Author Affiliations
Abstract
Aortic stenosis (AS) is a pathological condition in which the aortic valve, which is present between the left ventricle and aorta, becomes narrowed. Patients with AS had a high mortality rate due to left ventricular dysfunction, which leads to improper blood supply to the body and associated coronary artery disease. Echocardiography is the modality to evaluate cardiac disease. Patients are referred to further modalities for confirmation of disease or preoperative evaluation. The objective of this study is to investigate the feasibility of f coronary angiography (CTCA) in diagnosing left ventricular dysfunction in patients with AS. This cross-sectional descriptive study was conducted at the Punjab Institute of Cardiology, Lahore. Sample size of our research was 40. The participants ranged in age from 17 to 73 years (72.5% male), with most of the patients being between 21 and 40 years old. 12.5% of those aged over 65 while 87.5% were between 17 to 64 years affected with AS. 9(22.5%) patients had congenital bicuspid aortic valve causing AS while 31(77.5%) had trileaflet aortic valve which acquire AS. 13(32.5%) patients out of 40 had left ventricle ejection fraction (LVEF) < 50% out of which 6(46.2%) patients had dilated LV with moderate Left ventricle (LV) systolic dysfunction and 7(53.8) patients had dilated LV with severe LV systolic dysfunction were diagnosed on CTCA. CTCA is more feasible for early and accurate diagnosis of left ventricular dysfunction in AS patients. CTCA can improve the diagnostic potential and treatment plans for patients with AS and associated left ventricular dysfunction.
Keywords
Computed tomography coronary angiography (CTCA), Left ventricle (LV), Aortic stenosis (AS), Ejection Fraction (EF), Aortic valve.
Introduction
AS occurs when the aortic valve narrows, and blood cannot flow normally. Progressive valve narrowing and the ensuing left ventricular remodeling response are two hallmarks of AS [1]. The most prevalent kind of valvular heart disease is AS. Aortic sclerosis, a modest form of fibrocalcific leaflet alterations, is the first stage of aortic valve disease and is linked to a 50% higher risk of unfavorable cardiovascular events even in the presence of normal leaflet opening. Aortic sclerosis frequently advances to more severe leaflet calcification; the disease’s final stage is marked by blockage of left ventricular outflow, which makes it unable to raise cardiac output sufficiently during exertion. If LV outflow blockage is not relieved once even minor symptoms appear, heart failure and death result [2]. Once primarily caused by rheumatic fever, the most common pathogenesis today is an active inflammatory process with some features that are like atherosclerosis [3]. The lesion of AS begins with leaflet thickening and sclerosis with progression to hemodynamically significant narrowing, usually said to be present when the aortic valve area is less than 2 cm2 [4]. Degenerative illness is the most prevalent cause of AS, although AS can also be brought on by endocarditis, systemic inflammatory illness, congenital valve abnormalities, and numerous other conditions. Bicuspid aortic valve is thought to be a common congenital valve defect causing AS, and the prevalence of bicuspid aortic valve is about 0.5–1% in children [5].
In an unselected older population, hypertension, diabetes, and dyslipidemia all independently and dose responsively correlated with incident AS; taken together, they accounted for almost one-third of the risk of severe AS [6]. Both bicuspid and tricuspid aortic valves may acquire calcific AS because of abnormal aortic function [7]. Development of significant AS tends to occur earlier in persons with congenital bicuspid aortic valves as well as in people who have problems with the metabolism of calcium, including renal failure [8]. Calcific AS is the most prevalent heart valve disorder in developed countries. After systemic arterial hypertension and coronary artery disease, AS is the third most common cardiovascular disease in developed nations, with a frequency of 1.7% in people over 65 and 0.4% in the general population [9]. Many patients with a normal trileaflet aortic valve experience calcification; however, 60% of Aortic Valve Replacements in patients under the age of 70 and 40% in those over 70 have bileaflet architecture [2]. Aortic valve stenosis affects 3% of persons older than 65 years and is the most significant cardiac valve disease in developed countries. The patient is asymptomatic for many years and relatively safe in this latent period. The prognosis deteriorates and the death rate rises sharply at the onset of symptoms [10]. An estimated 4.2 to 5.6 million adults in the United States have some form of clinically important valve disease [11]. Asia is the largest continent in the world and consists of markedly heterogeneous ethnicities. With documented prevalence rates of 1.86 in China, 4.54 in India, and 1.3 in Bangladesh per 1,000 people, rheumatic AS is most prevalent in Asia [12]. The main method for diagnosing AS is transthoracic echocardiography [13]. Reduced ejection fraction is associated with an increased risk of perioperative mortality; however, patients without surgery have a poor prognosis, and valve replacement improves long-term outcomes. Ejection fraction reductions are directly related to afterload increases and will reverse after valve replacement [14].
CTCA is established for the assessment of severe AS considered for transcatheter aortic valve replacement [15]. Therapeutic management of AS is essentially determined by the severity of the stenosis, the patient’s symptomatic status, and the status of LV systolic function [16]. There is currently no effective medical treatment available to slow the course of AS or to enhance prognosis [17]. Surgical aortic valve replacement has been the gold standard treatment for decades. However, transcatheter aortic valve replacement has emerged as an attractive, less invasive option for appropriately selected patients [18]. Patients with severe AS who are at high surgical risk can be effectively treated with transcatheter aortic valve replacement. These patients are frequently old and affected by multiple cardiovascular comorbidities, such as hypertension, coronary artery disease, peripheral vascular disease, and diabetes mellitus. CTCA can be used to assess peripheral vascular access sites and coronary artery anatomy [19].
It is strongly advised to perform a coronary angiography to determine transcatheter aortic valve replacement eligibility [20]. Among asymptomatic patients with very severe AS, early surgical aortic valve replacement led to a considerably decreased risk of death from cardiovascular causes during the follow-up period compared with conservative therapy [21]. The aim of this study is to assess left ventricle dysfunction using CTCA in AS patients. After this study, we will seek to provide valuable insights into the potential role of CT as an adjunct or alternative imaging modality to echocardiography, thereby enhancing the diagnostic capabilities and management strategies for patients with AS and associated left ventricle dysfunction.
Methodology
A cross-sectional descriptive study was performed by using a non-probability convenient sampling technique in the Punjab Institute of Cardiology, Lahore, Pakistan. In this study, Patients diagnosed with AS on echocardiography were included. Patients with severe renal impairment contraindicating the use of the contrast agent, Uncooperative patients, and patients who declined to give consent were excluded. Study was performed on a CT machine model Toshiba Alexion of 640 slices, 5th generation. A complete analysis of the cross-sectional radiological examination of 40 patients was carried out for assessment of AS on CTCA. The data collected included age, gender, risk factors, and CTCA parameters. Data was evaluated and analyzed with statistical package for social sciences (SPSS) 25 and Microsoft excel of 2016. For continuous variables, the mean and standard deviation (SD) were computed.
Results
Participants in the study ranged in age from 17 to 73 (of which 72.5% were male), with the majority of patients being between 21 and 40 years old. 12.5% of those aged over 65, while 87.5% were between 17 to 64 years affected with AS. Patients of 9(22.5%) had congenital bicuspid aortic valve causing AS while 31(77.5%) had Trileaflet aortic valve which acquire AS. 13(32.5%) hypertension out of 40 had LVEF < 50% out of which 6(46.2%) patients had dilated LV with moderate LV systolic dysfunction and 7(53.8) patients had dilated LV with severe LV systolic dysfunction were diagnosed on CTCA.
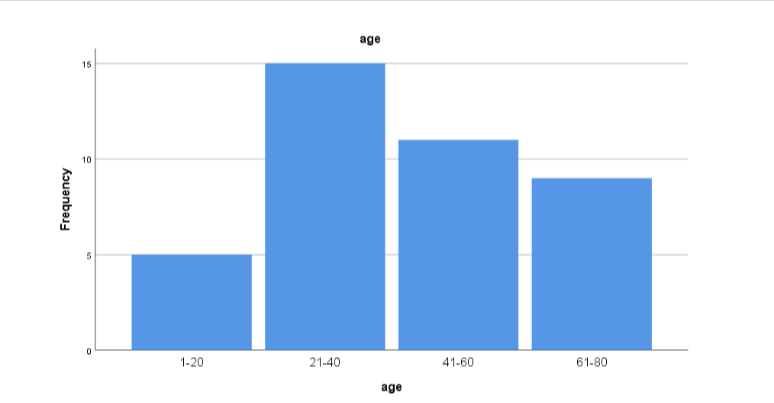
Figure 1: Age According to this, most patients were of the age between 21 to 40 years, while the least group of the age was 1-20 years
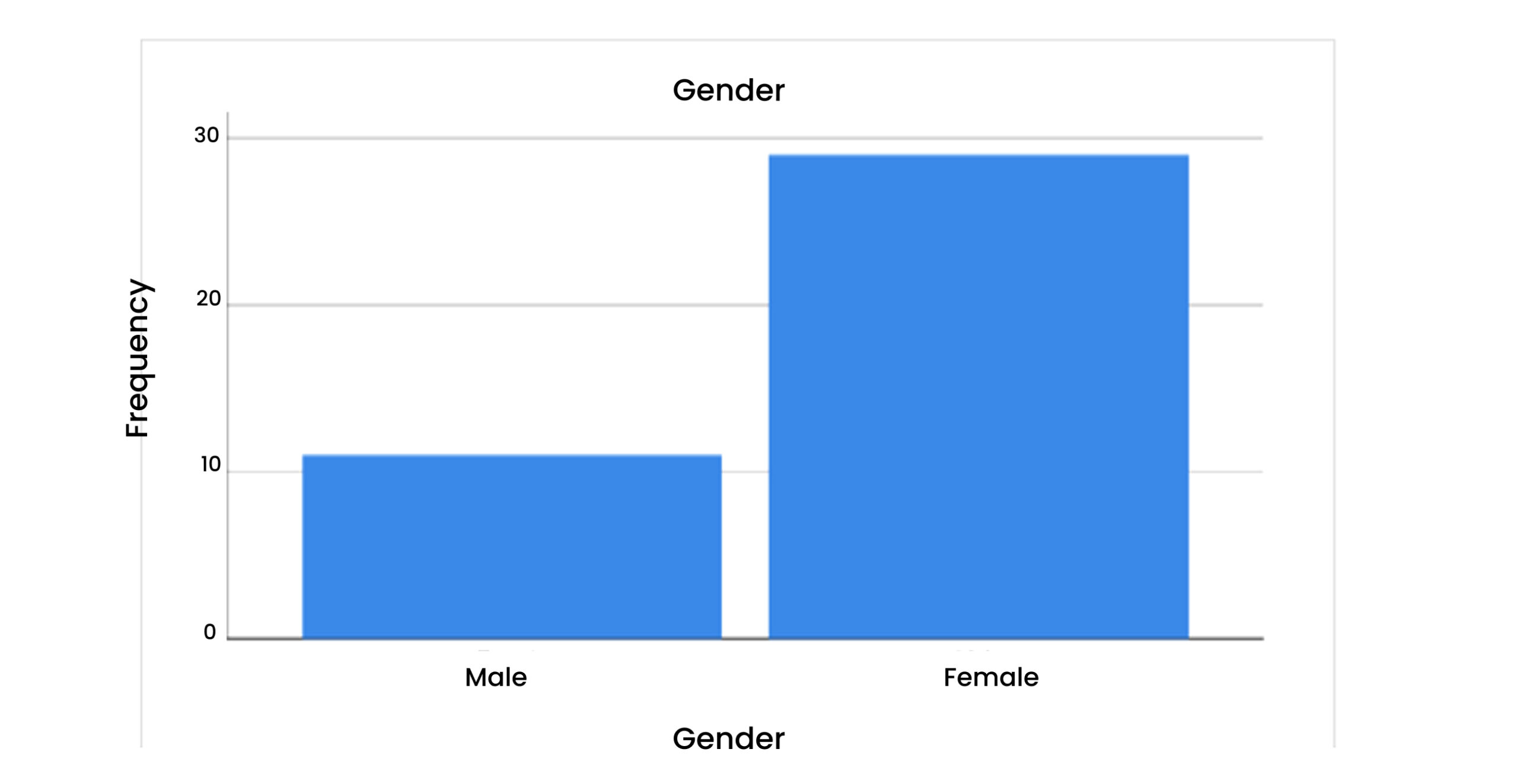
Figure 2: Gender
According to this, most of the patients were males, while females were less in number.
Table 1 presents a concise overview of the frequency of different health conditions, including hypertension, diabetes, obesity, hyperlipidemia, Familial hypercholesterolemia (FH), and smoking, among the participants. The mean values represent the frequency of each condition, while the SD measures the extent of variability in the data.
| N | Minimum | Maximum | Mean | SD | |
| Hypertension | 40 | 0 | 1 | 0.28 | 0.452 |
| Diabetes | 40 | 0 | 1 | 0.10 | 0.304 |
| Obesity | 40 | 0 | 1 | 0.08 | 0.267 |
| Hyperlipidemia | 40 | 0 | 1 | 0.10 | 0.304 |
| FH | 40 | 0 | 1 | 0.10 | 0.304 |
| Smoke | 40 | 0 | 1 | 0.15 | 0.362 |
Table 1: Descriptive Statistics
Table 2 & Figure 3: Hypertension
According to this, 72% of patients with hypertension were absent. While in 27% it was present.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 29 | 72.5 | 72.5 |
| Present | 11 | 27.5 | 27.5 |
| Total | 40 | 100.0 | 100.0 |
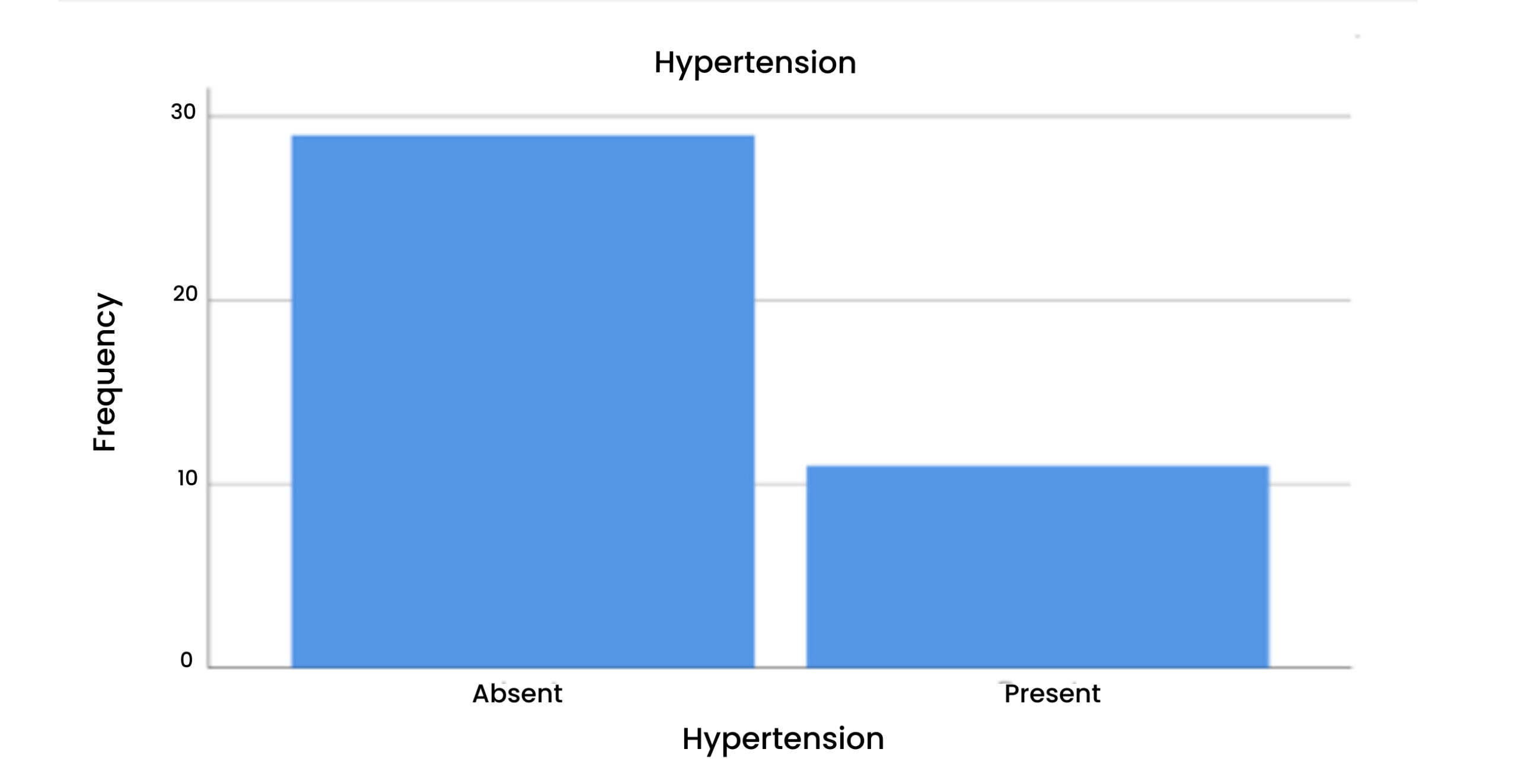
Figure 3: Hypertension
Table 3 & Figure 4: Diabetes
According to this, 90% of patients with diabetes were absent. While in 10% it was present.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 36 | 90.0 | 90.0 |
| Present | 4 | 10.0 | 10.0 |
| Total | 40 | 100.0 | 100.0 |
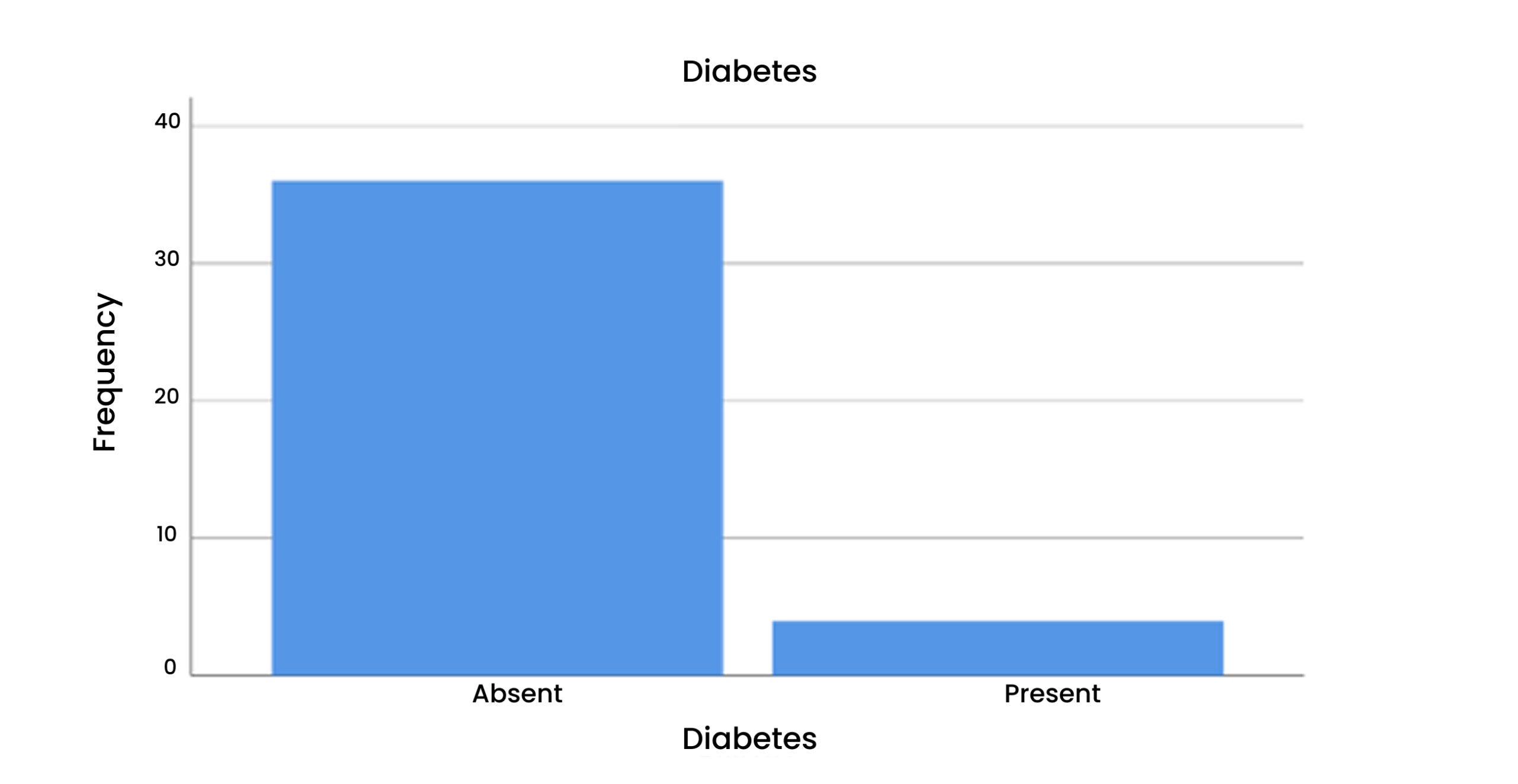
Figure 4: Diabetes
Table 4 & Figure 5: Hyperlipidemia
According to this, 90% of the patients hyperlipidemia was absent. While in 10% it was absent.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 36 | 90.0 | 90.0 |
| Present | 4 | 10.0 | 10.0 |
| Total | 40 | 100.0 | 100.0 |
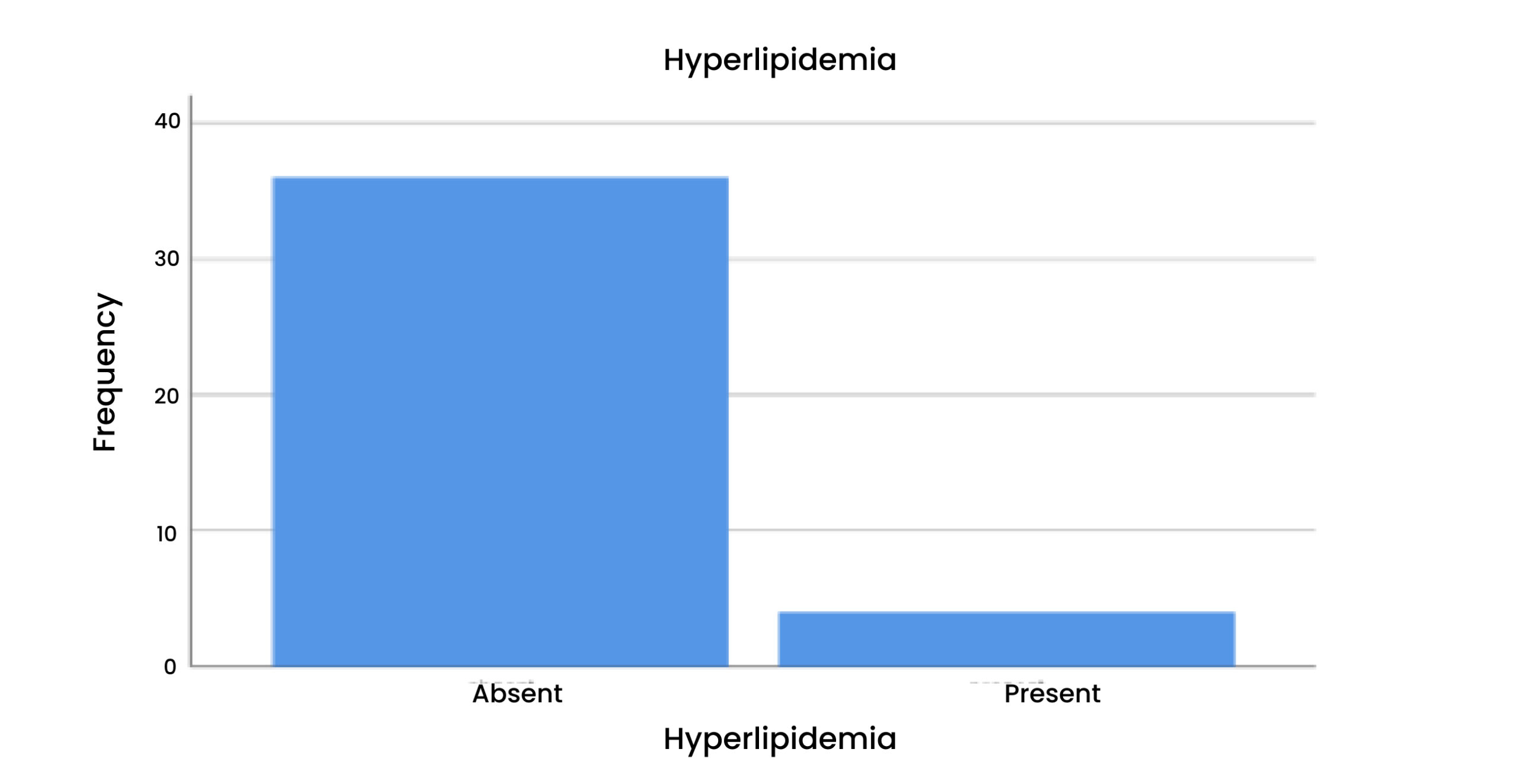
Figure 5: Hyperlipidemia
Table 5 & Figure 6: FH
According to this, 90% of patients FH were absent. While in 10% it was present.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 36 | 90.0 | 90.0 |
| Present | 4 | 10.0 | 10.0 |
| Total | 40 | 100.0 | 100.0 |
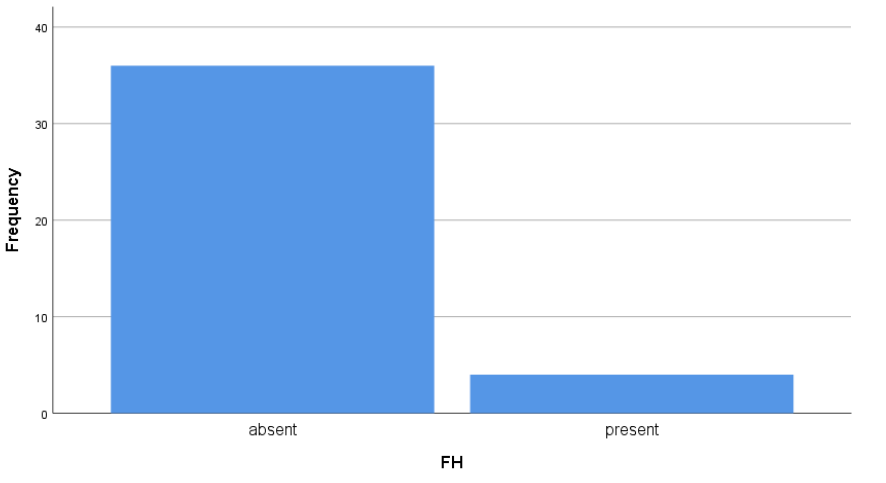
Figure 6: FH
Table 6 & Figure 7: Obesity
According to this, 92% of patients with obesity were absent. While in 7% it was present.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 37 | 92.5 | 92.5 |
| Present | 3 | 7.5 | 7.5 |
| Total | 40 | 100.0 | 100.0 |
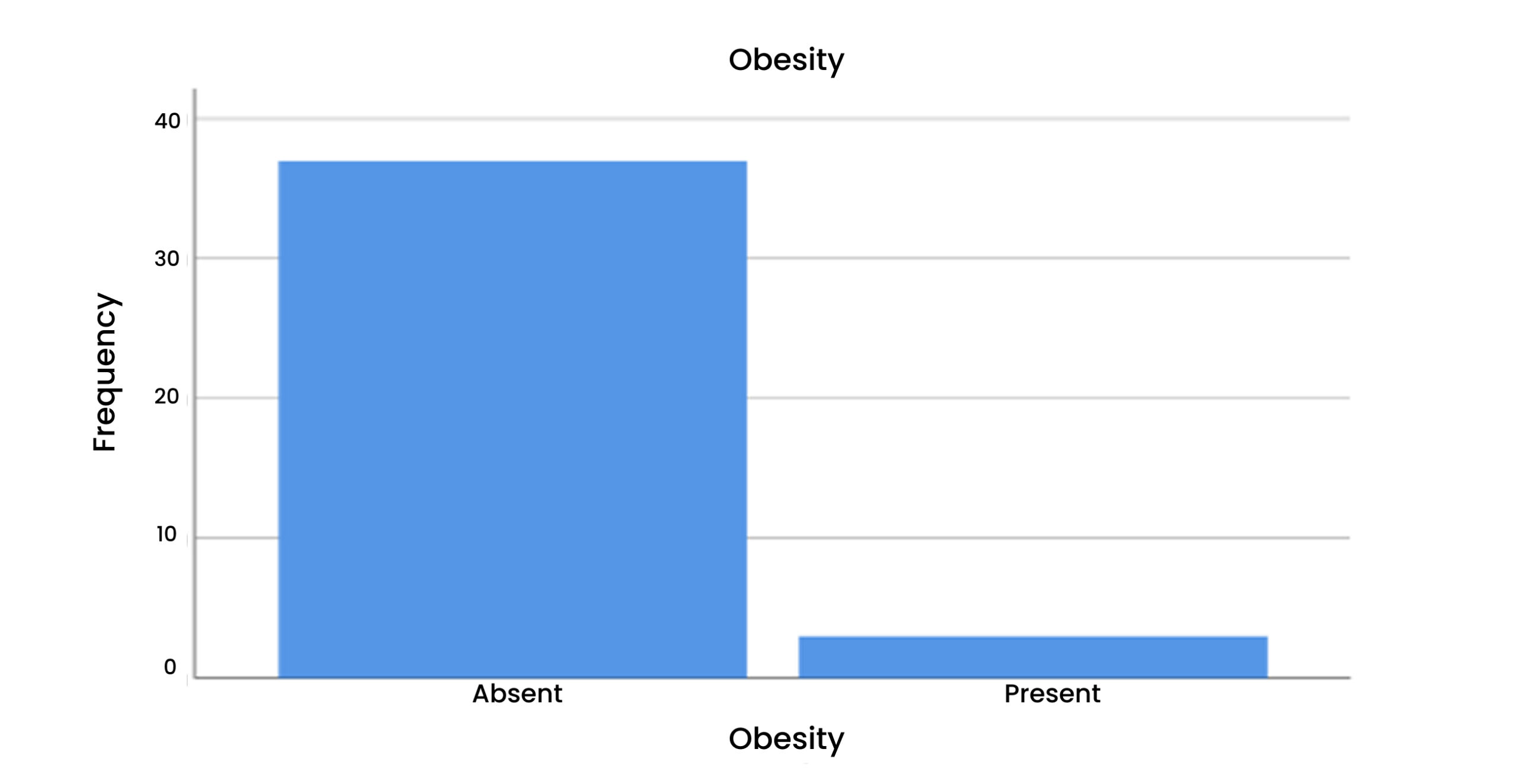
Figure 7: Obesity
Table 7 & Figure 8: Smoke
According to this, 85% of the smoke was absent. While in 15% it was present.
| Valid | Frequency | Percent | Valid Percent |
| Absent | 34 | 85.0 | 85.0 |
| Present | 6 | 15.0 | 15.0 |
| Total | 40 | 100.0 | 100.0 |
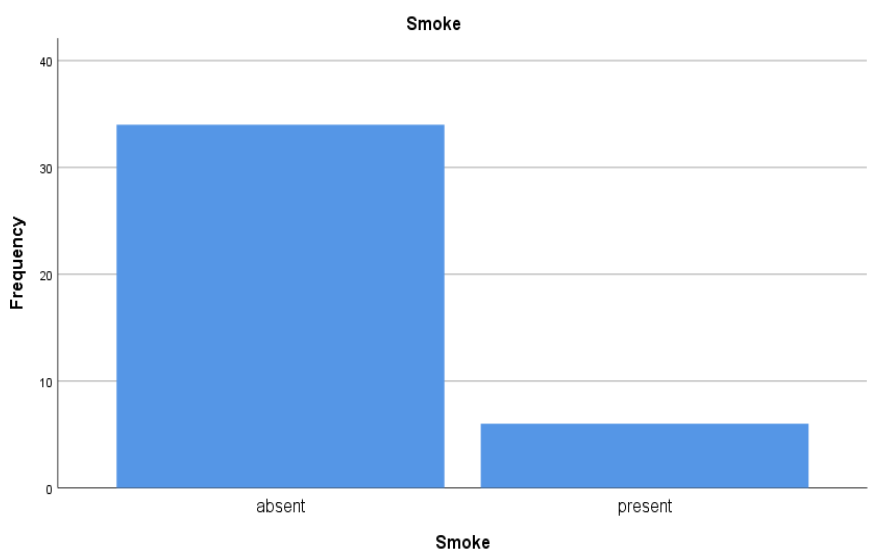
Figure 8: Smoke
Table 8 & Figure 9: End-diastolic volume (EDV)
According to this, a maximum of 42.5% of the patients were between 100- 200ml. A minimum of 5% of the patients with EDV presented with 300- 500 mL.
| Valid | Frequency | Percent | Valid Percent |
| 0-100ml | 3 | 7.5 | 7.5 |
| 101-200ml | 17 | 42.5 | 42.5 |
| 201-300ml | 16 | 40.0 | 40.0 |
| 300-400ml | 2 | 5.0 | 5.0 |
| 401-500ml | 2 | 5.0 | 5.0 |
| Total | 40 | 100.0 | 100.0 |
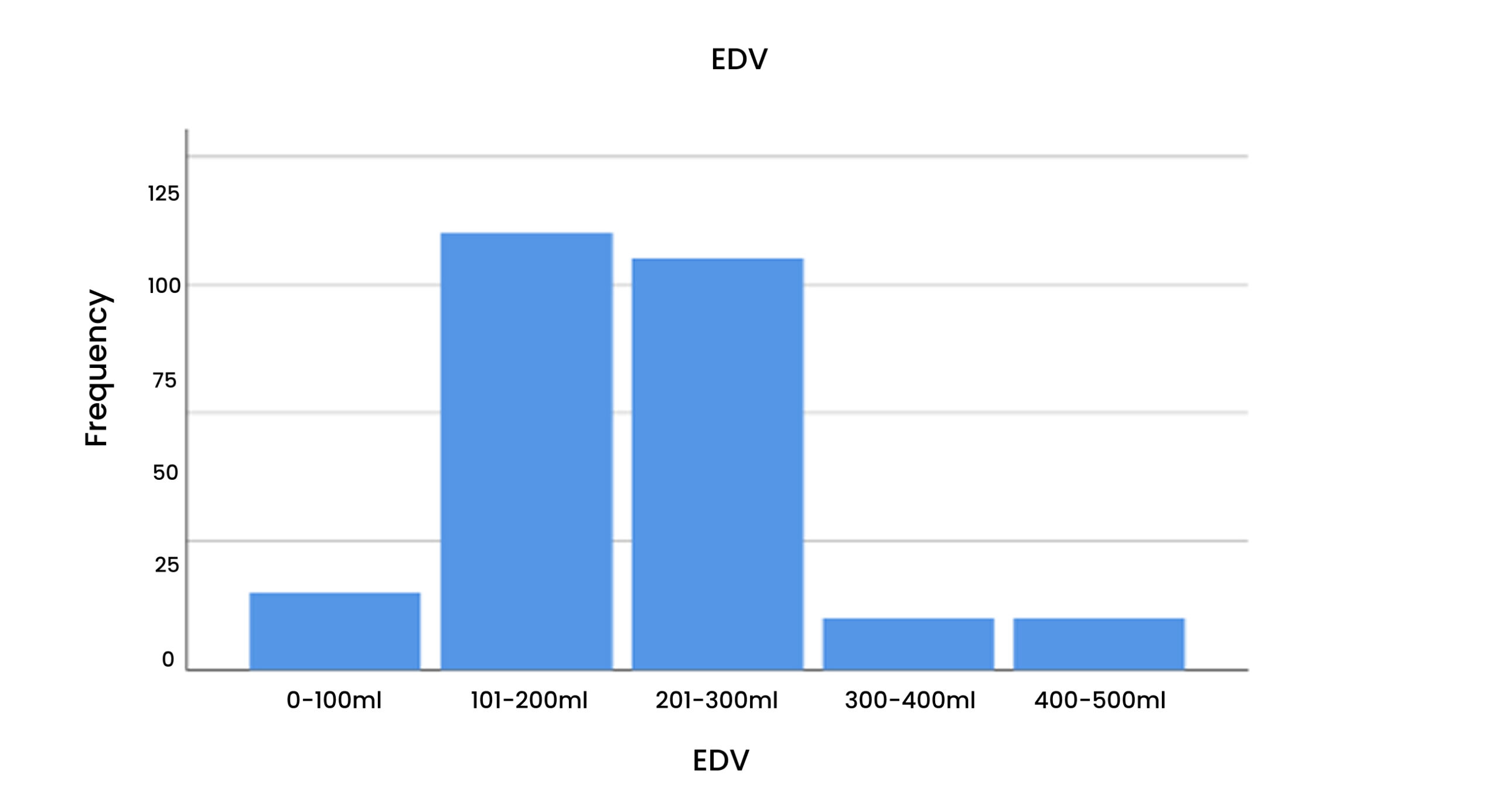
Figure 9: EDV
Table 9 & Figure 10: End-systolic volume (ESV)
According to this, a maximum of 35% of the patients were between 0- 50ml. A minimum of 2% of the patients with ESV presented in 200- 250ml.
| Valid | Frequency | Percent | Valid Percent |
| 0-50ml | 14 | 35.0 | 35.0 |
| 51-100ml | 8 | 20.0 | 20.0 |
| 101-150ml | 8 | 20.0 | 20.0 |
| 151-200ml | 9 | 22.5 | 22.5 |
| 201-150ml | 1 | 2.5 | 2.5 |
| Total | 40 | 100.0 | 100.0 |
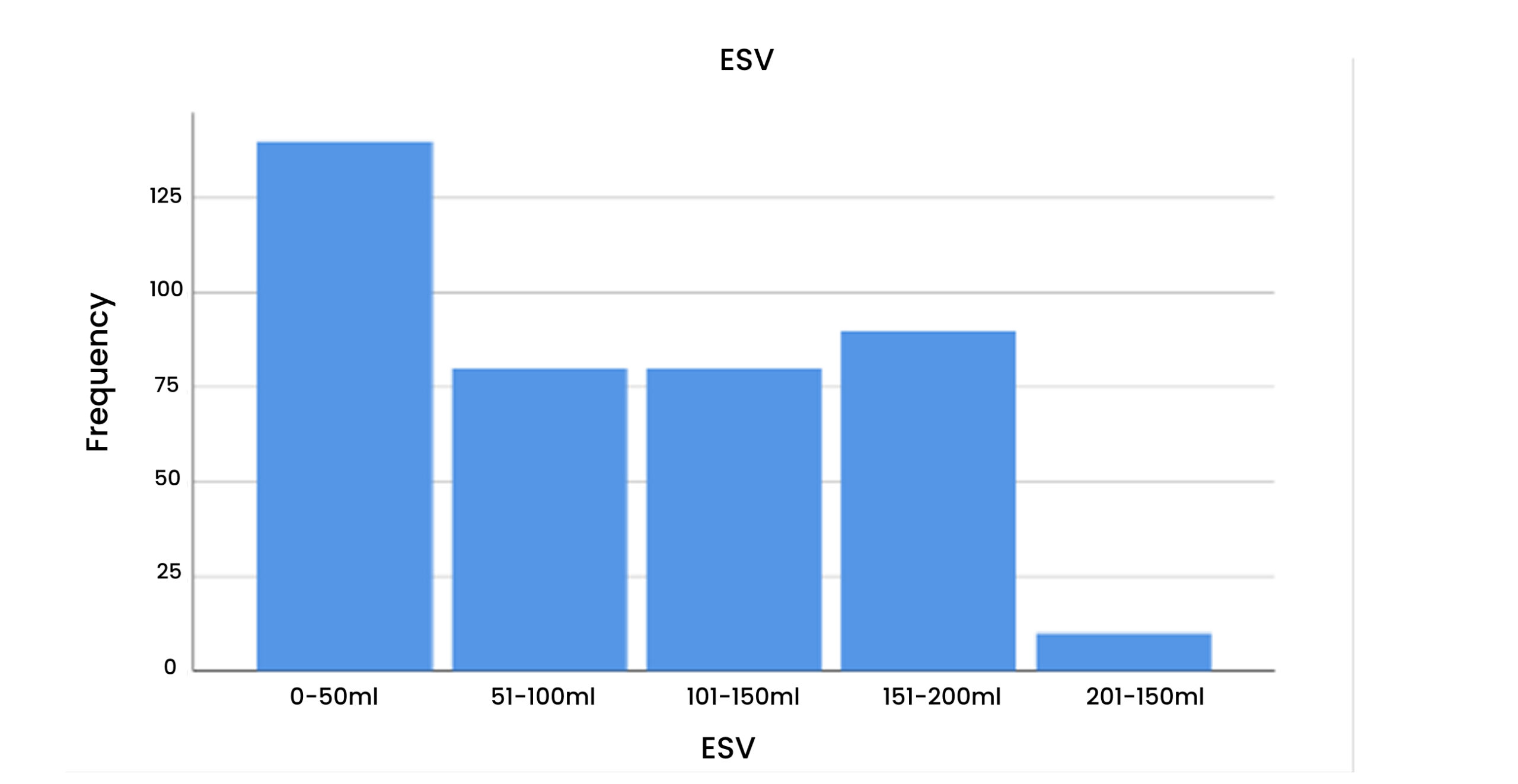
Figure 10: ESV
Table 10 & Figure 11: Aortic Valve
| Valid | Frequency | Percent | Valid Percent |
| Thickened Calcified Bicuspid | 6 | 15.0 | 15.0 |
| Thickened Calcified Trileaflet | 22 | 55.0 | 55.0 |
| Thickened Aortic Root | 8 | 20.0 | 20.0 |
| Heavily Calcified Bileaflet | 4 | 10.0 | 10.0 |
| Total | 40 | 100.0 | 100.0 |
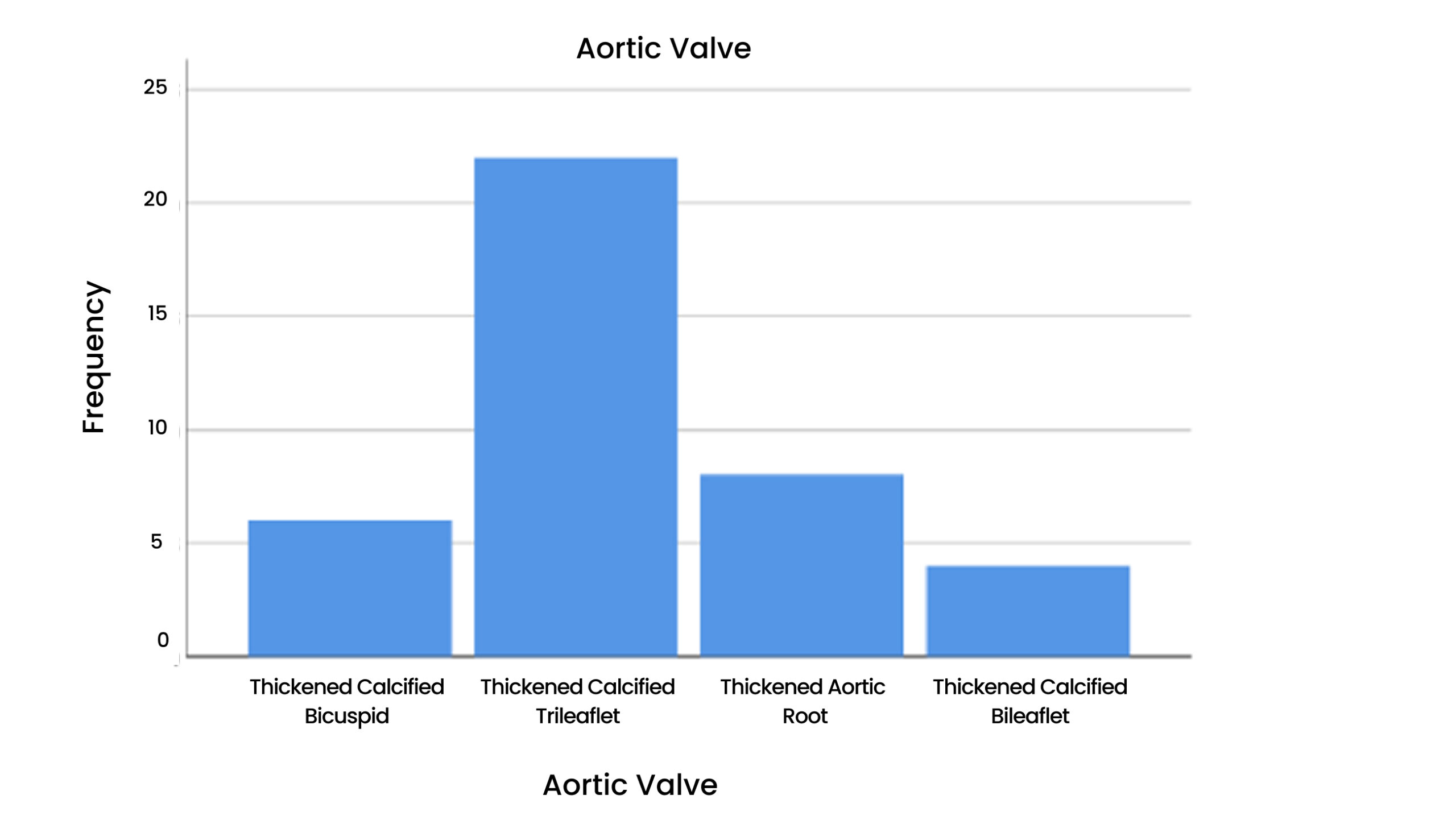
Figure 11: Aortic Valve
Table 11 & Figure 12: LV
| Valid | Frequency | Percent | Valid Percent |
| Good LV Function | 18 | 45.0 | 15.0 |
| Fair LV Function | 8 | 20.0 | 55.0 |
| Moderate LV Dysfunction | 7 | 17.5 | 20.0 |
| Severe LV Dysfunction | 7 | 17.5 | 10.0 |
| Total | 40 | 100.0 | 100.0 |
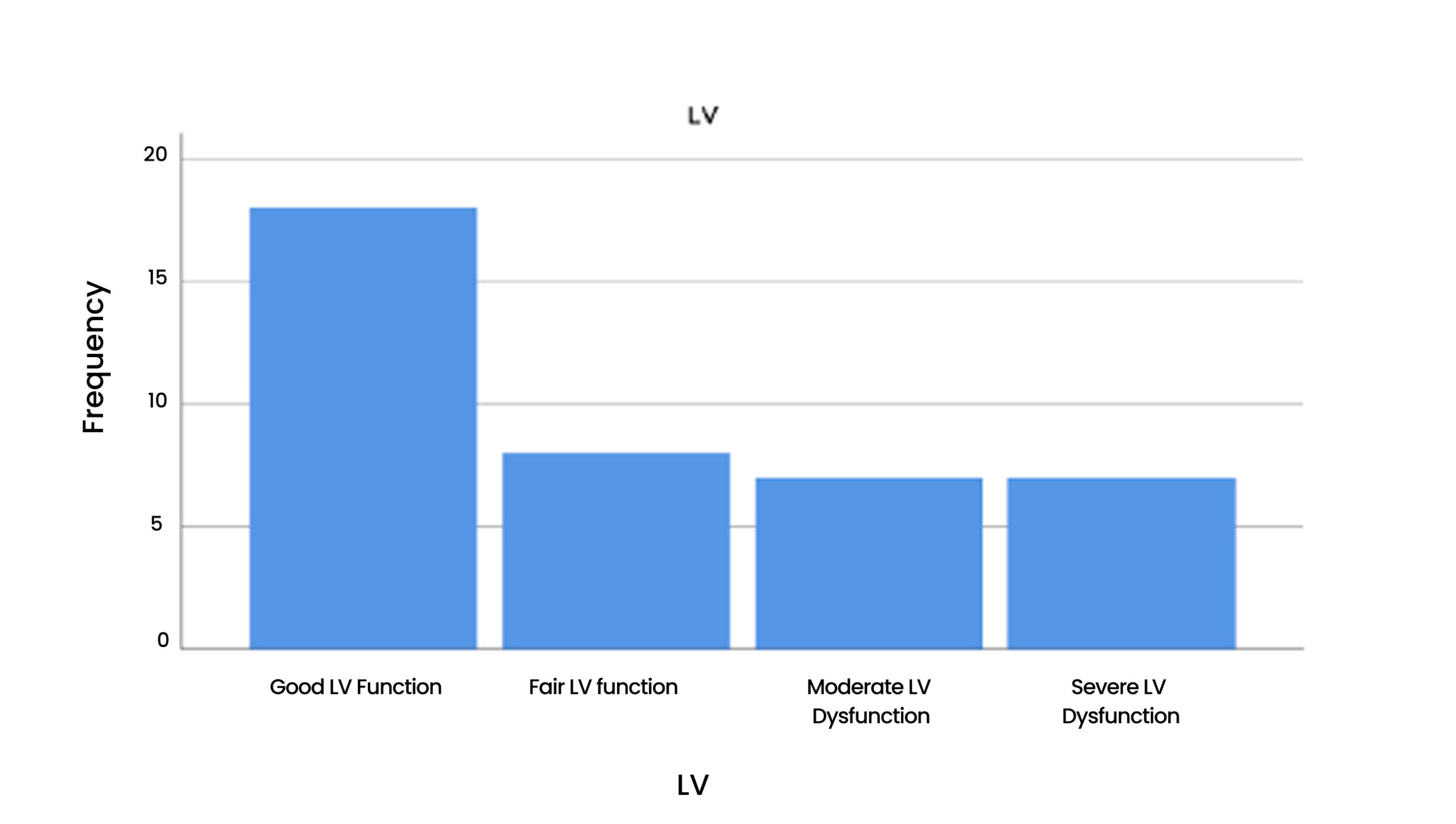
Figure 12: LV
Table 12 & Figure 13: Aortic Valve * LV Cross tabulation
| LV Aortic valve | Good LV Function | Fair LV Function | Moderate LV Dysfunction | Severe LV Dysfunction | Total |
| Thickened Calcified Bicuspid | 3 | 1 | 2 | 0 | 6 |
| Thickened Calcified Trileaflet | 9 | 6 | 5 | 2 | 22 |
| Thickened Aortic Root | 5 | 1 | 0 | 3 | 9 |
| Heavily Calcified Bileaflet | 1 | 0 | 0 | 2 | 3 |
| Total | 18 | 8 | 7 | 7 | 40 |
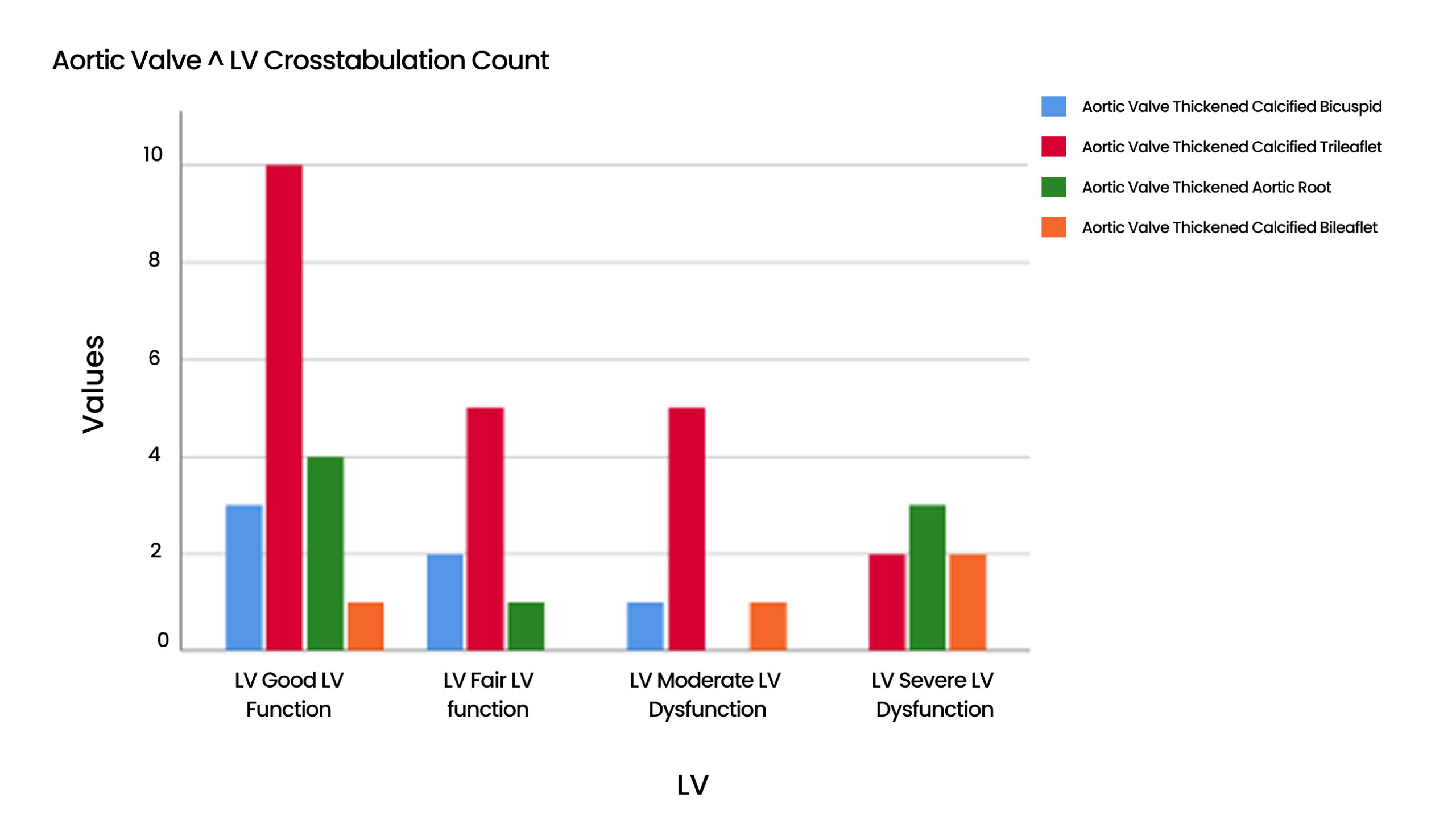
Figure 13: Aortic valve *LV Cross tabulation
Table 13: Aortic Valve * Hypertension Cross tabulation
| Hypertension Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 2 | 4 | 6 |
| Thickened Calcified Trileaflet | 17 | 5 | 22 |
| Thickened Aortic Root | 7 | 1 | 8 |
| Heavily Calcified Bileaflet | 3 | 1 | 4 |
| Total | 29 | 11 | 40 |
Table 14: Aortic Valve * Diabetes Cross tabulation
| Diabetes Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 6 | 0 | 6 |
| Thickened Calcified Trileaflet | 20 | 2 | 22 |
| Thickened Aortic Root | 7 | 1 | 8 |
| Heavily Calcified Bileaflet | 3 | 1 | 4 |
| Total | 36 | 4 | 40 |
Table 15: Aortic Valve * hyperlipidemia Cross tabulation
| Hyperlipidemia Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 5 | 1 | 6 |
| Thickened Calcified Trileaflet | 20 | 2 | 22 |
| Thickened Aortic Root | 7 | 1 | 8 |
| Heavily Calcified Bileaflet | 4 | 0 | 4 |
| Total | 36 | 4 | 40 |
Table 16: Aortic Valve * FH Cross tabulation
| FH Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 6 | 0 | 6 |
| Thickened Calcified Trileaflet | 20 | 2 | 22 |
| Thickened Aortic Root | 7 | 1 | 8 |
| Heavily Calcified Bileaflet | 3 | 1 | 4 |
| Total | 36 | 4 | 40 |
Table 17: Aortic Valve * Smoke Cross tabulation
| Smoke Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 5 | 1 | 6 |
| Thickened Calcified Trileaflet | 20 | 2 | 22 |
| Thickened Aortic Root | 7 | 1 | 8 |
| Heavily Calcified Bileaflet | 2 | 2 | 4 |
| Total | 34 | 6 | 40 |
Table 18: Aortic Valve * Obesity Cross tabulation
| Obesity Aortic valve | Absent | Present | Total |
| Thickened Calcified Bicuspid | 5 | 1 | 6 |
| Thickened Calcified Trileaflet | 21 | 1 | 22 |
| Thickened Aortic Root | 8 | 0 | 8 |
| Heavily Calcified Bileaflet | 3 | 1 | 4 |
| Total | 37 | 3 | 40 |
Discussion
The most prevalent valvular heart disease that results in death is AS. To evaluate the validity and viability of computed tomography-derived fractional flow reserve in patients with severe AS, Michail et al. 2021 carried out a prospective study. Patients received invasive FFR and CTCA according to normal protocol. To evaluate CT-FFR independently, CTCA pictures were evaluated. 39 patients (92.3%) out of 42 had interpretable CTCA, which allowed for the estimation of CT-FFR. 76.2±6.7 years was the mean age (71.8% male). With 76.7% diagnostic accuracy, the corresponding values for sensitivity, specificity, positive predictive value, and negative predictive value were 73.9%, 78.4%, 68.0%, and 82.9%, respectively. According to available data, CT-FFR’s diagnostic accuracy may make it possible for it to be used in clinical settings and lay the groundwork for further studies [22].
In 2017, a retrospective study led by Lennart van et al. aimed to ascertain the clinical outcome of individuals who had both simultaneous moderate AS and LV systolic dysfunction. The study included 305 patients, with a mean age of 73 ± 11 years, and 75% of them were male [23]. In our study total of 40 patients were studied with ages ranging between 17 to 73 years (72.5%male), most patients were of the age between 21 to 40 years. An ejection fraction determined from CTCA can be used to assess LV dysfunction in patients with AS. In conclusion, CTCA has improved the visualization of the aortic valve and left ventricle anatomy and pathology. A trustworthy and thorough explanation revealed by a CTCA scan serves as a pre-operative road map.
According to a 2014 study by Czarny MJ et al. 2% of people 65 years of age or older have valvular AS, a degenerative illness [24]. According to a Qais et al. 2023 study, AS, which affects 5% of adults over 65, is the most frequent valvular heart disease, with a 1-3% incidence of severe stenosis. Congenital or acquired AS is both possible; 9(22.5%) patients had congenital bicuspid aortic valve causing AS, while 31(77.5%) had trileaflet aortic valve, which acquired AS [10].
A 2019 study by JS Dahl et al. found that one unfavorable effect of severe AS’s pressure overload is left ventricular systolic failure. A LVEF cut point of less than 50% has been applied in the case of the asymptomatic patient [25]. Retrospective research by JS Dahl et al. in 2015 involved asymptomatic individuals with severe AS; guidelines suggest that LVEF of less than 50% be used as the cutoff point for aortic valve replacement referrals because it is linked to death [26]. In our study, 13(32.5%) patients out of 40 had LVEF < 50% out of which 6(46.2%) patients had dilated LV with moderate LV systolic dysfunction, and 7(53.8) patients had dilated LV with severe LV systolic dysfunction. When treating severe AS, early and accurate detection of LV dysfunction aids in the best possible time of intervention.
Conclusion
CTCA is more feasible for early and accurate diagnosis of left ventricular dysfunction in AS patients. CTCA can improve the diagnostic potential and treatment plans for patients with AS and concomitant left ventricular dysfunction.
References
- Bing R, Cavalcante JL, Everett RJ, Clavel MA, Newby DE, Dweck MR. Imaging and impact of myocardial fibrosis in AS. JACC Cardiovasc Imaging. 2019;12(2):283-296. PubMed | Crossref | Google Scholar
- Rashedi N, Otto CM. AS: Changing disease concepts. J Cardiovasc Ultrasound. 2015;23(2):59. PubMed | Crossref | Google Scholar
- Carabello BA. Introduction to AS. Circ Res. 2013;113(2):179-185. PubMed | Crossref | Google Scholar
- Thaden JJ, Nkomo VT, Enriquez-Sarano M. The global burden of AS. Prog Cardiovasc Dis. 2014;56(6):565-571. PubMed | Crossref | Google Scholar
- Akahori H, Tsujino T, Masuyama T, Ishihara M. Mechanisms of AS. J Cardiol. 2018;71(3):215-220. PubMed | Crossref | Google Scholar
- Yan AT, Koh M, Chan KK, et al. Association between cardiovascular risk factors and AS: The CANHEART AS study. J Am Coll Cardiol. 2017;69(12):1523-1532. PubMed | Crossref | Google Scholar
- Boudoulas KD, Triposkiadis F, Boudoulas H. The AS complex. 2018;140(3):194-198. PubMed | Crossref | Google Scholar
- Grimard BH, Safford RE, Burns EL. AS: Diagnosis and treatment. Am Fam Physician. 2016;93(5):371-378. Aortic Stenosis: Diagnosis and Treatment | AAFP
- Lindman BR, Clavel MA, Mathieu P, et al. Calcific AS. Nat Rev Dis Primers. 2016;2(1):1-28.
PubMed | Crossref | Google Scholar - Qais Z, Qais K, Dhillon S. AS. In: Clinical and Surgical Aspects of Congenital Heart Diseases: Text and Study Guide. Springer International Publishing; 2023:105-112. Crossref
- Kanwar A, Thaden JJ, Nkomo VT. Management of patients with aortic valve stenosis. Mayo Clin Proc. 2018;93(4):488-508. PubMed | Crossref | Google Scholar
- Tay EL, Ngiam JN, Kong WK, Poh KK. Management of severe AS: The Singapore and Asian perspective. Singapore Med J. 2018;59(9):452. PubMed | Crossref | Google Scholar
- Boskovski MT, Gleason TG. Current therapeutic options in AS. Circ Res. 2021;128(9):1398-1417. PubMed | Crossref | Google Scholar
- Everett RJ, Clavel MA, Pibarot P, Dweck MR. Timing of intervention in AS: A review of current and future strategies. Heart. 2018;104(24):2067-2076. PubMed | Crossref | Google Scholar
- Rossi A, De Cecco CN, Kennon SR, et al. CT angiography to evaluate coronary artery disease and revascularization requirement before trans-catheter aortic valve replacement. J Cardiovasc Comput Tomogr. 2017;11(5):338-346. PubMed | Crossref | Google Scholar
- Clavel MA, Magne J, Pibarot P. Low-gradient AS. Eur Heart J. 2016;37(34):2645-2657.
PubMed | Crossref | Google Scholar - Marquis-Gravel G, Redfors B, Leon MB, Genereux P. Medical treatment of AS. 2016;134(22):1766-1784. PubMed | Crossref | Google Scholar
- Joseph J, Naqvi SY, Giri J, Goldberg S. AS: Pathophysiology, diagnosis, and therapy. Am J Med. 2017;130(3):253-263. PubMed | Crossref | Google Scholar
- Chieffo A, Giustino G, Spagnolo PF, et al. Routine screening of coronary artery disease with computed tomographic coronary angiography in place of invasive coronary angiography in patients undergoing transcatheter aortic valve replacement. Circ Cardiovasc Interv. 2015;8(7):e002025. PubMed | Crossref | Google Scholar
- Paradis JM, Fried J, Nazif T, et al. AS and coronary artery disease: What do we know? What don’t we know? A comprehensive review of the literature with proposed treatment algorithms. Eur Heart J. 2014;35(31):2069-2082. PubMed | Crossref | Google Scholar
- Kang DH, Park SJ, Lee SA, et al. Early surgery or conservative care for asymptomatic AS. N Engl J Med. 2020;382(2):111-119. PubMed | Crossref | Google Scholar
- Michail M, Ihdayhid AR, Comella A, et al. Feasibility and validity of computed tomography-derived fractional flow reserve in patients with severe AS: The CAST-FFR study. Circ Cardiovasc Interv. 2021;14(1):e009586. PubMed | Crossref | Google Scholar
- van Gils L, Clavel MA, Vollema EM, et al. Prognostic implications of moderate AS in patients with left ventricular systolic dysfunction. J Am Coll Cardiol. 2017;69(19):2383-2392. PubMed | Crossref | Google Scholar
- Czarny MJ, Resar JR. Diagnosis and management of valvular AS. Clin Med Insights Cardiol. 2014;8:CMC-S15716. PubMed | Crossref
- Dahl JS, Magne J, Pellikka PA, Donal E, Marwick TH. Assessment of subclinical left ventricular dysfunction in AS. JACC Cardiovasc Imaging. 2019;12(1):163-171. PubMed | Crossref | Google Scholar
- Dahl JS, Eleid MF, Michelena HI, et al. Effect of left ventricular ejection fraction on postoperative outcome in patients with severe AS undergoing aortic valve replacement. Circ Cardiovasc Imaging. 2015;8(4):e002917. PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
Not reported
Author Information
Corresponding Author:
Mehreen Anwar
Department of Radiological Sciences and Medical Imaging Technology (MSMIT)
Bachelor of Science in Medical Imaging Technology (BSMIT), Superior University Lahore, Pakistan
Email: mehrinanwar27@hotmail.com
Co-Author:
Uswa Shabbir, Jahanzaib, Abdul Qadir Khan, Rashid Majeed, Marwa Nawaz
Department of MSMIT, BSMIT, Superior University Lahore, Pakistan
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation and writing – review & editing to refine the manuscript. Additionally, all authors participated in the Supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Informed Consent
Not applicable
Conflict of Interest Statement
Not reported
Guarantor
None
DOI
Cite this Article
Mehreen Anwar, Jahanzaib, Uswa S, Abdul Qadir K, Rashid M, Marwa N. Feasibility of Computed Tomography Coronary Angiography in Diagnosing Left Ventricle Dysfunction in Patients with Aortic Stenosis. medtigo J Med. 2024;2(3):e3062241. doi:10.63096/medtigo3062241 Crossref









