Author Affiliations
Abstract
Background: Renal artery variations (RAV) are common anatomical deviations that can significantly impact clinical outcomes in surgical and interventional procedures. There is limited comprehensive data correlating these variations with clinical evidence in southwestern Nigeria.
Purpose: To correlate renal artery variation in Southwestern Nigeria with clinical findings using computed tomography (CT).
Materials and Methods: A prospective cross-sectional study was conducted involving 250 participants who were referred for an abdominal CT scan. The RAV spectrum was examined based on gender, age, and body mass index (BMI).
Results: The participants consisted of 44.4% females and 55.6% males, with an overall average age of 47.97 years. The average BMI was 24.67 kg/m², with 46% of participants having a normal weight. Participants with normal renal artery were 44% while the remainder had variations as follows: aberrant origin of the renal artery (AORA) – 2.4%, bilateral accessory renal arteries (BARA) – 3.2%, bilateral renal artery stenosis (BRAS) – 0.8%, double accessory renal artery (DARA) – 3.6%, early division renal artery (EDRA) – 27% and single accessory renal artery (SARA) – 19%. There was no significant difference in the number of variations across variables (p > 0.05). However, there was a substantial difference in the number of variations within the spectrum of renal arteries (p < 0.001). A significant association was noted between RAV and clinical findings (p = 0.0252). The accuracy, sensitivity, and specificity of RAV in detecting abnormal arteries were 53.02%, 69.5%, and 48.2%, respectively.
Conclusion: The study shows the renal artery variations of a population in southwestern Nigeria, and recognizing these anatomical variations in clinical practice can enhance diagnostic accuracy and improve surgical outcomes.
Keywords
Renal artery variations, Accessory renal arteries, Early branching, Aberrant origin, Clinical findings, Computed tomography.
Introduction
The kidneys are structures or organs located bilaterally in the retroperitoneal region, at the upper quadrants of the abdomen. The blood supply of the kidney is provided by the renal artery, which emerges from the abdominal aorta around the first lumbar vertebra (L1), typically located at the level of the L1/L2 intervertebral disc, immediately inferior to the origin of the superior mesenteric artery.[1] It branches laterally from the aorta and proceeds to the kidneys, where it splits into smaller arteries and eventually forms arterioles, which provide oxygenated blood to various parts of the kidney. They are generally 4 cm to 6 cm long, 5 mm to 6 mm in diameter, and run laterally and posteriorly due to the hilum’s location.[2,3]
Understanding the anatomical variation of the renal artery is essential for surgeons and radiologists to prevent complications during procedures, especially in kidney transplantation. Additionally, the presence of multiple renal vessels, including accessory renal arteries and veins, has been documented, underscoring the significance of this knowledge for surgical and radiological practices. Moreover, anatomical variations in the renal vascular pedicle have been reported to be higher than previously thought, indicating the need for thorough assessment in clinical settings.[4,5] Studies have also discussed variations in the middle segmental artery of the kidney and their clinical implications, further emphasizing the importance of understanding renal vascular anatomy. Furthermore, the presence of double renal arteries originating from the aorta and venous communication between the right and left kidneys has also been documented, highlighting the complexity of renal vascular anatomy.[6]
Accurate identification of renal artery variants is crucial for diagnosing conditions such as renal artery aneurysms, fibromuscular dysplasia, and renal artery thrombosis.[7] Aneurysms of the renal arteries can develop when there are multiple renal arteries and aberrant branching.[8] Aremu et al.[9] found that variation in renal vascular anatomy is a frequent finding among the Nigerian population. Research into the clinical implications of renal artery variations reveals significant correlations with procedural success and complications. Similarly, variations could impact the technical success of endovascular interventions and the management of conditions such as renovascular hypertension.
The gold standard for non-invasive renal artery imaging is still computed tomography angiography (CTA) today.[10,11] Contemporary CT technology provides high-resolution pictures and three-dimensional reconstructions that accurately assess renal artery architecture. This aids in diagnosis, therapy planning, and post-treatment monitoring of patients with a range of renal and vascular disorders. With this background, it becomes important that the relationship between clinical findings and renal artery variance in Nigeria be known. The correlation between renal artery variation and radiological findings among south-western Nigerians is currently unavailable, and thus, the essence of this study, which is to analyze renal artery variation, branching, and course and to correlate it with clinical findings, as well as age and gender, in southwestern Nigeria, using multi-detector computed tomography of the abdomen and its correlation with age, gender and BMI.
Methodology
This study was carried out at a tertiary hospital in the Southwestern region of Nigeria. The study was conducted according to the principles of the declaration of helsinki in 1964.[12] Ethical approval was granted by the ethics and research committee of the institution. Additionally, permission and support were obtained from the radiology department’s managing consultants. Informed written consent was obtained from all subjects before the examination, where the reason for the study, benefits, possible effects, and stages of examination were explained to the subjects.
This prospective study comprised 250 consenting patients from Southwestern Nigeria who were referred for an abdominal computed tomography scan from March 2024 to May 2024 using a random sampling technique. Adults aged 18-80 years with confirmed abdominal CT scan requests were included in the study. Patients with prior renal surgery that might alter the normal anatomy, incomplete or poor-quality CT images with a Hounsfield unit below 200Hu, as well as patients with known congenital kidney anomalies (e.g., horseshoe kidney, polycystic kidney disease), were excluded due to possible distortions of normal anatomy.
On the day of the study, the participants’ weight and height were measured to determine the amount of contrast media (Omnipaque 350 mg/ml) to be injected, using a Leica mechanical height and weight scale model 15 R, after which the patient was positioned lying supine on the multi-detector computed tomography (MDCT) couch, with the leg first entering the MDCT gantry. All axial and coronal views, including the arterial and delay phase, were acquired using MDCT (64 slides), and a three-dimensional picture of the volume rendering (VR) and maximum intensity projection (MIP) of the arterial phase was generated using a CT Workstation Software (Version 2019r). Two expert radiologists assessed the images separately. Variations in renal artery anatomy were documented. Relevant clinical data were collected, including demographic information (age, gender), clinical indications for abdominal computed tomography, and a history of hypertension or renal artery stenosis. The images were analyzed and then compared for variability due to age and gender, and a correlation was made between renal artery variation and clinical findings.
Standard values for classification of the BMI (kg/m2) were used as follows:
- Underweight: BMI < 18.5
- Normal weight: BMI 18.5 – 24.9
- Overweight: BMI 25 – 29.9
Obesity:
- Class I: BMI 30 – 34.9
- Class II: BMI 35 – 39.9
- Class III (severe or morbid obesity): BMI ≥ 40
Microsoft Excel in Windows 11 was used for data entry and cleaning. Analysis of variance (ANOVA) for used to compare more than two means. All analyses were performed using R statistical computing programming version 4.3.1 in the RStudio environment. The level of significance for all inferential statistics was set at p < 0.05.
Results
A total of two hundred and fifty participants were recruited for this study. The females were 44.4% (111) and the males 55.6% (139). The overall average age, majority age group, average BMI, and common BMI status were 47.97 years, ≥ 60 years (27%), 24.67 Kg/m2, and normal weight (46%), respectively (Table 1). Table 2 shows the spectrum of renal artery variations (RAV) based on gender. A normal renal artery supply was present in 44% (Figure 1) of the participants while the rest had renal artery variations as follows: AORA (2.4%), BARA (3.2%) (Figure 2), BRAS (0.8%), DARA (3.6%), EDRA (27%), and SARA (19%) (Figure 3). Figure 4 shows the distribution of the location of renal artery variations amongst the participants. The comparison of the number of variations by RAV, gender, age, and BMI is shown in Figure 5. The ANOVA analysis showed insignificant differences in the number of variations by age, gender, and BMI (all p > 0.05). However, there was substantial statistical evidence of a difference in the number of renal artery variations (p < 0.0001). The distribution of renal artery in the stratification of age is shown in Table 3.
| Parameter | N | Overall, N = 250 | Gender | p-value | |
| Female, N = 111 | Male, N = 139 | ||||
| Age (Years) | 250 | 47.97±17.59 | 47.56±17.02 | 48.27±18.09 | 0.923 |
| Height (m) | 250 | 1.66±0.08 | 1.66±0.09 | 1.66±0.07 | 0.358 |
| Weight (Kg) | 250 | 67.64±11.27 | 68.76±10.27 | 66.75±12.07 | 0.262 |
| BMI (Kg/m2) | 250 | 24.67±4.63 | 24.99±4.43 | 24.41±4.78 | 0.345 |
| BMI Status | 250 | 0.444 | |||
| · Normal | 116 (46%) | 49 (44%) | 67 (48%) | ||
| · Obese | 36 (14%) | 14 (13%) | 22 (16%) | ||
| · Over | 75 (30%) | 39 (35%) | 36 (26%) | ||
| · Under | 23 (9.2%) | 9 (8.1%) | 14 (10%) | ||
Table 1: Sociodemographic and biometric distribution
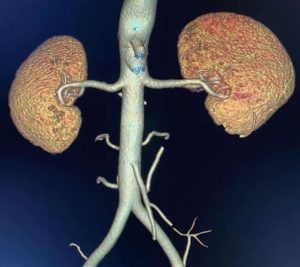
Figure 1: Normal renal arterial supply
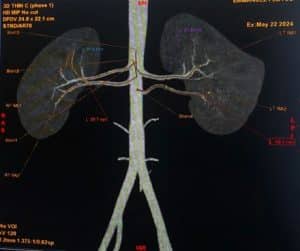
Figure 2: Maximum intensity projection (MIP) image showing bilateral accessory renal arteries
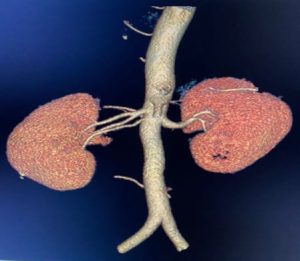
Figure 3: Right accessory renal artery
| Parameter | Overall
N = 250 |
Gender | |
| Female
N = 111 |
Male
N = 139 |
||
| Renal artery variation | |||
| Aberrant origin of renal artery | 6 (2.4%) | 4 (3.6%) | 2 (1.4%) |
| Bilateral accessory renal artery | 8 (3.2%) | 6 (5.4%) | 2 (1.4%) |
| Bilateral renal artery stenosis | 2 (0.8%) | 0 (0%) | 2 (1.4%) |
| Double accessory renal artery | 9 (3.6%) | 2 (1.8%) | 7 (5.0%) |
| Early division of renal artery | 68 (27%) | 28 (25%) | 40 (29%) |
| Normal | 110 (44%) | 46 (41%) | 64 (46%) |
| Single accessory renal artery | 47 (19%) | 25 (23%) | 22 (16%) |
Table 2: Distribution of renal artery variation stratified by gender
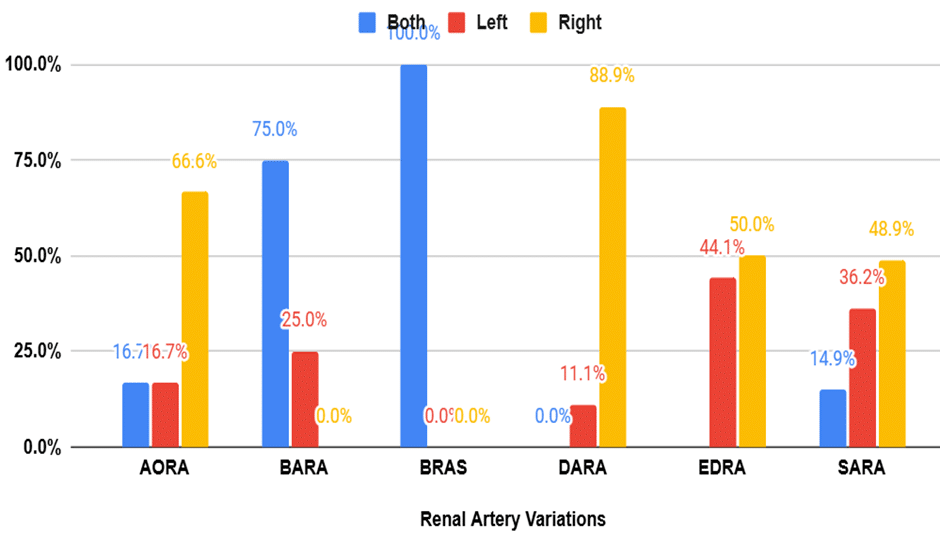
Figure 4: Distribution of the location of renal artery variations
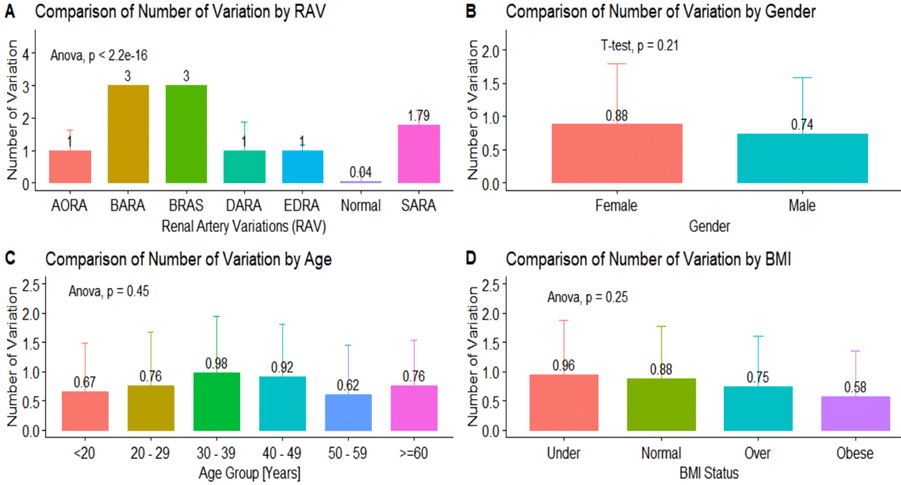
Figure 5: Distribution of the number of variations
(A) Average number of variations by RAV (B) Average number of variations by gender (C) Average number of variations by age (D) Average number of variations by BMI
| Parameter | Overall,
N = 250 |
Age group | |||||
| <20
N = 6 |
≥60
N = 67 |
20 – 29
N = 46 |
30 – 39
N = 42 |
40 – 49
N = 50 |
50 – 59
N = 39 |
||
| AORA | 6 (2.4%) | 0 (0%) | 2 (3.0%) | 1 (2.2%) | 2 (4.8%) | 0 (0%) | 1 (2.6%) |
| BARA | 8 (3.2%) | 0 (0%) | 0 (0%) | 2 (4.3%) | 3 (7.1%) | 2 (4.0%) | 1 (2.6%) |
| BRAS | 2 (0.8%) | 0 (0%) | 0 (0%) | 1 (2.2%) | 0 (0%) | 0 (0%) | 1 (2.6%) |
| DARA | 9 (3.6%) | 1 (17%) | 3 (4.5%) | 0 (0%) | 1 (2.4%) | 3 (6.0%) | 1 (2.6%) |
| EDRA | 68 (27%) | 1 (17%) | 21 (31%) | 11 (24%) | 8 (19%) | 16 (32%) | 11 (28%) |
| Normal | 110 (44%) | 3 (50%) | 28 (42%) | 23 (50%) | 17 (40%) | 18 (36%) | 21 (54%) |
| SARA | 47 (19%) | 1 (17%) | 13 (19%) | 8 (17%) | 11 (26%) | 11 (22%) | 3 (7.7%) |
Table 3: Distribution of renal artery in the stratification of age
Discussion
The study focuses on the critical importance of understanding the variations in renal artery anatomy, especially as the frequency of renal transplants, vascular reconstructions, and various surgical, urological, and radiological interventions continues to increase. This study investigated the prevalence and spectrum of RAV among 250 participants and its correlation with clinical findings, gender, age, and BMI. The overall prevalence of RAV in this study was 56.0%, which is consistent with previous literature. For example, Sampaio and Passos.[13] found a similar incidence of renal artery variants in a comparable group, showing anatomical similarities. These variations can pose challenges during renal surgeries and diagnostic imaging, necessitating careful preoperative assessment and planning. Additionally, the prevalence of RAV has implications for radiologists and clinicians in diagnostic settings. Variations in renal artery anatomy have been associated with increased risks of hypertension and renal insufficiency, further highlighting the importance of recognizing and managing these variations effectively. This is supported by research conducted by Herrmann.[14]
The distribution of specific RAV types in our study showed that EDRA was the most prevalent (27.0%), followed by SARA (19.0%) and BARA (3.2%). Similar trends were observed in a study by Aremu et al.[9], where EDRA was identified as a common variation, particularly in older adults. The age-specific prevalence rates in our study also support this, with the highest proportion of EDRA found in participants aged 40-49 years (32%), and this could be attributed to anatomical and physiological changes that occur with ageing. Moreover, the relatively high occurrence of SARA (19.0%) in our study aligns with findings from previous research. For example, Safaa et al.[15] found that renal artery variants were found in 11% of subjects, with accessory renal arteries in 6% of the study group. The presence of accessory renal arteries can complicate surgical planning and has been linked to increased risks of vascular complications during procedures such as nephrectomy and renal angioplasty.[16]
Gender differences in RAV were notable, with females showing a higher prevalence of AORA (3.6% in females vs. 1.4% in males) and BARA (5.4% in females vs. 1.4% in males). Males, on the other hand, exhibited a higher prevalence of DARA (5.0% in males vs. 1.8% in females) and EDRA (40% in males vs. 25% in females). These findings align with the results of a study by Elrahim[17], which also reported significant gender differences in renal artery anatomy. Such differences are crucial for personalized medical approaches and highlight the need for gender-specific diagnostic protocols. Such differences are crucial for personalized medical approaches and highlight the need for gender-specific diagnostic protocols.[18,19,20]
Recent studies have continued to explore the implications of RAVs in renal diagnostics. For instance, research by Benny et al.[22] demonstrated that the presence of multiple renal arteries can complicate the assessment of renal artery stenosis, potentially leading to both false positives and false negatives in imaging studies. This further underscores the need for high-resolution imaging techniques and the integration of clinical data to improve diagnostic accuracy.
The statistical analysis revealed no significant differences in the number of RAVs based on age, gender, or BMI (p > 0.05), indicating that the presence of RAVs is not influenced by these demographic factors. On the other hand, the spectrum of renal artery variations showed significantly different (p < 0.001). This is supported by Alberto’s findings.[23], who also reported significant variability in the types of RAVs among the population of the study. To further support the findings of this study, similar research has underscored the significance of considering anatomical variations of renal arteries in clinical practice. For example, a study by Mishra et al.[24] emphasized the critical role that understanding these variations plays in surgical planning and patient management, as well as the direct impact it can have on the success of various medical procedures. Furthermore, a better understanding of the environmental factors that contribute to RAVs could inform public health strategies aimed at minimizing risk factors during pregnancy or early development. This knowledge could also guide recommendations for preventive measures that might reduce the likelihood of developing complex renal artery variations, ultimately leading to improved patient outcomes.
The study identified a moderate correlation between RAV and clinical findings, with an agreement rate of 53.2%. This statistic means that just over half of the time, the presence of RAVs aligns with the observed clinical symptoms or outcomes. However, this level of agreement also highlights a key challenge: while RAVs are relatively common, their clinical significance is not always clear-cut. In other words, the presence of RAVs doesn’t consistently predict specific health issues or symptoms in patients. For some individuals, RAVs might be associated with certain renal conditions or complications, while for others, these variations might not have any noticeable impact on their health.[19,20] This variability makes it difficult to draw definitive conclusions about the clinical importance of RAVs based solely on their presence. Clinicians, therefore, need to be cautious when evaluating the significance of RAVs in their patients. The presence of these variations should be considered as part of a broader assessment, rather than being viewed as a definitive indicator of specific renal conditions.
In terms of diagnostic performance, the accuracy, sensitivity, and specificity of detecting abnormal arteries through RAV were 53.02%, 69.5%, and 48.2%, respectively. These figures suggest that while RAV can serve as a potential indicator of underlying renal conditions, they are not highly reliable as a standalone diagnostic marker. The sensitivity of 69.5% indicates that RAVs are relatively good at identifying cases where abnormal arteries are present, but the lower specificity of 48.2% reflects a significant rate of false positives, where RAVs might suggest abnormalities that are not actually present. This finding aligns with comparative studies in the field, such as those conducted by Zhang[21], which reported similar diagnostic performance metrics. It is crucial to consider additional diagnostic tools and clinical assessments to accurately diagnose renal conditions.[22]
Recent studies have continued to explore the implications of RAVs in renal diagnostics. For instance, research demonstrated that the presence of multiple renal arteries can complicate the assessment of renal artery stenosis, potentially leading to both false positives and false negatives in imaging studies. This further underscores the need for high-resolution imaging techniques and the integration of clinical data to improve diagnostic accuracy.[23]
The statistical analysis revealed no significant differences in the number of RAVs based on age, gender, or BMI (p > 0.05). However, the spectrum of renal arteries showed significant variability (p < 0.001), indicating that while the presence of RAVs is not influenced by these demographic factors, the type of variation may be. This is supported by findings from García-Barrios A et al.[24], who also reported significant variability in the types of RAVs among the population of the study. Further supporting the findings of this study, similar research has underscored the significance of considering anatomical variations, particularly in the renal arteries, in clinical practice. For example, a study by Mishra et al.[25] emphasized the critical role that understanding these variations plays in surgical planning and patient management, as well as the direct impact it can have on the success of various medical procedures. Furthermore, a better understanding of the environmental factors that contribute to RAVs could inform public health strategies aimed at minimizing risk factors during pregnancy or early development. This knowledge could also guide recommendations for preventive measures that might reduce the likelihood of developing complex renal artery variations, ultimately leading to improved patient outcomes.
Limitations of the study: One significant limitation of this study is its failure to consider the potential impact of various factors on RAV, such as underlying health conditions (e.g., diabetes, hypertension), genetic predispositions, and other comorbidities.
Conclusion
In conclusion, this study provides a comprehensive analysis of the prevalence and spectrum of renal artery variations (RAV) among 250 participants, highlighting significant gender and age-specific differences. The overall prevalence of RAV was 56.0%, with EDRA and Single SARA being the most common types. The findings emphasize the importance of recognizing these anatomical variations in clinical practice to enhance diagnostic accuracy and improve surgical outcomes. The moderate agreement between RAV and clinical findings suggests that while RAV can indicate underlying renal conditions, it should not be solely relied upon for diagnosis.
References
- Leslie SW, Sajjad H. Anatomy, Abdomen and Pelvis, Renal Artery. StatPearls Publishing; updated August 8, 2023. Anatomy, Abdomen and Pelvis, Renal Artery.
- Siqueira D, Guillaumon A. Angiography for Renal Artery Disease. IntechOpen Publishing; 2019. doi:10.5772/intechopen.79232 Crossref | Google Scholar
- Gulas E, Wysiadecki G, Szymański J, et al. Morphological and clinical aspects of the occurrence of accessory (multiple) renal arteries. Arch Med Sci. 2018;14(2):442-453. doi:10.5114/aoms.2015.55203 PubMed | Crossref | Google Scholar
- Oda O, Zamakhshary M, Namshan MA, Jadaan SA, Shalaan HA. Bowel ischemia in a baby with unspecified renovascular hypertension: a case report. J Med Case Rep. 2011;5:569. doi:10.1186/1752-1947-5-569
PubMed | Crossref | Google Scholar - Karadağ C, Birge O, Bakir MS, et al. Encountering the accessory polar renal artery during retroperitoneal lymphadenectomy. Clin Case Rep. 2020;9(1):177-179. doi:10.1002/ccr3.3493 Crossref | Google Scholar
- Burihan MC, Garbelotti SA, Cândido PL, et al. Multiple variation of right renal and gonadal vascularization: report of two cases. J Vasc Bras. 2024;23:e20230044. doi:10.1590/1677-5449.202300442 PubMed | Crossref | Google Scholar
- Baradhi KM, Bream P. Fibromuscular Dysplasia. StatPearls Publishing; 2023. Fibromuscular Dysplasia
- Akpunonu B. Bilateral accessory (aberrant) renal arteries associated with uncontrolled hypertension: role of renin-angiotensin-aldosterone antagonist drugs for treatment goal. Transl Univ Toledo J Med Sci. 2023;11(1):e1-1. doi:10.46570/utjms.vol11-2023-669 Crossref | Google Scholar
- Aremu A, Igbokwe M, Olatise O, Lawal A, Maduadi K. Anatomical variations of the renal artery: a computerized tomographic angiogram study in living kidney donors at a Nigerian Kidney Transplant Center. Afr Health Sci. 2021;21(3):1155-1162. doi:10.4314/ahs.v21i3.24 PubMed | Crossref | Google Scholar
- Arévalo Pérez J, Gragera Torres F, Marín Toribio A, et al. Angio CT assessment of anatomical variants in renal vasculature: its importance in the living donor. Insights Imaging. 2013;4(2):199-211. doi:10.1007/s13244-012-0217-5
PubMed | Crossref | Google Scholar - Cho ES, Yu JS, Ahn JH, et al. CT angiography of the renal arteries: comparison of lower-tube-voltage CTA with moderate-concentration iodinated contrast material and conventional CTA. AJR Am J Roentgenol. 2012;199(1):96-102. doi:10.2214/AJR.11.7450 PubMed | Google Scholar
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. 2013;310(20):2191-2194. doi:10.1001/jama.2013.281053 PubMed | Crossref | Google Scholar
- Sampaio FJ, Passos MA. Renal arteries: anatomic study for surgical and radiological practice. Surg Radiol Anat. 1992;14(2):113-117. doi:10.1007/BF01794885 PubMed | Crossref | Google Scholar
- Herrmann SM, Textor SC. Renovascular Hypertension. Endocrinol Metab Clin North Am. 2019;48(4):765-778. doi:10.1016/j.ecl.2019.08.007 PubMed | Crossref | Google Scholar
- Mohammed S, AbdAlla E, Elhag A, et al. The prevalence of accessory renal arteries in Sudanese population in Khartoum State: a cross-sectional CT study from 2017 to 2020. BMC Nephrol. 2024;25:135. doi:10.1186/s12882-024-03573-3 PubMed | Crossref | Google Scholar
- Wu F, Yuan X, Sun K, et al. Effect of accessory renal arteries on essential hypertension and related mechanisms. J Am Heart Assoc. 2024;13(4):e030427. doi:10.1161/JAHA.123.030427 PubMed | Crossref | Google Scholar
- Abd Elrahim E. Computed tomography evaluation of renal artery morphometry in adults: the impact of age and gender. Saudi Med J. 2020;41(1):34-37. doi:10.15537/smj.2020.1.24795 PubMed | Crossref | Google Scholar
- Olin JW, Gornik HL, Bacharach JM, et al. Fibromuscular dysplasia: state of the science and critical unanswered questions: a scientific statement from the American Heart Association. Circulation. 2014;129(9):1048-1078. doi:10.1161/01.cir.0000442577.96802.8c PubMed | Crossref | Google Scholar
- Gounden V, Bhatt H, Jialal I. Renal Function Tests. StatPearls Publishing; updated July 27, 2024. Renal Function Tests
- Luyckx VA, Tonelli M, Stanifer JW. The global burden of kidney disease and the sustainable development goals. Bull World Health Organ. 2018;96(6):414-422D. doi:10.2471/BLT.17.206441 PubMed | Crossref | Google Scholar
- Zhang HL, Sos TA, Winchester PA, et al. Renal artery stenosis: imaging options, pitfalls, and concerns. Prog Cardiovasc Dis. 2009;52(3):209-219. doi:10.1016/j.pcad.2009.10.003 PubMed | Crossref | Google Scholar
- Rountas C, Vlychou M, Vassiou K, et al. Imaging modalities for renal artery stenosis in suspected renovascular hypertension: prospective intraindividual comparison of color Doppler US, CT angiography, GD-enhanced MR angiography, and digital subtraction angiography. Ren Fail. 2007;29(3):295-302. doi:10.1080/08860220601166305
PubMed | Crossref | Google Scholar - Drieghe B, Madaric J, Sarno G, et al. Assessment of renal artery stenosis: side-by-side comparison of angiography and duplex ultrasound with pressure gradient measurements. Eur Heart J. 2008;29(4):517-524. doi:10.1093/eurheartj/ehm631 PubMed | Crossref | Google Scholar
- García-Barrios A, Cisneros-Gimeno AI, Celma-Pitarch A, Whyte-Orozco J. Anatomical study about the variations in renal vasculature. Folia Morphol (Warsz). doi:10.5603/FM.a2023.0039 PubMed | Crossref | Google Scholar
- Mishra GP, Bhatnagar S, Singh B. Anatomical variations of upper segmental renal artery and clinical significance. J Clin Diagn Res. 2015;9(8):AC01-3. doi:10.7860/JCDR/2015/12326.6280 PubMed | Crossref | Google Scholar
Acknowledgments
Not reported
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Information
Corresponding Author:
Abdullahi Alfa
Department of Radiology
Afe Babalola Multisystem Hospital, Nigeria
Email: abdullahialfa48@gmail.com
Co-Authors:
Linus Anderson Enye
Department of Human Anatomy
Afe Babalola University, Nigeria
Nicholas Kayode Irurhe
Department of Radiology
Lagos University Teaching Hospital, Nigeria
Muzammil Aminu
Department of Radiology
Afe Babalola Multisystem Hospital, Ekiti State, Nigeria
Ikechukwu Emmanuel Onwunta, Ikenna Caleb Onyemauwa, Stephen Chukwuebuka Ugwuobu
Department of Radiography and Radiological Science
University of Nigeria, Nigeria
Ebube Lilian Agbo
Department of Biochemistry
Alex Ekwueme Federal University, Ebonyi State, Nigeria
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing – original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
This study adhered to the ethical standards outlined in the 1964 Declaration of Helsinki. Ethical approval was obtained from the Ethics and Research Committee of Afe Babalola University, Ado Ekiti, College of Health Sciences (ABUAD) under the reference number ABUADHREC/26/06/2024/490, and from the Afe Babalola Multi-System Hospital, Ado Ekiti (AMSH) under the reference number AMSH/REC/24/069. Additionally, permission and support were granted by the managing consultants of the Radiology Department. Informed written consent was obtained from all participants before the examination, during which the purpose of the study, potential benefits, possible risks, and the steps involved in the examination were clearly explained.
Conflict of Interest Statement
The author declares no conflict of interest.
Guarantor
None
DOI
Cite this Article
Alfa A, Enye LA, Irurhe NK, et al. Evaluation of Renal Artery Variations Among Patients Undergoing Abdominal Computed Tomography in an African Population. medtigo J Med. 2025;3(1):e30623120. doi:10.63096/medtigo30623120 Crossref









