Author Affiliations
Abstract
Introduction: Gastrectomy, whether for bariatric weight loss or gastric cancer resection, demands precise anesthetic management due to the complexity of the procedure and patient physiology. With rising global obesity and persistent gastric malignancy rates, optimizing perioperative care through advanced anesthetic strategies is critical. Traditional opioid-based anesthesia is associated with delayed recovery and adverse effects, particularly in obese individuals. Consequently, interest has grown in opioid-free anesthesia (OFA), multimodal analgesia (MMA), and balanced techniques that minimize opioid use while enhancing recovery.
Methodology: A systematic literature review was conducted using PubMed and Google Scholar databases per preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines. Clinical trials and observational studies from January 2015 to June 2025 involving human subjects undergoing gastrectomy were included. Studies were screened and selected based on inclusion/exclusion criteria and methodological quality using a scale for the assessment of narrative review articles (SANRA) scores. Four high-quality studies were included for final analysis.
Results: OFA significantly improved early postoperative recovery (Quality of recovery-40 (QoR-40 scores)), reduced opioid consumption, and enabled earlier oral intake and discharge readiness. Balanced anesthesia using propofol-etomidate offered better hemodynamic stability and faster cognitive recovery than individual agents. Sevoflurane-based anesthesia reduced neuromuscular blocker requirements and post-anesthesia care unit (PACU) stay duration. Gabapentin premedication delayed opioid initiation and reduced 12-hour oxycodone use without increasing adverse effects.
Conclusion: Opioid-sparing strategies like OFA, balanced anesthesia, and gabapentin premedication enhance perioperative outcomes in gastrectomy. These approaches support individualized anesthetic protocols to optimize recovery and minimize complications, though larger trials are needed to confirm efficacy and guide standardization.
Keywords
Opioid-free anesthesia, Laparoscopic sleeve gastrectomy, Multimodal analgesia, Gabapentin, Hemodynamic stability, Postoperative recovery.
Introduction
Gastrectomy, whether performed as a bariatric procedure or a radical intervention for gastric cancer, represents a major surgical challenge that demands carefully coordinated anesthetic management. With the increasing global prevalence of obesity and the continued burden of gastric malignancies, the role of anesthesia in optimizing surgical outcomes during gastrectomy has gained significant attention.[1] While surgical techniques have rapidly evolved, especially with the widespread adoption of laparoscopic approaches, effective perioperative anesthetic strategies remain critical to ensure patient safety, enhance recovery, and minimize postoperative complications.[2]
In the context of morbid obesity, laparoscopic sleeve gastrectomy (LSG) has become the most performed bariatric surgery due to its simplicity, efficacy, and relatively lower complication profile.[3] LSG not only achieves substantial and sustained weight loss but also leads to improvement or resolution of obesity-related comorbidities.[4] Despite these advantages, hesitancy around undergoing bariatric surgery persists among patients due to concerns about postoperative pain, nausea, prolonged recovery time, and the potential need for opioids, all of which are intricately linked to anesthesia.[5] As such, anesthesia protocols that can facilitate early recovery and reduce opioid-related side effects are increasingly emphasized, particularly under enhanced recovery after surgery (ERAS) programs.[6]
ERAS protocols aim to improve perioperative outcomes and patient satisfaction by implementing multimodal strategies that include preoperative education, optimized anesthesia, pain control, and early mobilization.[7] Among the core components of ERAS, anesthetic management has a direct impact on the speed and quality of recovery. Traditional anesthetic approaches often rely heavily on opioids, which are known to cause nausea, vomiting, respiratory depression, ileus, and delayed extubation, which are particularly detrimental in the obese population.[8] Given these concerns, there is growing interest in OFA, a technique that limits or completely eliminates perioperative opioid use in favor of non-opioid analgesics and regional anesthesia.[9]
OFA regimens commonly incorporate agents such as dexmedetomidine, lidocaine, ketamine, and magnesium sulfate.[10] Dexmedetomidine, an α2 adrenergic receptor agonist, offers sedative, analgesic, and sympatholytic effects without significant respiratory depression, making it highly suitable for obese patients undergoing gastrectomy.[11] Similarly, intravenous lidocaine has shown analgesic and anti-inflammatory effects, while ketamine provides N-methyl-d-aspartate(NMDA)-receptor-mediated analgesia with opioid-sparing potential.[12] When used in combination, these agents can reduce intraoperative hemodynamic fluctuations and postoperative pain scores, accelerate ambulation, and shorten the time to oral intake.[13]
Despite the theoretical advantages of OFA, existing literature presents mixed findings. Several reviews have attempted to compare OFA to conventional opioid-based anesthesia in bariatric populations, yet inconsistencies in study design, drug combinations, and outcome measures have limited the strength of their conclusions.[14] A recent randomized controlled trial sought to address this gap by evaluating the quality of early postoperative recovery between OFA and standard multimodal analgesia in patients undergoing laparoscopic sleeve gastrectomy under ERAS protocols.[15] The study used the validated quality of recovery-40 (QoR-40) questionnaire and found that OFA was associated with superior recovery scores at both 6 and 24 hours postoperatively. Secondary outcomes such as earlier ambulation, reduced opioid consumption, and faster readiness for discharge further underscored the benefits of opioid avoidance in this setting.[16]
Beyond bariatric surgery, radical gastrectomy for gastric cancer remains a complex, high-risk procedure frequently requiring prolonged operative time, significant hemodynamic management, and robust analgesia.[17] In China, where gastric cancer incidence is among the highest globally, the optimization of anesthetic techniques during gastrectomy has emerged as a priority.[18] Common anesthetic agents such as propofol and etomidate are widely used, each with distinct pharmacological profiles. Propofol offers rapid onset and recovery but can suppress cardiovascular and respiratory function at high doses. Etomidate, by contrast, preserves hemodynamic stability but may lead to myoclonus, nausea, and suppression of adrenal function if used continuously.[19]
Recent innovations in anesthetic drug formulation have introduced mixtures such as etomidate + propofol (Etofol), a 1:1 combination of propofol and etomidate, which aim to balance the advantages of both agents while minimizing their individual side effects. Studies suggest that such combinations can reduce intraoperative muscle tremors and postoperative nausea while ensuring adequate sedation and faster awakening.[20] These effects are particularly valuable in radical gastrectomy, where intraoperative stress responses and postoperative complications can compromise outcomes.
To further enhance the anesthetic depth and surgical field exposure during laparoscopic gastrectomy, particularly in obese patients, deep neuromuscular blockade (dNMB) has become a valuable tool.[21] Achieved through agents like rocuronium, dNMB helps relax abdominal muscles and allows for lower pneumoperitoneum pressures, reducing tissue trauma and the likelihood of postoperative complications. However, deep blockade must be carefully managed to avoid residual neuromuscular weakness, which can prolong recovery and increase the risk of respiratory events.[22] Inhalational agents like sevoflurane, when combined with intravenous anesthesia, have been shown to enhance muscle relaxation, reduce the total rocuronium dose needed, and facilitate quicker postoperative extubation and recovery in bariatric patients.[23]
Pharmacokinetic and pharmacodynamic alterations in obese individuals further complicate anesthetic decision-making. Increased fat mass altered regional blood flow, and changes in respiratory mechanics influence drug distribution, metabolism, and elimination. These changes necessitate precise dosing, vigilant monitoring, and tailored regimens to optimize outcomes.[24] The use of balanced anesthesia combining inhalational and intravenous agents can provide the required depth of anesthesia while mitigating the risks of overdose or delayed emergence.
Finally, managing postoperative pain remains one of the central concerns in anesthesia for gastrectomy. Multimodal analgesia, incorporating both opioid and non-opioid agents, has been widely adopted to control pain while minimizing opioid-related side effects. Gabapentin, an antiepileptic drug with analgesic properties, has emerged as a promising agent for pre-emptive analgesia in bariatric surgery. A randomized trial evaluating the preoperative use of 1200 mg gabapentin showed a significant delay in postoperative opioid requirement and reduced 12-hour oxycodone consumption compared to placebo, without increasing the incidence of nausea or delaying discharge. However, despite its favorable profile, the routine use of gabapentin remains debated due to mixed findings in meta-analyses and variable clinical protocols.[25]
In summary, anesthesia for gastrectomy, whether for bariatric or oncologic purposes, requires a nuanced, individualized approach. Innovations such as opioid-free anesthesia, deep neuromuscular blockade, novel anesthetic combinations, and preemptive analgesics like gabapentin are shaping the future of perioperative care. As surgical techniques continue to advance, anesthetic strategies must evolve in parallel to ensure optimal recovery, reduce complications, and improve patient satisfaction.[26]
This study aims to systematically evaluate and compare the effects of various anesthetic strategies, specifically OFA, MMA, and balanced anesthesia, on postoperative outcomes in patients undergoing gastrectomy. By analyzing clinical trials and observational studies from 2015 to 2025, the review seeks to assess pain control, opioid consumption, recovery profiles, and adverse events associated with these approaches. The objective is to identify optimal, evidence-based anesthetic protocols that enhance perioperative care and recovery in bariatric and oncologic gastrectomy patients.
Methodology
A systematic literature search was conducted using PubMed and Google Scholar databases according to PRISMA guidelines. The search included publications from January 1, 2015, to June 30, 2025, and was limited to studies published in English.
Inclusion criteria
- Clinical trials, observational studies, and randomized controlled trials
- Studies involving human subjects
- Articles published in English
- Studies including male and female participants
- Studies published between January 1, 2015, and June 30, 2025
Exclusion criteria
- Books, commentaries, editorials, letters, documents, and book chapters
- Case reports, case series, and literature reviews
- Articles published in languages other than English
- Animal studies and in vitro (laboratory) studies
- Articles published before January 1, 2015, or after June 30, 2025
- Studies lacking a reported results section
The study followed a systematic review methodology in accordance with the PRISMA guidelines. Initially, a total of 288 records were identified through database searches based on inclusion and exclusion criteria. Before the screening process began, 111 records were removed for various reasons, such as duplication or irrelevance, leaving 177 records for screening. These records were screened based on titles and abstracts, resulting in the exclusion of 133 records that did not meet the inclusion criteria. Of the remaining 44 reports, full-text retrieval was attempted; however, 29 could not be retrieved due to access issues or unavailability. The remaining 15 full-text articles were then assessed for eligibility. During this phase, 11 articles were excluded because they scored below 10 on the SANRA, indicating insufficient methodological quality. Ultimately, four studies fulfilled all the inclusion criteria and were included in the final review.
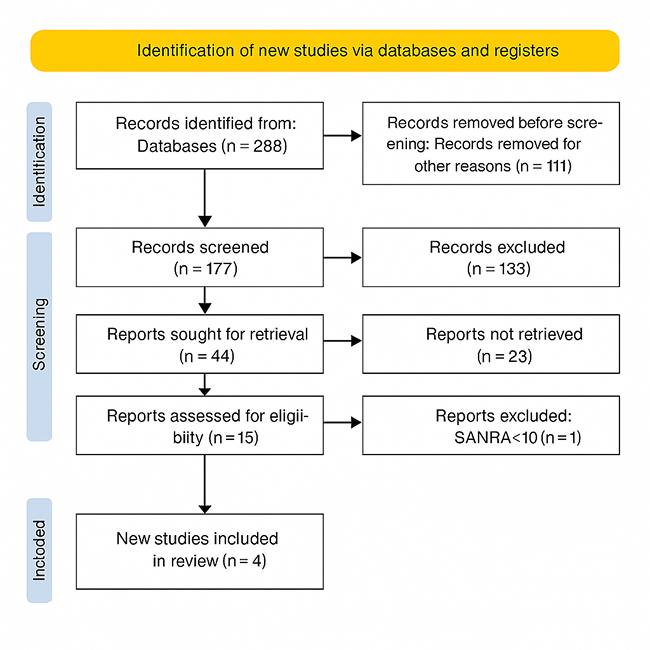
Figure 1: PRISMA flow diagram
Results
Effect of opioid free anesthesia on pain control following gastrectomy
According to Ibrahim M et al., a total of 108 patients were initially considered for the study. Of these, two refused participation and three were excluded due to protocol violations. Two received non-protocol postoperative analgesia by a new on-call physician, and one received sedation in the PACU due to severe agitation. The remaining 103 patients were randomized into the MMA and OFA groups, with similar baseline characteristics observed between both groups. All participants successfully completed the QoR-40 questionnaire. At 6 hours postoperatively, the mean difference in QoR-40 scores between groups was −4.15 (95% CI: −5.78 to −2.5, P < 0.0001), indicating better recovery in the OFA group. By 24 hours, the difference was no longer statistically significant (0.738; 95% CI: −0.8 to 2.28, P = 0.345). Since the QoR-40 scores did not follow a Gaussian distribution, the authors employed the Mann-Whitney test, which confirmed the significance observed with the parametric test.
The OFA group demonstrated faster tolerance to oral fluids and earlier readiness for discharge when compared to the MMA group (P = 0.022 and P = 0.001, respectively), though no significant difference was found in time to ambulation (P = 0.169). Pain scores, assessed using the numeric rating scale (NRS), were significantly lower in the OFA group at the PACU, as well as at 2 and 6 hours postoperatively. Moreover, the incidence of NRS >3 was lower in the OFA group (median value 0.50) compared to the MMA group (0.58), with statistical significance (P = 0.008). Opioid consumption was also reduced in the OFA group. According to the study, both PACU opioid use and total 24-hour consumption were significantly lower in the OFA group compared to the MMA group (P = 0.005 and P = 0.024, respectively).
In terms of hemodynamic stability, 16 patients (30.7%) in the MMA group required rescue fentanyl for mean arterial pressure (MAP) elevation, with a mean dose of 11.83 ± 20.79 μg. Among them, eight patients needed one dose, seven required two doses, and one patient required three doses. The OFA group had fewer MAP elevation events (median 0.10 vs. 0.28, P = 0.002). Three patients in the OFA group experienced bradycardia (heart rate (HR) < 45 bpm), which was effectively managed with atropine. None of the patients in the OFA group required labetalol for elevated HR or MAP.
Extubation time was shorter in the MMA group (6.85 ± 3.81 min) compared to the OFA group (9.24 ± 3.29 min), with statistical significance (P = 0.001). The incidence of postoperative nausea and vomiting (PONV) was slightly lower in the OFA group (35.3%) than in the MMA group (46.2%), though not statistically significant (P = 0.262). Interestingly, 17.6% of patients in the OFA group experienced hallucinations, whereas no such events were reported in the MMA group. No adverse effects related to the oblique subcostal transversus abdominis plane (OSTAP) block or systemic local anesthetic toxicity were observed in either group.[27]
Effect of anesthesia choice on recovery and cognitive function after gastrectomy
According to Liu J et al., baseline characteristics were comparable among the three study groups, with no statistically significant differences observed (P > 0.05). However, the use of vasoactive drugs was significantly higher in group P compared to groups E and PE (P < 0.05). In terms of systolic blood pressure (SBP), there were no differences between groups at baseline (T0). From 3 minutes post-induction (T1) to the start of surgery (T4), all groups showed significant decreases in SBP compared to baseline (P < 0.05). At intubation (T2) and extubation (T6), SBP in group E was significantly higher than in groups P and PE (P < 0.05). Group P had significantly lower SBP than the other groups during the T1–T4 interval (P < 0.05).
Diastolic blood pressure (DBP) was also similar at baseline across all groups (P > 0.05). In group P, DBP decreased significantly from T1 to T6 compared to baseline (P < 0.05). At T2 and T6, group E showed significantly higher DBP than groups P and PE (P < 0.05), and DBP from T1 to T4 was significantly lower in group P than in groups E and PE (P < 0.05). HR did not differ at baseline among groups. HRs in groups P and PE were significantly reduced from T1 to T4 when compared to their baseline values (P < 0.05). Group E had significantly higher HRs at T2 and T6 than the other two groups (P < 0.05). Group P exhibited greater HR reductions during the T1–T4 period compared to groups E and PE (P < 0.05).
Regarding postoperative recovery, group E had significantly longer awakened and response times to questions than groups P and PE (P < 0.05). Respiratory depression occurred in three patients from group P but was absent in groups E and PE (P < 0.05). Although nausea was common and vomiting rare across groups, adverse event rates were lower in groups P and PE compared to group E, but the difference was not statistically significant (P > 0.05). Postoperative cognitive function, assessed via Montreal Cognitive Assessment (MoCA) and Mini-Mental State Examination (MMSE) scores, showed no significant preoperative differences among groups. On postoperative day 7, MoCA scores in group P were significantly lower than those in groups E and PE (P < 0.05), while MMSE scores remained comparable across all groups (P > 0.05).[28]
Effect of sevoflurane concentration on anesthetic recovery in gastrectomy
According to Shao L et al., 78 patients were initially assessed for eligibility. Of these, 12 declined participation and seven did not meet inclusion criteria (2 were over 65 years old; 5 had body mass index (BMI) < 30 kg/m²). Ultimately, 59 patients were enrolled. After surgery, four patients in the S1.0 group and 5 in the I group were transferred to the intensive care unit (ICU) and excluded, leaving 25 patients in each group for final analysis. There were no statistically significant differences between the two groups in demographic variables including sex (P = 0.440), age (P = 0.111), height (P = 0.797), weight (P = 0.946), BMI (P = 0.560), or American Society of Anesthesiologists (ASA) classification (P = 0.338).
Intraoperative measurements showed that the total doses of propofol and remifentanil were significantly lower in the S1.0 group than in the I group (P < 0.001 for both). The total dose of rocuronium was also significantly reduced in the S1.0 group (P = 0.028). However, no significant differences were observed in body movement during surgery (P = 0.317) or in the total dose of sufentanil (P = 0.784 and P = 0.693). Extubation time was significantly shorter in the S1.0 group compared to the I group (P = 0.039).
Muscle relaxation parameters showed no difference in clinical muscle relaxant time (P = 0.566), but the in vivo action time of rocuronium was longer in the I group (58.7 ± 14.1 min) compared to the S1.0 group (50.5 ± 10.0 min; P = 0.022). Recovery time from 25% to 75% of T1 was significantly shorter in the S1.0 group (4.7 ± 4.6 min) than in the I group (11.5 ± 8.5 min; P = 0.001). Regarding surgical field quality, the Leiden-Surgical Rating Scale (LSR) scores did not differ significantly between groups at the beginning of surgery (P = 0.166), one hour into surgery (P = 0.510), or when rocuronium infusion was stopped (P = 0.561). However, LSR scores were significantly higher in the S1.0 group at the time of abdominal closure (P < 0.001).
In the PACU, pain scores were comparable between the groups (P = 0.144), but duration of stay was shorter in the S1.0 group (P = 0.039). One patient in the I group experienced dyspnea after extubation, though the difference was not statistically significant (P = 0.312). Patient satisfaction levels were similar across both groups (P = 0.297), and none of the patients reported being fairly dissatisfied during the perioperative period.[29]
Effect of gabapentin on oxycodone consumption following gastrectomy
According to Rupniewska-Ladyko A et al., 120 patients were enrolled over a 32-month period from March 2013 to November 2015. Data from seven patients were excluded due to patient-controlled analgesia (PCA) pump failure, chronic neurological disorder, allergy, or protocol violations. A total of 113 patients completed the study, with 56 in the control group and 57 in the gabapentin group. There were no significant differences between the two groups in terms of age, body mass index, duration of surgery, or anesthesia. However, the interval from the last dose of fentanyl to the first oxycodone dose in the recovery room was significantly longer in the gabapentin group (P < 0.001). Specifically, the mean time from the end of anesthesia to the initiation of analgesic therapy was 74.3 ± 37.8 minutes in the placebo group and 110.4 ± 65.4 minutes in the gabapentin group (mean difference: −36 minutes; 95% CI: −12 to −40; P = 0.0004).
Gabapentin also significantly reduced oxycodone consumption in the first 12 hours postoperatively. Patients in the gabapentin group used 26.3 ± 10 mg, compared to 31.5 ± 10 mg in the placebo group (mean difference: −5.2 mg; 95% CI: −9.08 to −1.35; P = 0.0085). Despite the reduction in opioid use, the incidence of postoperative nausea and vomiting (PONV) and the time to discharge were not significantly different between the two groups.[30]
| Study (Author) | Population/groups | Anesthesia Strategy/Agents | Pain control | Opioid use | Recovery outcomes | Adverse events |
| Ibrahim M et al.[27] | 103 patients (OFA vs MMA) | Dexmedetomidine, lidocaine, ketamine | ↓ NRS at PACU, 2h, 6h in OFA | ↓ PACU & 24h opioid use (P = 0.005, 0.024) | Faster oral fluid intake and discharge readiness (P = 0.022, 0.001) | Hallucinations in 17.6% (OFA); no block-related toxicity |
| Liu J et al.[28] | 90 patients (P vs E vs PE) | Propofol, Etomidate, Etofol | Not directly assessed | Not quantified, but PE had stable needs | Faster response & wake-up in PE group; Etomidate delayed cognition | Respiratory depression in propofol group; none in PE or E |
| Shao L et al.[29] | 50 patients (Sevo 1.0 MAC vs Inhalational group) | Sevoflurane + IV propofol/remifentanil/rocuronium | Comparable pain scores (P = 0.144) | ↓ propofol, remifentanil, rocuronium in Sevo 1.0 group | Shorter extubation and PACU stay (P = 0.039); better surgical field at closure | 1 dyspnea case (inhalational group); no dissatisfaction |
| Rupniewska-Ladyko A et al.[30] | 113 patients (Gabapentin vs placebo) | 1200 mg gabapentin 1 hr pre-op | Not directly reported | ↓ 12h oxycodone (26.3 ± 10 vs 31.5 ± 10 mg, P = 0.0085) | Longer time to first analgesic (P < 0.001); no delay in discharge | No increase in PONV or sedation |
Table 1: Comparative outcomes of opioid-free, multimodal, and balanced anesthesia in gastrectomy patients.
Discussion
Gastrectomy, whether performed for weight loss or cancer treatment, presents a unique challenge for anesthesiologists due to the physiological stress it imposes and the complexity of the patient population.[12] Obese patients undergoing LSG often have altered pharmacodynamics, increased respiratory and cardiovascular risk, and heightened sensitivity to opioid-induced complications. Consequently, anesthetic strategies in this context must balance effective analgesia, hemodynamic stability, and accelerated recovery while minimizing adverse effects.[14]
The analgesic benefits of perioperative gabapentin have been consistently supported across several studies, though variations in methodology and patient populations offer nuanced insights into its efficacy.[18] The comparative analysis of four pivotal studies by Ibrahim et al., Liu et al., Shao et al., and Rupniewska-Ladyko et al. underscores the clinical benefits and limitations of various opioid-free or reduced-opioid anesthetic strategies in obese patients undergoing LSG.
Ibrahim et al. conducted a randomized controlled trial evaluating the impact of OFA combined with loco-regional blocks under an ERAS protocol. Their results demonstrated a significantly improved QoR-40 at the 6-hour postoperative mark in the OFA group compared to the MMA group, although this difference was not maintained at 24 hours. OFA was also associated with significantly lower postoperative morphine consumption (10 mg vs. 20 mg), shorter time to oral fluid intake, and earlier discharge readiness. However, the time to ambulation did not differ significantly. These findings suggest that OFA under ERAS may accelerate early functional recovery, improve analgesia, and reduce opioid-related side effects in bariatric patients.[27]
In contrast, Liu et al. focused on hemodynamic stability and postoperative cognitive dysfunction (POCD) in radical gastrectomy patients anesthetized with dexmedetomidine combined with propofol-etomidate mixtures. Although the setting differs slightly—radical gastrectomy rather than sleeve gastrectomy—the study offers insights into the role of opioid-sparing anesthetics. Patients receiving propofol-only anesthesia (group P) had greater hemodynamic instability, with a significantly higher incidence of hypotension and postoperative respiratory depression. Conversely, groups using etomidate (group E) or a propofol-etomidate combination (group PE) demonstrated more stable systolic blood pressure and fewer respiratory complications. Notably, the addition of dexmedetomidine contributed to better intraoperative hemodynamics and possibly lower POCD rates, although these differences were not statistically significant. These findings highlight the importance of hemodynamic control and reduced neurocognitive risk in anesthetic planning, especially for high-risk populations.[28]
Shao et al. investigated the role of deep neuromuscular blockade (DNB) with sevoflurane versus intravenous anesthesia (I group) in obese patients undergoing LSG. Their study showed that the sevoflurane group (S1.0) required significantly lower intraoperative doses of propofol, remifentanil, and rocuronium. Additionally, this group exhibited shorter extubation times and PACU stays, implying more efficient recovery. However, pain scores and patient satisfaction did not differ between groups, and only one case of postoperative dyspnea was reported in the intravenous group. These findings suggest that volatile-based anesthesia, particularly sevoflurane with DNB, may contribute to anesthetic drug-sparing effects and enhanced recovery without compromising analgesia.[29]
Lastly, the study by Rupniewska-Ladyko et al. explored the preoperative administration of gabapentin (1200 mg) and its impact on postoperative opioid consumption. The gabapentin group had a significantly longer interval before needing postoperative analgesia and consumed approximately 5.2 mg less oxycodone in the first 12 hours postoperatively compared to the placebo group. These results reinforce the utility of preemptive gabapentinoids in reducing acute postoperative opioid requirements and enhancing early pain control.[30]
Comparatively, all four studies demonstrate that opioid-free or opioid-sparing strategies in bariatric and upper GI surgeries result in meaningful improvements in recovery metrics, be it reduced opioid use, shorter PACU stays, better pain control, or hemodynamic stability.[24] Ibrahim et al. emphasize functional recovery within ERAS, while Liu et al. caution against propofol-only regimens due to instability and respiratory depression. Shao et al. support the use of volatile anesthesia for smoother emergence, and Rupniewska-Ladyko et al. provide a strong case for preemptive oral adjuncts like gabapentin.
Limitations
- Limited number of high-quality, comparable studies due to variability in protocols, drug dosages, and outcome measures.
- Short-term focus of available data; lack of longitudinal outcomes such as chronic pain or cancer prognosis.
- Variability in gabapentin dosing and response, limiting universal application.
- Potential for publication bias and exclusion of non-English or inaccessible full-text studies in the review.
Future directions
- Further large-scale, multicenter randomized trials are needed to validate the efficacy and safety of OFA across diverse patient populations.
- Exploration of novel drug combinations (e.g., Etofol) and their pharmacodynamic effects in obese and oncologic patients.
- Research into personalized anesthetic protocols integrating patient genomics, pharmacokinetics, and machine-learning-based risk stratification.
- Long-term studies assessing the impact of anesthetic strategies on cancer recurrence, cognitive function, and quality of life after radical gastrectomy.
Conclusion
Anesthetic management in gastrectomy, particularly LSG, plays a crucial role in enhancing recovery, minimizing opioid-related side effects, and improving overall outcomes in high-risk populations such as the obese and those with gastric cancer. Evidence from recent studies highlights the growing utility of OFA, MMA, and balanced techniques. Ibrahim et al. demonstrated that OFA combined with regional blocks significantly improved early recovery metrics, reduced opioid consumption, and accelerated discharge readiness. Liu et al. emphasized the hemodynamic stability and lower risk of postoperative cognitive dysfunction with etomidate-based combinations compared to propofol-only anesthesia. Shao et al. reported that sevoflurane with deep neuromuscular blockade led to shorter extubation and PACU stays without compromising pain control. Additionally, Rupniewska-Ladyko et al. showed that preoperative gabapentin effectively delayed opioid requirement and reduced oxycodone use postoperatively. Collectively, these findings support integrating OFA protocols, non-opioid adjuncts like gabapentin, and volatile-intravenous combinations tailored to patient risk profiles. A personalized, evidence-based anesthetic approach, especially under ERAS protocols, can significantly enhance functional recovery, reduce complications, and improve patient satisfaction following bariatric or oncologic gastrectomy.
References
- Kim J, Lee HJ. Function Preserving Gastrectomy and Quality of Life. J Gastric Cancer. 2025;25(1):247-260. doi:10.5230/jgc.2025.25.e7
PubMed | Crossref | Google Scholar - Javed H, Olanrewaju OA, Owusu FA, et al. Challenges and solutions in postoperative complications: a narrative review in general surgery. Cureus. 2023;15(12):e50942. doi:10.7759/cureus.50942
PubMed | Crossref | Google Scholar - Iannelli A, Dainese R, Piche T, Facchiano E, Gugenheim J. Laparoscopic sleeve gastrectomy for morbid obesity. World J Gastroenterol. 2008;14(6):821-827. doi:10.3748/wjg.14.821
PubMed | Crossref | Google Scholar - Arishi AA, Gosadi IM, Hakami IA, et al. Bariatric surgery reduces weight loss, comorbidities prevalence, and improves quality of life in the southern region of Saudi Arabia. Medicina (Kaunas). 2023;59(10):1695. doi:10.3390/medicina59101695
PubMed | Crossref | Google Scholar - Eipe N, Budiansky AS. Perioperative Pain Management in Bariatric Anesthesia. Saudi J Anaesth. 2022;16(3):339-346. doi:10.4103/sja.sja_236_22
PubMed | Crossref | Google Scholar - Simpson JC, Bao X, Agarwala A. Pain Management in Enhanced Recovery after Surgery (ERAS) Protocols. Clin Colon Rectal Surg. 2019;32(2):121-128. doi:10.1055/s-0038-1676477
PubMed | Crossref | Google Scholar - Yang Q, Yang H, Zhao J, Ren L. Enhanced Recovery After Surgery (ERAS) Rehabilitation Protocols Significantly Improve Postoperative Pain and Recovery in Ankle Fracture Surgery. Ther Clin Risk Manag. 2025;21:841-850. doi:10.2147/TCRM.S517790
PubMed | Crossref | Google Scholar - Guay J, Nishimori M, Kopp SL. Epidural Local Anesthetics Versus Opioid-Based Analgesic Regimens for Postoperative Gastrointestinal Paralysis, Vomiting, and Pain After Abdominal Surgery: A Cochrane Review. Anesth Analg. 2016;123(6):1591-1602. doi:10.1213/ANE.0000000000001628
PubMed | Crossref | Google Scholar - Chia PA, Cannesson M, Bui CCM. Opioid free anesthesia: feasible?. Curr Opin Anaesthesiol. 2020;33(4):512-517. doi:10.1097/ACO.0000000000000878
PubMed | Crossref | Google Scholar - Schenkel L, Vogel Kahmann I, Steuer C. Opioid-Free Anesthesia: Physico Chemical Stability Studies on Multi-Analyte Mixtures Intended for Use in Clinical Anesthesiology. Hosp Pharm. 2022;57(2):246-252. doi:10.1177/00185787211016336
PubMed | Crossref | Google Scholar - Kallio A, Scheinin M, Koulu M, et al. Effects of dexmedetomidine, a selective alpha 2-adrenoceptor agonist, on hemodynamic control mechanisms. Clin Pharmacol Ther. 1989;46(1):33-42. doi:10.1038/clpt.1989.103
PubMed | Crossref | Google Scholar - Jendoubi A, Naceur IB, Bouzouita A, et al. A comparison between intravenous lidocaine and ketamine on acute and chronic pain after open nephrectomy: A prospective, double-blind, randomized, placebo-controlled study. Saudi J Anaesth. 2017;11(2):177-184. doi:10.4103/1658-354X.203027
PubMed | Crossref | Google Scholar - Jayadevan D, Kumar L, Varghese R, et al. Evaluation of analgesic effects and hemodynamic responses of epidural ropivacaine in laparoscopic abdominal surgeries: Randomised controlled trial. J Anaesthesiol Clin Pharmacol. 2022;38(2):245-251. doi:10.4103/joacp.JOACP_153_20
PubMed | Crossref | Google Scholar - Dagher C, Mattar R, Aoun M, et al. Opioid-free anesthesia in bariatric surgery: a prospective randomized controlled trial. Eur J Med Res. 2025;30(1):320. doi:10.1186/s40001-025-02565-9
PubMed | Crossref | Google Scholar - Papasavas P, Seip RL, McLaughlin T, et al. A randomized controlled trial of an enhanced recovery after surgery protocol in patients undergoing laparoscopic sleeve gastrectomy. Surg Endosc. 2023;37(2):921-931. doi:10.1007/s00464-022-09512-3
PubMed | Crossref | Google Scholar - Gornall BF, Myles PS, Smith CL, et al. Measurement of quality of recovery using the QoR-40: a quantitative systematic review. Br J Anaesth. 2013;111(2):161-169. doi:10.1093/bja/aet014
PubMed | Crossref | Google Scholar - Fan S, Jiang H, Xu Q, et al. Risk factors for pneumonia after radical gastrectomy for gastric cancer: a systematic review and meta-analysis. BMC Cancer. 2025;25(1):840. doi:10.1186/s12885-025-14149-1
PubMed | Crossref | Google Scholar - Tsekrekos A, Okumura Y, Rouvelas I, et al. Gastric cancer surgery: balancing oncological efficacy against postoperative morbidity and function detriment. Cancers (Basel). 2024;16(9):1741. doi:10.3390/cancers16091741
PubMed | Crossref | Google Scholar - Valk BI, Struys MMRF. Etomidate and its Analogs: A Review of Pharmacokinetics and Pharmacodynamics. Clin Pharmacokinet. 2021;60(10):1253-1269. doi:10.1007/s40262-021-01038-6
PubMed | Crossref | Google Scholar - Yang WL, Wen YL, Xu WM, et al. Effect of deep neuromuscular block on the quality of early recovery after sleeve gastrectomy in obese patients: a randomized controlled trial. BMC Anesthesiol. 2024;24(1):101. doi:10.1186/s12871-024-02465-1
PubMed | Crossref | Google Scholar - Eom BW, Yoon HM, Kim YW, et al. Quality of Life and Nutritional Outcomes of Stomach-Preserving Surgery for Early Gastric Cancer: A Secondary Analysis of the SENORITA Randomized Clinical Trial. JAMA Surg. 2024;159(8):900-908. doi:10.1001/jamasurg.2024.1210
PubMed | Crossref | Google Scholar - Kim HS, Lee DC, Lee MG, et al. Effect of pneumoperitoneum on the recovery from intense neuromuscular blockade by rocuronium in healthy patients undergoing laparoscopic surgery. Korean J Anesthesiol. 2014;67(1):20-25. doi:10.4097/kjae.2014.67.1.20
PubMed | Crossref | Google Scholar - Hakkenbrak NAG, Jansma EP, van der Wielen N, et al. Laparoscopic versus open distal gastrectomy for gastric cancer: A systematic review and meta-analysis. Surgery. 2022;171(6):1552-1561. doi:10.1016/j.surg.2021.11.035
PubMed | Crossref | Google Scholar - Casati A, Putzu M. Anesthesia in the obese patient: pharmacokinetic considerations. J Clin Anesth. 2005;17(2):134-145. doi:10.1016/j.jclinane.2004.01.009
PubMed | Crossref | Google Scholar - Chincholkar M. Gabapentinoids: pharmacokinetics, pharmacodynamics and considerations for clinical practice. Br J Pain. 2020;14(2):104-114. doi:10.1177/2049463720912496
PubMed | Crossref | Google Scholar - Gupta L, Agarwal J, Saxena KN. Opioid-free anaesthesia: The conundrum and the solutions. Indian J Anaesth. 2022;66(Suppl 2): S91-94. doi:10.4103/ija.ija_256_22
PubMed | Crossref | Google Scholar - Ibrahim M, Elnabtity AM, Hegab A, et al. Combined opioid free and loco-regional anaesthesia enhances the quality of recovery in sleeve gastrectomy done under ERAS protocol: a randomized controlled trial. BMC Anesthesiol. 2022;22(1):29. doi:10.1186/s12871-021-01561-w
PubMed | Crossref | Google Scholar - Liu J, Yang J, Yang X, et al. Application of dexmedetomidine combined with propofol-etomidate mixture in radical gastrectomy under general anesthesia. Medicine (Baltimore). 2024;103(44):e39669. doi:10.1097/MD.0000000000039669
PubMed | Crossref | Google Scholar - Shao L, Liu Y, Hao J, et al. Effect of sevoflurane on the deep neuromuscular blockade in obese patients undergoing laparoscopic sleeve gastrectomy: a single center prospective randomized controlled study. Drug Des Devel Ther. 2023;17:3193-3203. doi:10.2147/DDDT.S413535
PubMed | Crossref | Google Scholar - Rupniewska-Ladyko A, Malec-Milewska M, Kraszewska E, et al. Gabapentin before laparoscopic sleeve gastrectomy reduces postoperative oxycodone consumption in obese patients: a randomized double-blind placebo-controlled trial. Minerva Anestesiol. 2018;84(5):565-571. doi:10.23736/S0375-9393.17.12194-2
PubMed | Crossref | Google Scholar
Acknowledgments
Not applicable
Funding
No funding
Author Information
Corresponding Author:
Samatha Ampeti
Department of Pharmacology
Kakatiya University, University College of Pharmaceutical Sciences, Warangal, TS, India
Email: ampetisamatha9@gmail.com
Co-Authors:
Sonam Shashikala BV, Mansi Srivastava, Shubham Ravindra Sali, Nirali Patel, Raziya Begum Sheikh
Independent Researcher
Department of Content, medtigo India Pvt Ltd, Pune, India
Authors contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation and writing-review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
Not applicable
Conflict of Interest Statement
None
Guarantor
None
DOI
Cite this Article
Sonam SBV, Samatha A, Mansi S, Shubham RS, Nirali P, Raziya BS. Evolving Anesthesia Strategies for Enhanced Recovery in Gastrectomy. medtigo J Anesth Pain Med. 2025;1(2):e3067123. doi:10.63096/medtigo3067123 Crossref









