Author Affiliations
Abstract
Background: Guidelines are used in clinical practice to provide clear direction and supporting evidence for a range of conditions pertinent to that practitioner. In Developmental Dysplasia of the Hip (DDH), several guidelines exist detailing management.
Case report: This case chronicles a 13-month-old toddler who demonstrated indications of hip dysplasia. Ultrasound at 31 days of age revealed physiologically immature hips. A recommendation for observation was initiated at this point. At 14 weeks of age, ultrasound revealed an 11-degree alpha angle variance (72° right, 61° left) and a recommendation to review with a general medical practitioner at nine months of age. At nine months of age, x-ray imaging revealed a right acetabular angle of 24°, a left acetabular angle of 34.5°, and a clear disruption to Shenton’s line signifying left acetabular dysplasia. Several guidelines are available, yet errors in management are still occurring, which may reflect a lack of clarity and action within these guidelines regarding clinical intervention. Without regular assessment, dysplastic changes were not detected until nine months of age.
Conclusion: Results of examination and screening ultrasound should undergo careful interpretation, with significant asymmetry interpreted as abnormal development and treated appropriately to avoid long-term disability. Emphasis must be on early detection for appropriate management.
Keywords
Developmental dysplasia of the hip, Infants, Newborn, Hip joint, Primary health care.
Introduction
DDH refers to a spectrum of abnormalities of the femoral head and acetabulum. DDH encompasses laxity of the joint, unstable, subluxated, and dislocated hips, as well as malformation in the shape of the acetabulum.[1,2] Normal development of the hip joint relies on the co-dependent relationship between the femoral head and the acetabulum. A lack of sphericity and congruence will occur if either structure is abnormal.[3]
Previously referred to as congenital hip dysplasia, DDH more accurately reflects the progressive and developmental nature of the condition, as these abnormalities are not always present at birth.[4]
Due to the developmental nature of the condition, there is a lack of definitive parameters, both clinically and radiologically, as to what constitutes DDH.[5] The developmental nature of DDH means that a single measure of incidence for DDH is overly simplistic. Diagnosis of DDH is often made in the first year of life, but evidence of DDH can be detected in children up to 5 years old.[2,4,6]
The American Academy of Pediatrics screening guidelines suggest hip checks at birth, 1, 2, 4, 6, 9, and 12 months of age; however, guideline-concordant screening is not always followed by primary care providers until walking age.[1,6]
Children born with known risk factors should raise the level of suspicion for the presence of DDH, as early diagnosis and treatment are important in reducing or preventing the long-term sequelae, which include early osteoarthritis, hip preservation surgery, early hip replacement, and chronic pain and loss of productivity.[5,7]
| Female | First born |
| Breech presentation | Family history |
| Oligohydramnios | Birth weight > 5 kg |
| Post maturity | Multiple birth |
Table 1: Risk factors for the development of DDH
Allied health professionals are well placed to screen for and participate in the initial management of DDH. Chiropractors often see infants for routine clinical observation and for screening and management of a range of musculoskeletal conditions, which may include DDH.[8]
This case study chronicles an infant who presented for further advice and management of DDH following delayed treatment and non-guideline concordant medical advice. It highlights the importance of careful interpretation of examination and screening. Consent was attained from the patient’s caregiver for the use of case details in publication. This case was prepared utilizing the Case Report (CARE) guidelines.[9]
Case Presentation
The patient was born weighing 2.68 kg at 37+6 weeks’ gestation via emergency caesarean section at a private hospital in Australia. It was the mother’s first pregnancy, during which she was diagnosed with anemia and low serum pregnancy-associated plasma protein-A (PAPP-A). There was no oligohydramnios nor family history of hip dysplasia; however, she was born in a breech position. There were no concerns with breastfeeding, nor with the introduction of solids.
Case Management
The patients’ initial assessment was performed by a pediatrician at 4 days of age. Hip assessment demonstrated normal hip abduction with subluxation, but no dislocation was present. This prompted an ultrasound review at 4 weeks of age. At 31 days of age, ultrasound demonstrated physiological hip immaturity (Graf hip type 2a+ bilaterally) and asymmetry (Alpha angle 57° right, 50° left). A further review was recommended; however, it was determined that bracing was not required.
At 8 weeks of age (1M28D), repeat ultrasound was performed, and this showed physiological immaturity present bilaterally (Graf hip type 2a+); however, alpha angles were now considered consistent with hip dysplasia (Alpha angle 57° right, 56° left). As physical examination showed normal hip abduction and a negative Barlow test, the recommendation was observation without bracing and review in a further 6 weeks.
After 6 weeks (3M9D), ultrasound revealed that alpha angles had increased to 72° right and 61° left, with mild changes only to the left acetabular superior bony rim. At this point, with both hips’ alpha angles being greater than 60°, they were classified as sonographically normal (Graf type 1b). Combined with the clinical review showing normal abduction and Barlow’s test negative bilaterally, the recommendation was given to avoid swaddling, and against the recommended hip screening guideline, a follow-up review with her general practitioner (GP) at 9 months of age.
At 9 months 29 days of age (8M29D), an A-P lumbopelvic x-ray was arranged by a GP. This revealed a right acetabular angle of 24 degrees, but a left acetabular angle of 34.5° degrees. A clear disruption to Shenton’s line signifying left acetabular dysplasia was also apparent. A private pediatric orthopedic consultation resulted in the recommendation of rhino bracing for 23 hours per day for a period of 12 weeks. The parents were advised that there may be difficulty with tolerating the brace, and due to a rash appearing after one week, Denis Browne bar bracing was utilized during naps and night-time in conjunction with the rhino brace.
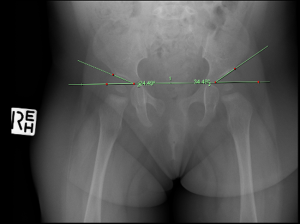
Figure 1: X-ray at 8M29D Showing Acetabular Angles Prior to Bracing
Figure 1 shows a right acetabular angle measure of 24.49° and a left of 34.49°. Repeat x-ray at 12 months of age (12M8D) showed a one-degree improvement in left acetabular angle to 33 degrees, with the right acetabular angle not measured. The pediatric orthopedic specialist recommended a further 12 weeks of bracing at 23 hours per day. Due to the poor response to bracing and compliance issues, the parents at this point ceased bracing.
| Age | Imaging modality (angle measured) | Right | Left |
| 31D | Ultrasound (alpha angle) | 57° | 50° |
| 1M28D | Ultrasound (alpha angle) | 57° | 56° |
| 3M9D | Ultrasound (alpha angle) | 72° | 61° |
| 8M29D | X-ray (Acetabular angle) | 24.5° | 34.5° |
| 12M8D | X-ray (Acetabular angle) | * | 33° |
Table 2: Summary of hip angle progression over time (D; days, M; months, *; angle not reported)
Discussion
This case chronicles an infant with several risk factors and mild dysplasia evident on ultrasound who was not monitored at all between 3.5 and 9 months of age. The long-term sequelae of DDH are well documented.[1,2] Newborn screening followed by routine surveillance until walking age is recognized as best practice care by the American Academy of Pediatrics.[3] Routine screening includes, but is not limited to, the Ortolani and Barlow maneuvers to test stability, Allis test (Galeazzi sign) assessing thigh shortening. After 12 weeks of age, hip abduction asymmetry is the main clinical sign.[4,5] Selective ultrasound screening is used for patients with known risk factors such as breech presentation and a positive family history, or where clinical examination is suggestive or inconclusive.[6] Sonography allows for the visualization of the femoral head relative to the acetabulum. It is useful up to approximately 4-6 months of age, after which the ossifying nucleus of the femoral head appears, and radiographic studies become the most appropriate form of evaluation.[3] Sonographic assessment may be performed dynamically to assess instability or statically, using the Graf criteria to assess and grade morphology.[7] Graf et al described the alpha angle formed between the bony ilium and bony roof of the acetabulum and the beta angle between the ilium and the cartilaginous labrum. The alpha angle, whose significance is like that of the acetabular index, reflects the degree of inclination of the bony acetabular roof, with > 60° classified as normal.[10] Whilst the Graf rating system is widely used, it has a poor intra- and interobserver reliability and relatively high interscan variability.[8,11,12]
This report describes the presentation and management of an infant initially classified with physiologically immature hips (Graf 2a) at 1 month of age with alpha angles of 57° on the right and 50° on the left. A repeat ultrasound at 14 weeks of age measured alpha angles of 72° on the right and 61° on the left, and was classified as normal (Graf 1b), and a review was recommended at 9 months of age. Several considerations exist in the literature suggesting that this may not have been best practice. As the inter-observer variation allowance of the Graf criteria is 2°, it could be argued that the left hip measurement of 61° could be classified borderline at the 14-week assessment and at risk of developing poorly. Considering that the rate of normalization of newborn girls with Graf 2a hips is much lower than that of boys, the infant should therefore have been managed with a higher level of concern.[7,13] There is evidence in the literature to suggest that an asymmetry between the two hips greater than 4° is suggestive of a potential development of a dysplastic hip.[14] In this case, there was an 11° difference, which warranted closer supervision. The 6-month interval between hip assessments in this case, which exceeded guideline-recommended screening intervals for low-risk infants, resulted in a delayed diagnosis with a presentation at 9 months of age with a clearly dysplastic left hip.
In Australia, several guidelines/protocols for screening DDH are available; however, only half of them outline routine screening timeframes. Child Health Books for each state and territory, except for Victoria and Northern Territory, mentioned routine serial screening for DDH, but were not consistent in the timeframes suggested.[15] The increase in incidence of late-detected DDH in Australia may in part be due to the lack of clearly defined guidelines and referral pathways. However, in one Australian study, only 43% of clinicians reported using guidelines, and 51% reported receiving any training in DDH screening beyond their primary clinical training.[16]
Despite a large body of literature regarding DDH, discrepancies in management strategies also exist, primarily due to a lack of prospective randomized comparative trials, a lack of homogeneity in grading DDH, and no common definition of a successful outcome.[17–20] Current treatment with dynamic bracing, that allows some limited movement of the legs, particularly if provided within the first 4-5 months of life, has been shown to be both safe and effective, with the Pavlik harness having a success rate, defined as a regression of dysplasia and recovery of the hip, of 91.1%.[21] Static splinting, most commonly with a Rhino brace or Denis Browne bar, involves a fixed hip abduction and flexion with no possibility of hip movement. It is often recommended beyond six months of age; however, there is limited evidence to support the use, with only some improvements in the acetabular angle observed.[22] There is also limited evidence for the use of part-time bracing as was recommended in this case, with some reduction in the acetabular index previously demonstrated, yet not achieving a normative range. The optimal duration of splinting has also yet to be determined.
While the long-term complications and impact of DDH are well documented, there are few long-term studies evaluating the expense associated with late-diagnosed DDH.[23-25] A 2014 cost analysis of medical expenses in individuals with late-detected DDH found that it was £4,352 per child in 2008 when surgical intervention was required.[26] In comparison, a 2015 3-year analysis of cases of late diagnosed DDH was $10,655.25 USD per child over the course of three years.[27] Contrast this to the cost of early detection; £601 and $1,730USD per child, respectively. With an incidence ranging from 0.77 to 1.28 per 1000 births, this represents significant medical costs to a substantial percentage of the population.[28,29] In the case discussed, with failed harnessing and likely needing surgical intervention, Woodacre (2014) highlights a greater expense of £7,025 per child in 2008, approximately £10,418.18 or $17,837.90AUD allowing for inflation in 2023.
In this case, a period of static bracing for 12 weeks (at 9-12 months of age) resulted in a one-degree change in acetabular angle, which is within expected measuring variation. Despite years of research, there remains a paucity of high-quality evidence to guide the diagnosis and management of late-diagnosed DDH.[30] Current medical guidelines recommend observation for minor hip abnormalities or mild DDH until 4 months of age, with insufficient evidence to recommend treatment over observation.[31,32] Regardless of the presence or suspicion of DDH in infants, best practice recommends surveillance screening until walking age, with timeframes outlined in at least 4 Australian guidelines in accordance with the American Academy of Pediatrics.[32-35]
Conclusion
There are several guidelines published in Australia detailing the assessment and management of infants with an indication of DDH. However, there appears to be poor translation into clinical practice: in a recent analysis of practice regarding DDH screening, more than half of the clinicians involved failed to appropriately apply these guidelines. This case highlights the ramifications of a lack of concordance with clinical practice guidelines and surveillance protocols. With the increase in prevalence of late diagnosed DDH in Australia, and the known long-term costs and disability resulting from the condition, emphasis should be placed on ensuring clinicians are aware of the importance of serial examination and screening, clear documentation, and appropriate referral pathways for at-risk infants and children.
References
- Cady RB. Developmental dysplasia of the hip: definition, recognition, and prevention of late sequelae. Pediatr Ann. 2006;35(2):92-101. doi.10.3928/0090-4481-20060201-09 PubMed | Crossref| Google Scholar
- Wedge JH, Wasylenko MJ. The natural history of congenital disease of the hip. J Bone Joint Surg Br. 1979;61-B(3):334-338. doi:10.1302/0301-620X.61B3.158025 PubMed | Crossref | Google Scholar
- Yang S, Zusman N, Lieberman E, Goldstein RY. Developmental Dysplasia of the Hip. Pediatrics. 2019;143(1):e20181147. doi:10.1542/peds.2018-1147 PubMed | Crossref | Google Scholar
- Swarup I, Penny CL, Dodwell ER. Developmental dysplasia of the hip: an update on diagnosis and management from birth to 6 months. Curr Opin Pediatr. 2018;30(1):84-92. doi:10.1097/MOP.0000000000000574 PubMed | Crossref | Google Scholar
- Čustović S, Šadić S, Vujadinović A, et al. The predictive value of the clinical sign of limited hip abduction for developmental dysplasia of the hip (DDH). Med Glas (Zenica). 2018;15(2):174-178. doi:10.17392/954-18 PubMed | Crossref | Google Scholar
- Shorter D, Hong T, Osborn DA. Screening programmes for developmental dysplasia of the hip in newborn infants. Cochrane Database Syst Rev. 2011;2011(9):CD004595. doi:10.1002/14651858.CD004595.pub2 PubMed | Crossref | Google Scholar
- Graf R. Fundamentals of sonographic diagnosis of infant hip dysplasia. J Pediatr Orthop. 1984;4(6):735-740. doi:10.1097/01241398-198411000-00015 PubMed | Crossref | Google Scholar
- Bucher F, Görg C, Weber S, Peterlein CD. Paediatric hip ultrasound: uncertainties in examination and choice of treatment. J Child Orthop. 2021;15(1):42. doi.10.1302/1863-2548.15.200084 PubMed | Crossref | Google Scholar
- Riley DS, Barber MS, Kienle GS, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol. 2017;89:218-235. doi:10.1016/j.jclinepi.2017.04.026 PubMed | Crossref | Google Scholar
- Liu B, Hu X, Li L, Gao S. Morphological development of the hip in normal infants under six months of age by the Graf ultrasound method. Front Pediatr. 2022;10:914545. doi.10.3389/FPED.2022.914545 PubMed | Crossref | Google Scholar
- Kolb A, Benca E, Willegger M, Puchner SE, Windhager R, Chiari C. Measurement considerations on examiner-dependent factors in the ultrasound assessment of developmental dysplasia of the hip. Int Orthop. 2017;41(6):1245-1250. doi:10.1007/s00264-017-3455-9 PubMed | Crossref | Google Scholar
- Roposch A, Graf R, Wright JG. Determining the reliability of the Graf classification for hip dysplasia. Clin Orthop Relat Res. 2006;447:119-124. doi:10.1097/01.blo.0000203475.73678.be PubMed | Crossref | Google Scholar
- Omeroğlu H, Caylak R, Inan U, Köse N. Ultrasonographic Graf type IIa hip needs more consideration in newborn girls. J Child Orthop. 2013;7(2):95-98. doi:10.1007/s11832-012-0476-1 PubMed | Crossref | Google Scholar
- Vasilescu D, Cosma D, Vasilescu DE, Solomon CM, Corbu A, Dudea SM. Ultrasonography of borderline and asymmetric infant hips – a challenge in the management of developmental dysplasia of the hip. Clujul Med. 2018;91(4):422-426. doi:10.15386/cjmed-1047 PubMed | Crossref | Google Scholar
- Marriott E, Twomey S, Lee M, Williams N. Variability in Australian screening guidelines for developmental dysplasia of the hip. J Paediatr Child Health. 2021;57(12):1857-1865. doi:10.1111/jpc.15744 PubMed | Crossref | Google Scholar
- Wright S, Cotterell E, Schmidt D. Screening for developmental dysplasia of the hip in a rural health district: An analysis of practice. Aust J Rural Health. 2018;26(3):199-205. doi:10.1111/ajr.12400 PubMed | Crossref | Google Scholar
- Cooper AP, Doddabasappa SN, Mulpuri K. Evidence-based management of developmental dysplasia of the hip. Orthop Clin North Am. 2014;45(3):341-354. doi:10.1016/j.ocl.2014.03.005 PubMed | Crossref | Google Scholar
- Taylor IK, Burlile JF, Schaeffer EK, et al. Developmental Dysplasia of the Hip: An Examination of Care Practices of Pediatric Orthopaedic Surgeons in North America. J Pediatr Orthop. 2020;40(4):e248-255. doi:10.1097/BPO.0000000000001505 PubMed | Crossref | Google Scholar
- Zomar BO, Mulpuri K, Schaeffer EK. Examining the Short-Term Natural History of Developmental Dysplasia of the Hip in Infancy: A Systematic Review. Indian J Orthop. 2021;55(6):1372-1387. doi:10.1007/s43465-021-00510-6 PubMed | Crossref | Google Scholar
- Dygut J, Sułko J, Guevara-Lora I, Piwowar M. Secondary (Late) Developmental Dysplasia of the Hip with Displacement: From Case Studies to a Proposition for a Modified Diagnostic Path. Diagnostics (Basel). 2022;12(6):1472. doi:10.3390/diagnostics12061472 PubMed | Crossref | Google Scholar
- Gans I, Flynn JM, Sankar WN. Abduction bracing for residual acetabular dysplasia in infantile DDH. J Pediatr Orthop. 2013;33(7):714-718. doi:10.1097/BPO.0b013e31829d5704 PubMed | Crossref | Google Scholar
- Pavone V, de Cristo C, Vescio A, et al. Dynamic and Static Splinting for Treatment of Developmental Dysplasia of the Hip: A Systematic Review. Children (Basel). 2021;8(2):104. doi:10.3390/children8020104 PubMed | Crossref | Google Scholar
- Thomas SR. A review of long-term outcomes for late presenting developmental hip dysplasia. Bone Joint J. 2015;97-B(6):729-733. doi:10.1302/0301-620X.97B6.35395 PubMed | Crossref | Google Scholar
- Petra M, Benson MKD. Long-term outcomes in developmental dysplasia of the hip. Hip Int. 2001;11(4):175-185. doi.10.1177/112070000101100401 Crossref | Google Scholar
- Lucchesi G, Sacco R, Zhou W, Li Y, Li L, Canavese F. DDH in the Walking Age: Review of Patients with Long-Term Follow-Up. Indian J Orthop. 2021;55(6):1503-1514. doi:10.1007/s43465-021-00531-1 PubMed | Crossref | Google Scholar
- Woodacre T, Dhadwal A, Ball T, Edwards C, Cox PJA. The costs of late detection of developmental dysplasia of the hip. J Child Orthop. 2014;8(4):325-332. doi.10.1007/s11832-014-0599-7 PubMed | Crossref | Google Scholar
- Anuar R, Mohd-Hisyamudin HP, Ahmad MH, Zulkiflee O. The Economic Impact of Managing Late Presentation of Developmental Dysplasia of Hip (DDH). Malays Orthop J. 2015;9(3):40-43. doi:10.5704/MOJ.1511.006 PubMed | Crossref | Google Scholar
- Broadhurst C, Rhodes AML, Harper P, Perry DC, Clarke NMP, Aarvold A. What is the incidence of late detection of developmental dysplasia of the hip in England?: a 26-year national study of children diagnosed after the age of one. Bone Joint J. 2019;101-B(3):281-287. doi:10.1302/0301-620X.101B3.BJJ-2018-1331.R1 PubMed | Crossref | Google Scholar
- Studer K, Williams N, Antoniou G, et al. Increase in late diagnosed developmental dysplasia of the hip in South Australia: risk factors, proposed solutions. Med J Aust. 2016;204(6):240. doi:10.5694/mja15.01082 PubMed | Crossref | Google Scholar
- Schaeffer EK, Study Group I, Mulpuri K. Developmental dysplasia of the hip: addressing evidence gaps with a multicentre prospective international study. Med J Aust. 2018;208(8):359-364. doi:10.5694/mja18.00154 PubMed | Crossref | Google Scholar
- Sioutis S, Kolovos S, Papakonstantinou ME, Reppas L, Koulalis D, Mavrogenis AF. Developmental Dysplasia of the Hip: A Review. J Long Term Eff Med Implants. 2022;32(3):39-56. doi:10.1615/JLongTermEffMedImplants.2022040393 PubMed | Crossref | Google Scholar
- Shaw BA, Segal LS; SECTION ON ORTHOPAEDICS. Evaluation and Referral for Developmental Dysplasia of the Hip in Infants. Pediatrics. 2016;138(6):e20163107. doi:10.1542/peds.2016-3107 PubMed | Crossref | Google Scholar
- Fludder CJ, Keil BG, Neave MJ. Case report: Morphological changes evident after manual therapy in two cases of late-diagnosed developmental dysplasia of the hip. Front Pediatr. 2023;10:1045812. doi:10.3389/fped.2022.1045812 PubMed | Crossref | Google Scholar
- Government of South Australia. Neonatal Hip Screening and Management of Developmental Dysplasia of the Hip. Updated April 11, 2025. Neonatal Hip Screening and Management of Developmental Dysplasia of the Hip
- Safer Care Victoria. Developmental dysplasia of the hip in neonates. 2017. Developmental dysplasia of the hip in neonates
Acknowledgments
Not applicable
Funding
The work had no special funding.
Author Information
Corresponding Author:
Christian Fludder
Allied Health Practitioner
Private Practice, Brisbane, Australia
Email: christianfludder@gmail.com
Co-Authors:
Braden Keil
Allied Health Practitioner
Private Practice, Melbourne, Australia
Melissa Neave
Allied Health Practitioner
Private Practice, Sydney, Australia
Authors Contributions
All authors were involved in the synthesis and development of the manuscript. CF collated case data and prepared a case presentation. All authors contributed to writing the manuscript. All authors read and approved the final manuscript.
Informed Consent
Written informed consent to participate in this study was provided by the participants’ legal guardian/next of kin. Written informed consent was obtained from the minor(s)’ legal guardian/next of kin for the publication of any potentially identifiable images and data included in this article.
Conflict of Interest Statement
The authors declare no conflicts of interest.
Guarantor
None
DOI
Cite this Article
Fludder C, Keil B, Neave M. Guideline discordance in the management of a toddler with developmental dysplasia of the hip. medtigo J Med. 2025;3(3):e3062334. doi:10.63096/medtigo3062334 Crossref









