Author Affiliations
Abstract
Objective: To evaluate the frequency of Glucose 6 Phosphate dehydrogenase (G6PD) deficiency by a G6PD quantitative method in jaundiced infants visiting the Tehsil head quarter (THQ) hospital, central Kurram Dogar.
Methodology: This descriptive study was conducted at the special care baby unit (SCBU) department of child health, Tehsil headquarters hospital, from November 2023 to April 2024. A total number of 283 newborns, aged 1-14 days of either sex, admitted with jaundice were included in the study. Biodata and clinical profile of all patients were collected on preformed proforma. G6PD decolorization time, baby and mother blood group, smear, retic count, and other related investigations were done. The results were analyzed using descriptive statistics.
Results: During the study time, a total of 710 newborns were admitted to the Special Care Baby Unit. Out of the total special care baby unit admissions, 283 (39.85%) neonates had jaundice. Among these 283 jaundiced newborn babies, the G6PD deficient were 83. Out of these 83 G6PD-deficient neonates, males were 63 (75.9%) and females were 20 (24.1%). All jaundiced neonates received phototherapy. Among G6PD deficiency jaundiced babies, 54 (65.06%) neonates with severe hyperbilirubinemia needed exchange transfusion. Nine babies (10.84%) developed kernicterus. The G6PD discoloration time test at the time of admission varied from 60-120 minutes. Serum bilirubin level ranged 9.5-40 mg.
Conclusion: G6PD deficiency is a relatively common cause of neonatal jaundice and has a greater prevalence in males sex. Babies suffering from G6PD deficiency present with early jaundice relatively earlier like other hemolytic jaundices including ABO and Rh incompatibility.
Keywords
Neonatal jaundice, Glucose 6 phosphate dehydrogenase, Hyperbilirubinemia, Infant, Bilirubin, Oxidative drugs.
Introduction
G6PD deficiency was first discovered in 1950, but the history goes back 2000 years when Egyptian priests were not allowed to take broad beans because of the occurrence of the disorder now called acute hemolytic crisis.[1] G6PD is a cytosolic enzyme encoded by an X-linked gene and expressed in all tissues of the body.[2] G6PD deficiency is the commonest enzyme defect not only in hematology but also in human physiology as a whole.[3-5] G6PD catalyzes the first step in the pentose phosphate pathway, generating nicotinamide adenine dinucleotide phosphate reduced (NADPH), which keeps the glutathione in reduced form, which has a protective role for red blood cells and other tissues from oxidative damage caused by medicines, infections, and environmental stresses.[6]
The normal enzyme found in Caucasians is G6PD-B.[7] G6PD gene is extremely polymorphic with over 130 mutations and approximately 400 variants.[8] In the majority of cases, these variants are due to structural gene mutation caused by a single amino acid substitution.[9] G6PD deficiency is the most common disorder and 400 million people throughout the world are affected in different parts of the world. The prevalence rate varies from as high as 62% among Kurdish Jews to as low as 0.1% in Japan, while it ranges from 3 to 6.9% in Pakistan, Southern China, and Southern Russia.[10,11] The commonest presentation of G6PD deficiency in Pakistan is neonatal jaundice and acute hemolysis.[12]
Neonatal Jaundice is an important condition and accounts for many SCBU admissions. Neonatal jaundice is commonly present in the first week of life. Pathological jaundice even appears on the first day of life and can lead to complications if no proper intervention is taken in a timely manner. Serum hyperbilirubinemia is defined as a total serum bilirubin (TSB) level above the 95th centile for that specific age in hours, occurring in 8-9% of neonates during the first week of life.[13,14] If the jaundice progresses and the serum bilirubin level exceeds 25-30 mg/dL, greater than the 99th centile, even healthy neonates are prone to develop neuronal damage leading to kernicterus, unless proper interventional measures are taken.[14] Indirect bilirubin content of the total serum bilirubin level is a notorious component because it crosses the blood-brain barrier and accumulates in the nervous tissue, causing grievous injuries to neuronal tissue and resulting in kernicterus. Several factors are responsible for neonates developing hyperbilirubinemia, including an imbalance between production, conjugation, and elimination of bilirubin, environmental factors, and ethnicity.[15] G6PD deficiency is one of the common aetiologic factors for neonatal hyperbilirubinemia in our country, however the data is limited. This study was conducted to determine the relative frequency of G6PD deficiency in neonatal jaundice.
Methodology
This hospital-based descriptive study was conducted at SCBU, the department of child health Tehsil headquarter hospital central Kurram Dogar, from November 2023 to April 2024. All full-term neonates aged 1-14 days who were presented with jaundice were included in the study. Low birth weight babies, neonates with congenital anomalies such as cleft palate were excluded from the study. Neonates with down syndrome, biliary atresia, hypothyroidism, and neonatal hepatitis syndrome were also excluded from the study. Complete history, detailed examination, and investigations of every neonate were recorded on a preformed proforma, including sex, age, address, appearance of jaundice, time of admission, and any history of jaundice and exchange transfusion in other siblings. The investigations done in these jaundiced babies were blood groups of the baby and mother, G6PD, full blood count (FBC), reticulocyte count, and serum bilirubin total (direct and indirect).
G6PD enzyme estimation was done in minutes using Sigma Diagnostic G6PD reagent. The test vial was observed at 25-minute intervals up to one hour for a change of color from its original deep blue to a maroon or reddish endpoint. The individual reactions were classified as normal or G6PD deficient based on their dye-decolorization time. Change of color within one hour indicated a normal level of glucose-6-phosphate dehydrogenase. The samples that did not decolorize after 70 minutes were labeled as G6PD deficient. Results were analyzed using descriptive statistics.
Results
The total number of babies admitted to special care baby units during the study period was 710. Out of these 283 (39.85%) neonates were admitted with jaundice. Out of these 283 jaundiced newborn babies, the G6PD deficient were 83 (29.3%). The 2nd common cause of neonatal jaundice was ABO incompatibility followed by Rh incompatibility constituting 25.4% and 12.4% of the cases respectively. The results are shown in table 1. Amongst the G6PD deficient male neonates were 63 (75.9%), and the females were 20 (24.1%) as given in figure 1. The G6PD decolorization test at the time of admission varied from 60-120 minutes. Serum bilirubin level in these newborns ranged from 9.5-40.0 mg, with a Mean value of 18.90 ± 6.62. The mean, standard deviation (SD), and range of hemoglobin, reticulocyte count, serum bilirubin, and G6PD discoloration time are given in Table 2.
The mean age of appearance of jaundice in G6PD-deficient babies was 1.85 ± 0.74 days, while the range was 1-3.5 days. The mean age of the jaundiced neonates at the time of admission was 6.29 ± 2.69 days, while the range was 1-14 days. All G6PD-deficient babies eventually received phototherapy. Among 83 G6PD-deficient neonates, 54 (65.06%) babies with severe hyperbilirubinemia required exchange blood transfusions. Exchanged blood transfusion was done once in 49 patients (59.04%) while in the remaining five cases (6.02%), it was done more than once, as given in Figure 2. Nine G6PD-deficient neonates (10.84%) developed Kernicterus.
| Causes of jaundice | No of patients | Percentage |
| G6PD deficiency | 83 | 29.3 % |
| ABO incompatibility | 72 | 25.4 % |
| Rh incompatibility | 35 | 12.4 % |
| Other causes | 93 | 32.9 % |
| Total | 283 | 100 % |
Table 1: Frequency of G6PD deficiency in jaundiced neonates (Causes of neonatal jaundice)
| Test (Unit) | Mean ± SD | Range |
| Hemoglobin (gm/dL) | 16.53 ± 4.25 | 12-18.5 |
| Reticulocyte count | 4.50 ± 1.25 | 2-8.5 |
| Serum total bilirubin (mg/dL) | 19.5 ± 6.54 | 7.5-30.5 |
| G6PD discoloration time (minutes) | 92.92 ± 23.07 | 60-120 |
Table 2: Laboratory profile with mean and range in G6PD-deficient jaundiced neonates
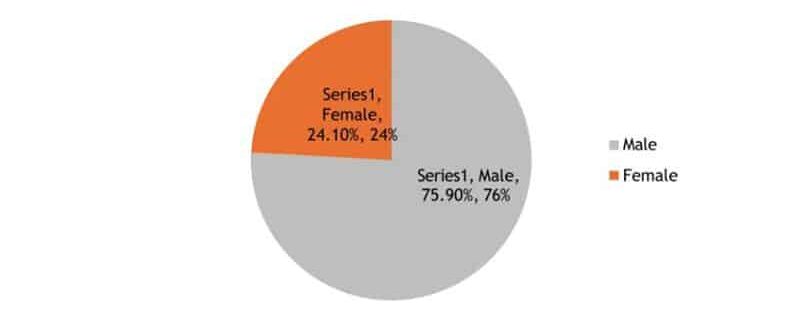
Figure 1: Gender-wise distribution of G6PD deficiency (n=83)
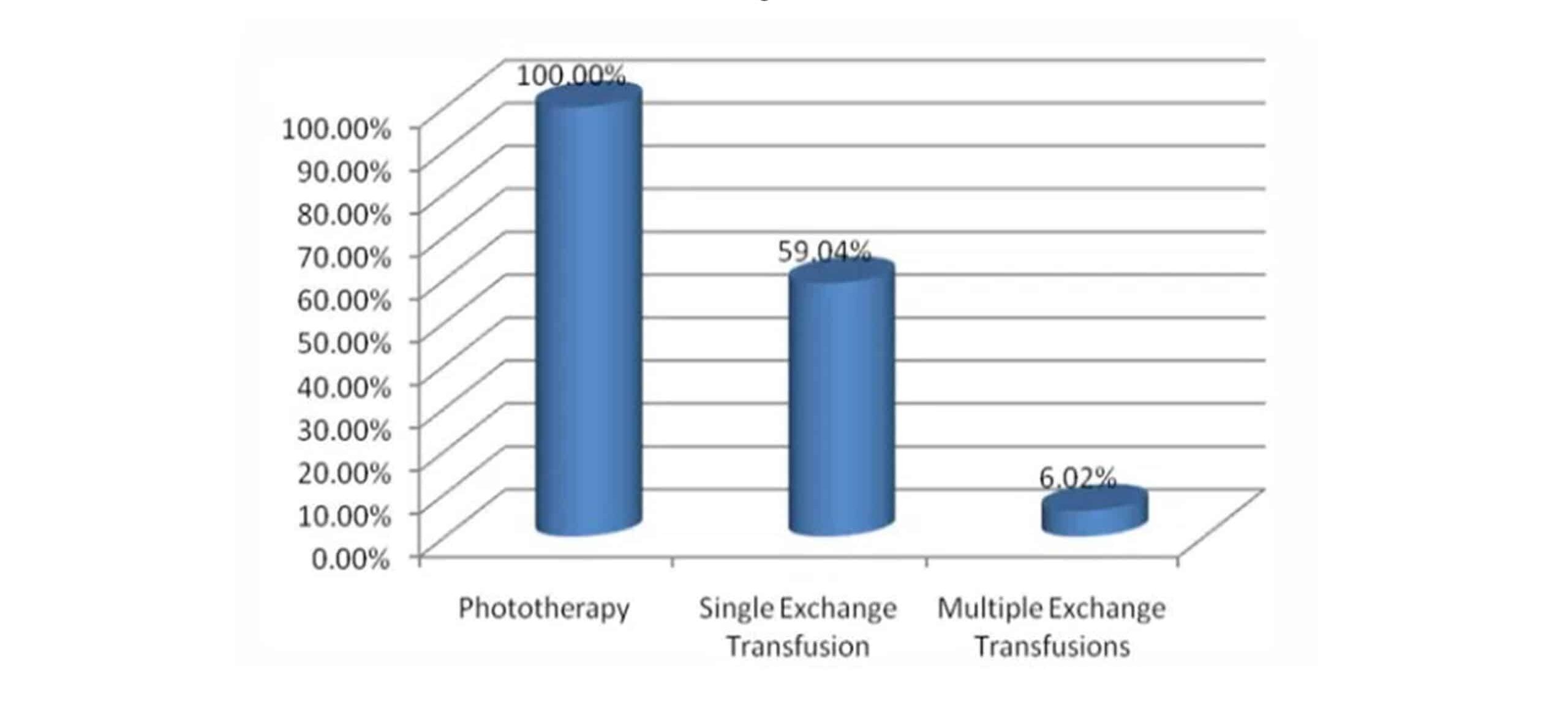
Figure 2: Therapeutic modalities in jaundiced neonates with G6PD deficiency
Discussion
In our study, we found that neonatal jaundice is an important neonatal condition, making up a huge part of our special care baby unit admissions. In this study, neonatal jaundice constituted 40 % of special care baby unit admissions, which is a significant amount. A study conducted at the Nursery Unit lady reading hospital by Hussain M et al showed that neonatal jaundice constituted 23 % of the nursery admissions.[16] Several other international studies support our study regarding neonatal jaundice as an important cause of the special care baby unit
admission.[17,18]
Neonatal jaundice has several causes. In our study we found three important causes of early neonatal jaundice, G6PD deficiency, ABO and Rhesus incompatibility. Almost the same etiologies of neonatal jaundice have been mentioned in various national and international studies.[19,20]
Frequency of G6PD deficiency was an important variable in our study. In our study we found that G6PD deficiency made a big proportion of the jaundiced neonates i.e. 29.3 %. The data regarding the frequency of G6PD deficiency is different in various parts of our country and abroad. The frequency was 12 %, 13%, 14 %, and 16 % in national studies conducted by Imran et al.[21], Khan A et al.[22], Rehman G et al.[23] and Hussain M et al.[16] respectively. These figures are comparatively lower than ours. Various reasons may be responsible for the increasing frequency. One reason may be the high prevalence of the pathology in Pakhtoon region. Study data available on various ethnic groups show a high frequency in Pathans as compared to the rest of the ethnic groups of the country, and secondly, the figures are rising because of the practice of cousin marriages in our part of the country.[5]
International data regarding the G6PD deficiency frequency varies a lot. Studies from Saudi Arabia, Iran, and India show the frequency of G6PD deficiency as low as 2%, 2.1%, and 7.5%, respectively.[24-26] These figures are quite low compared to our results. On the other hand, studies in China, Nigeria, and Thailand found a high occurrence of G6PD deficiency with figures of 18.42%, 25.5%, and 38 %, respectively.[27-29] These figures are almost comparable to our results, while the one from Nigeria is even above ours.
Gender wise distribution varies in G6PD deficiency. As it is a sex-linked recessive disorder, it is obvious to be a pathology of the male sex. But still female is not excluded. Male to female ratio has been observed different in various national and international studies. In our study, this ratio was almost 3:1, which means that females made up almost one-fourth of the study population. The sex ratio has varied in different studies. A study conducted at district Timergara by Rahim F et al.[30] shows a ratio of 7:1. The same results have been given by Ahmed I et al.[31] The difference between the ratios of male to female is quite high compared to ours. But studies are there where the difference was lower than ours, with a male-to-female ratio of 2:1.[32] Neonatal jaundice appeared early in our study. Almost the same was found in other hospital-based studies, while in another study, presentation was late as compared to ours.[22,30,31] One explanation for this difference may be that the last study was conducted mostly at a clinical laboratory.
Recommendations: Public awareness should be created so that the moment parents notice the jaundice, the baby should be taken to the nearest hospital for timely investigations and management. Early recognition of the pathology and management will significantly reduce the morbidity and mortality related to severe hyperbilirubinemia due to G6PD deficiency.
Limitation of study: The results of this study are subject to several limitations that may influence its overall reliability and generalizability. One of the primary constraints was the limited sample size, which restricts the ability to capture diverse perspectives and may not represent the broader population. Additionally, the study was conducted within a restricted timeframe, which imposed challenges on thorough data collection and analysis. Financial constraints further limited the resources available for an expansive or in-depth investigation. The data collection process was also influenced by the ease of access to participants or sources, potentially introducing bias or reducing the variety of data gathered. Finally, ethical considerations played a significant role in shaping the study, including adherence to privacy, informed consent, and maintaining the integrity of the research process. These factors collectively shaped the scope and depth of the study’s outcomes.
Conclusion
G6PD deficiency is a common cause of neonatal jaundice and has more preponderance for male sex. Babies suffering from G6PD deficiency present with early neonatal jaundice.
References
- Eghbal F, Fakoorziba MR, Eghbal MH, Latifnia S. Survey on causes of hemolysis in Glucose-6-Phosphate Dehydrogenase (G6PD) deficient pediatric patients. Pak J Med Sci.2012;28(4):666-669 Survey on causes of hemolysis in Glucose-6-Phosphate Dehydrogenase (G6PD) deficient pediatric patients.
- Ley B, Alam MS, O’Donnell JJ, et al. A Comparison of Three Quantitative Methods to Estimate G6PD Activity in the Chittagong Hill Tracts, Bangladesh. PLoS One. 2017;12(1):e0169930. doi:10.1371/journal.pone.0169930
PubMed | Crossref | Google Scholar - Merdin A, Avci F, Guzelay N. Glucose-6-phosphate dehydrogenase deficiency presented with convulsion: a rare case. Hematol Rep. 2014;6(1):5266. doi:10.4081/hr.2014.5266
PubMed | Crossref | Google Scholar - Pahlavanzadeh M, Hekmatimoghaddam S, Teremahi Ardestani M, Ghafoorzadeh M, Aminorraaya M. G6PD Enzyme Deficiency in Neonatal Pathologic Hyperbilirubinemia in Yazd. Iran J Ped Hematol Oncol. 2013;3(2):69-72 Enzyme Deficiency in Neonatal Pathologic Hyperbilirubinemia in Yazd
- Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet. 2008;371(9606):64-74. doi:10.1016/S0140-6736(08)60073-2 PubMed | Crossref | Google Scholar
- Usanga EA, Ameen R. Glucose-6-phosphate dehydrogenase deficiency in Kuwait, Syria, Egypt, Iran, Jordan and Lebanon. Hum Hered. 2000;50(3):158-161. doi:10.1159/000022906
PubMed | Crossref | Google Scholar - Agarwal A, Nayak M D, Patil A, Manohar C. Glucose 6 phosphate dehydrogenase deficiency unmasked by diabetic ketoacidosis: an underrated phenomenon. J Clin Diagn Res.
2013;7(12):3012-3013. doi:10.7860/JCDR/2013/6159.3892 Glucose 6 phosphate dehydrogenase deficiency unmasked by diabetic ketoacidosis: an underrated phenomenon - Peng Q, Li S, Ma K, et al. Large cohort screening of G6PD deficiency and the mutational spectrum in the Dongguan District in Southern China. PLoS One. 2015;10(3):e0120683. doi:10.1371/journal.pone.0120683
PubMed | Crossref | Google Scholar - Mehta A, Mason PJ, Vulliamy TJ. Glucose-6-phosphate dehydrogenase deficiency. Baillieres Best Pract Res Clin Haematol. 2000;13(1):21-38. doi:10.1053/beha.1999.0055 PubMed | Crossref | Google Scholar
- Fiorelli G, Martinez di Montemuros F, Cappellini MD. Chronic non-spherocytic haemolytic disorders associated with glucose-6-phosphate dehydrogenase variants. Baillieres Best Pract Res Clin Haematol. 2000;13(1):39-55. doi:10.1053/beha.1999.0056 PubMed | Crossref | Google Scholar
- Howes RE, Dewi M, Piel FB, et al. Spatial distribution of G6PD deficiency variants across malaria-endemic regions. Malar J. 2013;12:418. doi:10.1186/1475-2875-12-418 PubMed | Crossref | Google Scholar
- Ali N, Anwar M, Ayyub M, Bhatti FA, Nadeem M, Nadeem A. Frequency of glucose-6-phosphate dehydrogenase deficiency in some ethnic groups of Pakistan. J Coll Physicians Surg Pak. 2005;15(3):137-141 Frequency of glucose-6-phosphate dehydrogenase deficiency in some ethnic groups of Pakistan
- Bhutani VK, Johnson L, Sivieri EM. Predictive ability of a predischarge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy term and near-term newborns. Pediatrics. 1999;103(1):6-14. doi:10.1542/peds.103.1.6 PubMed | Crossref | Google Scholar
- Stevenson DK, Fanaroff AA, Maisels MJ, et al. Prediction of hyperbilirubinemia in near-term and term infants. Pediatrics. 2001;108(1):31-39. doi:10.1542/peds.108.1.31 PubMed | Crossref | Google Scholar
- Smitherman H, Stark AR, Bhutani VK. Early recognition of neonatal hyperbilirubinemia and its emergent management. Semin Fetal Neonatal Med. 2006;11(3):214-224. doi:10.1016/j.siny.2006.02.002
PubMed | Crossref | Google Scholar - Hussain M, Irshad M, Kalim M, Ali L, Ali L. Glucose-6-phosphate dehydrogenase deficiency in jaundiced neonates. J Postgrad Med Inst.2010;24(2):122-26 Glucose-6-phosphate dehydrogenase deficiency in jaundiced neonates
- Dzinović A, Heljić S, Maksić H, Hrnjić Z. Neonatal hyperbilirubinemia: evaluation and treatment.
Med Arh. 2002;56(3 Suppl 1):44-45 Neonatal Hyperbilirubinemia: Evaluation and Treatment - Smitherman H, Stark AR, Bhutani VK. Early recognition of neonatal hyperbilirubinemia and its emergent management. Semin Fetal Neonatal Med. 2006;11(3):214-224. doi:10.1016/j.siny.2006.02.002
PubMed | Crossref | Google Scholar - Bhutani VK, Johnson LH, Shapiro SM. Kernicterus in sick and preterm infants (1999-2002): a need for an effective preventive approach. Semin Perinatol. 2004;28(5):319-325. doi:10.1053/j.semperi.2004.09.006
PubMed | Crossref | Google Scholar - Atay E, Bozaykut A, Ozahi Ipek I. Glucose-6-phosphate dehydrogenase deficiency in neonatal indirect hyperbilirubinemia. J Trop Pediatr.2006;52(1):56-58. doi:10.1093/tropej/fmi042 PubMed | Crossref | Google Scholar
- Stevenson DK, Fanaroff AA, Maisels MJ, et al. Prediction of hyperbilirubinemia in near-term and term infants. Pediatrics. 2001;108(1):31-39. doi:10.1542/peds.108.1.31 PubMed | Crossref | Google Scholar
- Jan AZ, Zahid SB, Ahmad S. Frequency of G6PD deficiency and its severity in neonatal jaundice in Rehman Medical Institute, Peshawar. Khyber Med Univ J. 2013;5(1):36-39. https://www.kmuj.kmu.edu.pk/article/view/11345
- Par EJ, Hughes CA, DeRico P. Neonatal Hyperbilirubinemia: Evaluation and Treatment. Am Fam Physician. 2023;107(5):525-534. https://www.aafp.org/pubs/afp/issues/2023/0500/neonatal-hyperbilirubinemia.html
- Moiz B, Nasir A, Khan SA, Kherani SA, Qadir M. Neonatal hyperbilirubinemia in infants with G6PD c.563C > T Variant. BMC Pediatr. 2012;12:126. doi:10.1186/1471-2431-12-126 PubMed | Crossref | Google Scholar
- Muzaffer MA. Neonatal screening of glucose-6-phosphate dehydrogenase deficiency in Yanbu, Saudi Arabia. J Med Screen. 2005;12(4):170-171. doi:10.1258/096914105775220660
PubMed | Crossref | Google Scholar - Abolghasemi H, Mehrani H, Amid A. An update on the prevalence of glucose-6-phosphate dehydrogenase deficiency and neonatal jaundice in Tehran neonates. Clin Biochem.
2004;37(3):241-244. doi:10.1016/j.clinbiochem.2003.11.010 PubMed | Crossref | Google Scholar - Iranpour R, Akbar MR, Haghshenas I. Glucose-6-phosphate dehydrogenase deficiency in neonates. Indian J Pediatr. 2003;70(11):855-857. doi:10.1007/BF02730581
PubMed | Crossref | Google Scholar - Weng YH, Chou YH, Lien RI. Hyperbilirubinemia in healthy neonates with glucose-6-phosphate dehydrogenase deficiency. Early Hum Dev. 2003;71(2):129-136. doi:10.1016/s0378-3782(03)00002-1 PubMed | Crossref | Google Scholar
- Uko EK, Agwunobi SN, Udoh JJ. Glucose-6-phosphate dehydrogenase (G-6-PD) levels in jaundiced neonates in Calabar. Niger J Med. 2003;12(2):98-102 Glucose-6-phosphate dehydrogenase (G-6-PD) levels in jaundiced neonates in Calabar
- Thaithumyanon P, Visutiratmanee C. Double phototherapy in jaundiced term infants with hemolysis. J Med Assoc Thai. 2002;85(11):1176-1181 Double phototherapy in jaundiced term infants with hemolysis
- Rahim F, Zahid S, Shah SMA, Said H, Uzair M. Glucose-6-phosphate dehydrogenase (G6PD) deficiency in neonates presenting with jaundice. J Postgrad Med Inst.2008;22(2):102-106 Glucose-6-phosphate dehydrogenase (G6PD) deficiency in neonates presenting with jaundice
- Lee HY, Ithnin A, Azma RZ, Othman A, Salvador A, Cheah FC. Glucose-6-Phosphate Dehydrogenase Deficiency and Neonatal Hyperbilirubinemia: Insights on Pathophysiology, Diagnosis, and Gene Variants in Disease Heterogeneity. Front Pediatr. 2022;10:875877. doi:10.3389/fped.2022.875877 PubMed | Crossref | Google Scholar
- Abu-Osba YK, Mallouh AA, Hann RW. Incidence and causes of sepsis in glucose-6-phosphate dehydrogenase-deficient newborn infants. J Pediatr. 1989;114(5):748-752. doi:10.1016/s0022-3476(89)80131-3
PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
Not reported
Author Information
Corresponding Author:
Bilal Noor
Department of Pediatric Medicine
Doggar Hospital, Pakistan
Email: bilalzk261@gmail.com
Co-Authors:
Hamid Iqbal
Department of Pediatric Medicine
Nasir Ullah Babar Memorial Hospital Peshawar, Pakistan
Muhammad Suliman
Clinical Technologist
Department of Clinical Technology, Doggar Hospital, Pakistan
BSC (MLT) THQ Hospital central kurram Dogar
Nasar Ullah
Consultant Anesthesia
Department of Dentistry
MTI Abbottabad University, Khyber Medical University, Pakistan
Tahir Hayat
Consultant Pediatrician
Department of Pediatrics
Children Specialist, Khyber Pakhtunkhwa, Pakistan
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
Ethical approval for the study was granted by the THQ hospital central Kurram Dogar ethical board. Data was collected after getting written consent from the parents of enrolled children in our study.
Conflict of Interest Statement
The authors declare no conflict of interest.
Guarantor
None
DOI
Cite this Article
Bilal N, Hamid I, Muhammad S, Nasar U, Tahir H. Evaluation of the Frequency of G6PD Deficiency by a G6PD Quantitative Method in Jaundiced Infants Visiting Dogar Central Kurram Hospital. medtigo J Med. 2025;3(1):e30623110. doi:10. 63096/medtigo30623110 Crossref









