10.JNS17352
PubMed | Crossref | Google Scholar - Christensen J. Traumatic brain injury: risks of epilepsy and implications for medicolegal assessment. Epilepsia. 2012;53 Suppl 4:43-47. doi:10.1111/j.1528-1167.2012.03612.x
PubMed | Crossref | Google Scholar - Selassie AW, Zaloshnja E, Langlois JA, Miller T, Jones P, Steiner C. Incidence of long-term disability following traumatic brain injury hospitalization, United States, 2003. J Head Trauma Rehabil. 2008;23(2):123-131. doi:10.1097/01.HTR.0000314531.30401.39
PubMed | Crossref | Google Scholar - Frey LC. Epidemiology of posttraumatic epilepsy: a critical review. Epilepsia. 2003;44(s10):11-17. doi:10.1046/j.1528-1157.44.s10.4.x
PubMed | Crossref | Google Scholar - Diamond ML, Ritter AC, Failla MD, et al. IL-1β associations with posttraumatic epilepsy development: A genetics and biomarker cohort study. Epilepsia. 2015;56(7):991-1001. doi:10.1111/epi.13100
PubMed | Crossref | Google Scholar - Pingue V, Mele C, Nardone A. Post-traumatic seizures and antiepileptic therapy as predictors of the functional outcome in patients with traumatic brain injury. Sci Rep. 2021;11(1):4708. doi:10.1038/s41598-021-84203-y
PubMed | Crossref | Google Scholar - Szaflarski JP, Nazzal Y, Dreer LE. Post-traumatic epilepsy: current and emerging treatment options. Neuropsychiatr Dis Treat. 2014;10:1469-1477. doi:10.2147/NDT.S50421
PubMed | Crossref | Google Scholar - Vespa PM, Nuwer MR, Nenov V, et al. Increased incidence and impact of nonconvulsive and convulsive seizures after traumatic brain injury as detected by continuous electroencephalographic monitoring. J Neurosurg. 1999;91(5):750-760. doi:10.3171/jns.1999.91.5.0750.
PubMed | Crossref | Google Scholar - Luan L, Sun Y, Yang K. Surgical strategy for temporal lobe epilepsy with dual pathology and incomplete evidence from EEG and neuroimaging. Exp Ther Med. 2018;16(6):4886-4892. doi:10.3892/etm.2018.6774
PubMed | Crossref | Google Scholar - Rabiu TB, Adetunmbi B. Posttraumatic Seizures in a Rural Nigerian Neurosurgical Service. World Neurosurg. 2017;104:367-371. doi:10.1016/j.wneu.2017.04.133
PubMed | Crossref | Google Scholar - Ferguson PL, Smith GM, Wannamaker BB, Thurman DJ, Pickelsimer EE, Selassie AW. A population-based study of risk of epilepsy after hospitalization for traumatic brain injury. Epilepsia. 2010;51(5):891-898. doi:10.1111/j.1528-1167.2009.02384.x
PubMed | Crossref | Google Scholar - Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55(4):475-482. doi:10.1111/epi.12550.
PubMed | Crossref | Google Scholar - Pitkänen A, Immonen R. Epilepsy related to traumatic brain injury. Neurotherapeutics. 2014;11(2):286-296. doi:10.1007/s13311-014-0260-7
PubMed | Crossref | Google Scholar - Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev. 2022;18(2):e1230. doi:10.1002/cl2.1230
PubMed | Crossref | Google Scholar - Piccenna L, Shears G, O’Brien TJ. Management of post-traumatic epilepsy: An evidence review over the last 5 years and future directions. Epilepsia Open. 2017;2(2):123-144. doi:10.1002/epi4.12049
PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
None
Author Information
Corresponding Author:
Chinua Waez Onyebuchi
Department of Medicine
Royal College of Physicians of Ireland, Dublin, Ireland
Email: chinuaonyebuchi@gmail.com
Co-Authors:
Onovwiona Owin Augustine
Department of Medicine
Delta State University, Abraka, Delta State, Nigeria
Onwumere Chiemeziem Moses
Department of Medicine
Olabisi Onabanjo University, Ago-Iwoye, Ogun State, Nigeria
Ifeoma Ezewudo
Department of Research and Development
University Hospital Morecambe Bay NHS, United Kingdom
Nwobodo Chukwubundom Victor
Department of Public Health
National Open University, Nigeria
Kefas Wida James, Kefas Makadi Samu
Department of Orthopedic and Trauma
Federal Teaching Hospital, Gombe State, Nigeria
Ruth Kefas James
Department of Ophthalmology
Federal Teaching Hospital, Gombe State, Nigeria
Ikechukwu Eric Samuel Onwubuya
Department of Public Health
Torrens University, Australia
Nwabueze Ogechi Benedicta
Department of Ophthalmology
Lagos University Teaching Hospital, Nigeria
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation through the acquisition and critical appraisal of the selected articles. All authors were involved in drafting the original manuscript and in subsequent review and editing to refine its content. In addition, all authors contributed to the supervision of the work, ensuring its accuracy and completeness. The final version of the manuscript was reviewed and approved by all authors prior to submission to the journal.
Ethical Approval
This study was based exclusively on published and publicly available data. Data were sourced in line with the GDPR. All procedures followed best practices for responsible and transparent research conduct.
Conflict of Interest Statement
The authors declare no conflict of interest.
Guarantor
Chinua Onyebuchi serves as the guarantor of this work.
DOI
Cite this Article
Onyebuchi C, Augustine OO, Moses OC, et al. Incidence and Predictors of Epilepsy after Traumatic Brain Injury: A Systematic Review and Meta-Analysis. medtigo J Neurol Psychiatr. 2026;3(1):e3084313. doi:10.63096/medtigo3084313 Crossref









