Author Affiliations
Abstract
Epilepsy represents a widely recognized neurological disorder that necessitates the administration of antiepileptic drugs for its treatment. Although these pharmacological agents are proficient in seizure management, their utilization is often correlated with a spectrum of adverse drug effects that profoundly influence patient adherence and overall quality of life. The primary aim of this investigation was to assess the incidence, severity, and determinants of adverse drug effects associated with antiepileptic drugs (AEDs) among individuals seeking treatment at Pakistan Institute of Medical Sciences (PIMS) Hospital and Pakistan Ordnance Factories (POF) Hospital. A cross-sectional observational research design was employed from December 2024 to March 2025 within the neurology department of the PIMS and POF hospitals. A cohort of 60 patients diagnosed with epilepsy, who have been receiving AED therapy, was selected through systematic random sampling. Data collection was facilitated through structured interviews and validated questionnaires, followed by statistical analysis utilizing SPSS version 26.0. The most frequently reported adverse events included dizziness (61.7%), fatigue (58.3%), and muscle weakness (68.3%). Noteworthy statistical correlations were observed between the implementation of combination therapy and an increased incidence of dizziness (p = 0.012), weight gain (p = 0.015), and severe allergic reactions (p = 0.032). Logistic regression analysis identified severe dizziness (odds ratio (OR) = 3.1, p = 0.004) and age exceeding 45 years (OR = 2.4, p = 0.018) as significant predictors for discontinuation of therapy. Adverse drug effects are notably prevalent among patients who are on polytherapy. These findings emphasize the necessity for personalized treatment regimens for high-risk groups and vigilant monitoring to optimize therapeutic adherence and enhance patient outcomes.
Keywords
Epilepsy, Anti-epileptic drugs, Adverse drug effects, Polytherapy, Seizure severity, Neurological disorder, Side effects.
Introduction
Epilepsy, also referred to as a seizure disorder, is a disorder of the brain characterized by frequent and repeated seizures.[1] Epilepsy is generally the hyperexcitation of neurons in which a single stimulation results in a series of repeating action potentials without a refractory period, rather than a singular action potential.[2] This occurs because of an imbalance between inhibitory and excitatory neurotransmitters.[3] This disrupts the normal functioning of the brain regions involved, resulting in the clinical manifestations of the particular kind of epilepsy.[4] With an estimated 50 million cases globally, epilepsy contributes significantly to the global disease burden. The estimated prevalence of active epilepsy ranges from 4 to 10 per 1,000 individuals at any particular moment. Epilepsy affects almost 5 million individuals worldwide annually. The annual incidence of epilepsy in developed countries is around 49 cases per 100,000 individuals. This number may reach 139 per 100,000 in developing countries.[5] According to the Centers for Disease Control and Prevention, in 2021 and 2022, around 2.9 million U.S. people aged 18 and older reported experiencing epilepsy.[6] Epilepsy is the most common chronic disorder of neurons, impacting millions annually.[7] Approximately 50 million individuals globally have epilepsy, with Pakistan exhibiting an estimated frequency of 9.99 per 1,000 inhabitants. The largest frequency is seen in persons under 30, with around 2 million cases.[8,9]
According to a population-based epidemiological study of epileptic patients in Pakistan, a potential etiology was identified in 38.4% of cases: 32% had a positive family history of epilepsy, mostly among siblings. Commonly identified triggers were fever (29.2%) and emotional problems (16.6%). Merely 3% of individuals with epilepsy attributed their condition to supernatural origins.[10] The primary therapy for epilepsy usually involves AEDs.[11] These drugs mitigate seizures by diminishing the abnormal electrical discharge in the brain that precipitates them. The prescription AED will be dependent upon the epilepsy type, seizure frequency and intensity, and the patient’s general health status.[12] The objective of therapy for patients with epilepsy is to attain a seizure-free condition without adverse effects at a minimum cost of treatment.[13,14] Approximately 60-70% of patients on AEDs can achieve their treatment goals.[15]
Different types of antiseizure medications are now available, each with varying advantages and adverse effects. The majority of seizures can be managed with a single medication. The concomitant use of drugs may exacerbate adverse effects, including fatigue and dizziness. Nevertheless, combinations of medications are sometimes required for certain epileptic patients who are not treated by single-line therapy.[16] A medicine with low efficacy but a favorable safety profile may seem more advantageous than a treatment with higher efficacy but causes clinically significant side effects.[17]
The extended retention of patients on their antiepileptic drug treatment is now recognized as a definitive indicator of clinical efficacy. It serves as a significant cumulative assessment of both effectiveness and safety profile over time.[18] Behavioral and cognitive side effects are the most common side effects of AEDs. Phenytoin and valproic acid cause more weight gain than carbamazepine. Careful monitoring of patients who are on polytherapy is required because of frequent behavioral side effects.[19]
The newer medications like topiramate, tiagabine, oxcarbazepine, levetiracetam, and zonisamide may exhibit improved tolerability, reduced drug interactions, and seem to alter cognitive functioning to a lower degree than older medications.[20] Lamotrigine also has a safer adverse effect profile than carbamazepine.[21] Lamotrigine (LTG) is extensively approved for use as an adjunctive therapy and as monotherapy for partial and generalized seizures in pediatric and adult patients.[22] A randomized double-blind research study including individuals with recent-onset epilepsy reported no therapeutic difference between lamotrigine and carbamazepine, but LTG has better tolerability.[23]
Timely recognition of these AEs improves patient safety, compliance, and overall quality of life. This also relates to economic benefits, as it reduces medical expenses and prevents unnecessary hospitalizations. Additionally, proper drug monitoring ensures that each patient receives personalized treatment, thereby enhancing efficacy and safety while minimizing side effects.[24] Moreover, evaluating high-risk groups can help prevent severe issues early. Therefore, the ultimate purpose of this research was to determine patient-reported adverse effects associated with AEDs and identify which factors were associated with these complications among patients at PIMS Hospital and POF Hospital, Islamabad.[25,26]
Methodology
Study area and duration: The study was conducted at the neurology outpatient department of PIMS and POF, two primary tertiary care teaching hospitals located in Islamabad and Wah Cantt, Pakistan, respectively. These hospitals serve as specialized care institutions for a large population from Islamabad, Rawalpindi, Wah Cantt, and adjacent localities. They comprise over 1000 beds across various inpatient and outpatient departments. The neurology departments at PIMS & POF manage a substantial number of patients with neurological disorders, including epilepsy. Data collection was conducted over three months, from 15 December 2024 to 15 March 2025.
Study design and subject: This was a cross-sectional study conducted in a hospital setting. Adult patients with epilepsy who were on AED treatment and attended routine follow-up in the neurology department in the study period constituted the study population. Patients were considered cases if they had received an AED within the past year. They were divided into two groups: patients with AEDs for ≤1 year and those with AEDs for >1 year. Exclusion criteria were incomplete records, AEDs for <1 year, and uncooperative or refusing patients.
Sample size and sampling method: A cohort of 60 patients was included in this study. A systematic random sampling technique was used to mitigate the selection bias and enhance the validity of the results. This ensured the accurate representation of the target demographic and increased the reliability of findings for our cross-sectional study design.
Inclusion criteria: A confirmed diagnosis of epilepsy alongside concurrent administration of anti-epileptic drug pharmacotherapy.
Exclusion criteria: Patients who opted out of participation or were incapable of providing informed consent. This methodology promotes a systematic, impartial recruitment process appropriate for hospital-based clinical settings.
Data collection procedures: Data were collected through a structured, interviewer-administered questionnaire. It was specifically designed for this investigation, alongside an assessment of their medical records. The questionnaire encompassed both open-ended and closed-ended questions addressing critical domains. These included demographic information, type of epilepsy, AED regimen, comorbidity, and any documented AEDs within the previous three months.
Patients were approached individually. The data was collected through face-to-face interviews. Also, the interviews were conducted in confidential settings to encourage accurate and truthful responses. This process was performed by trained pharmacy students or clinical personnel under the supervision of the principal investigator. All data collectors underwent a comprehensive one-day training session. Interviews were conducted in the patient’s preferred language, either Urdu or English. The gathered data was then evaluated for completeness, accuracy, and consistency. After that, it was entered into a secure database designated for analysis.
Ethical consideration: Ethical approval was obtained through a letter of permission from the respective bodies of PIMS and POF. Patients were informed of the study’s objectives, and informed consent was obtained. Patient confidentiality and participant anonymity were strictly maintained throughout the research process. No personal identifiers, such as names and addresses, were recorded on the data collection forms, and all information was handled in accordance with ethical research standards to protect patient privacy.
Results
Socio-demographic profile of study participants: A total of 60 participants diagnosed with epilepsy were included in this comparative evaluation of the adverse effects of AEDs. Most participants were aged <30 years (47%, n = 24), with males constituting 53% (n = 27) of the cohort. Only 8% of the participants were employed. 83% of patients were unemployed. Demographic characteristics of the patients are given in Table 1 below.
| Variables | Categories | Frequency | Percent | Valid percent | Cumulative percent |
| Age | <15years 16-30 years 30-40 years 45-65 years >65 years |
13 20 15 11 1 |
21.7 33.3 25.0 18.3 1.7 |
21.7 33.3 25.0 18.3 1.7 |
21.7 55.0 80.0 98.3 100 |
| Total | 60 | 100.0 | 100.0 | ||
| Gender | Male Female |
36 24 |
60 40 |
60 40 |
60.0 100.0 |
| Weight | <15kg 16-35kg 36-50kg 51-75kg 76-90kg >90kg |
6 9 9 27 8 1 |
10.0 15.0 15.0 45.0 13.3 1.7 |
10.0 15.0 15.0 45.0 13.3 1.7 |
10.0 25.0 40.0 85.0 98.3 100.0 |
| Marital status | Married Single Other(divorced/widow) |
13 32 15 |
21.7 53.3 25.0 |
21.7 53.3 25.0 |
21.7 75.0 100.0 |
| Level of education completed. | Primary level High school /equivalent Bachelor’s degree Master’s level or higher Don’t want to answer |
20 11 5 5 19 |
33.3 18.3 8.3 8.3 31.7 |
33.3 18.3 8.3 8.3 31.7 |
33.3 51.6 59.9 68.2 100.0 |
| Employment status | Employed Unemployed Retired Other |
5 51 1 3 |
8.3 85.0 1.7 5.0 |
8.3 85.0 1.7 5.0 |
8.3 93.3 95.0 100.00 |
Table 1: Frequency, percent, and valid percent distribution of socio-demographic among the variables receiving anti-epileptic drugs
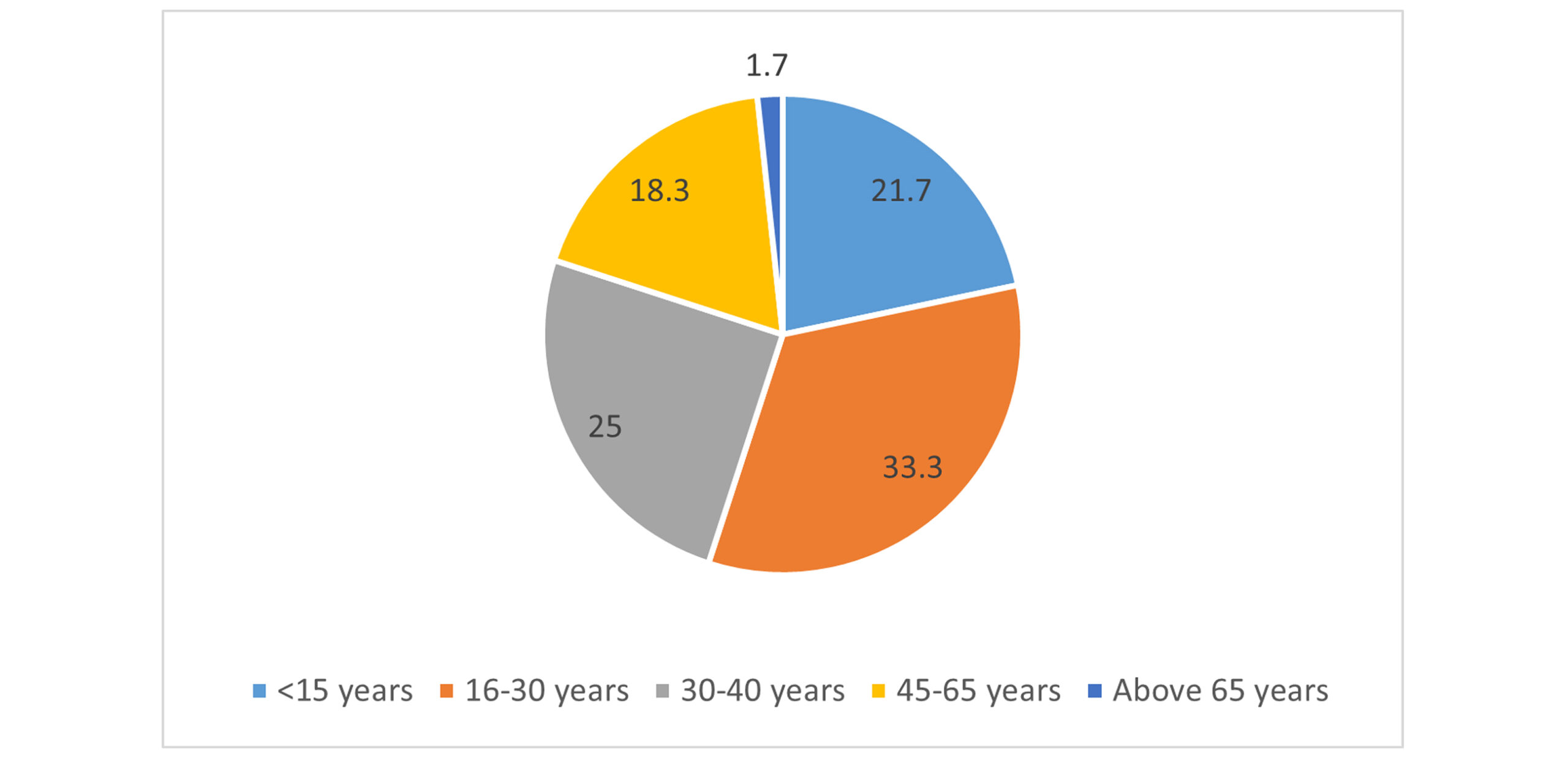
Figure 1: Age distribution of study cohort (n = 60)
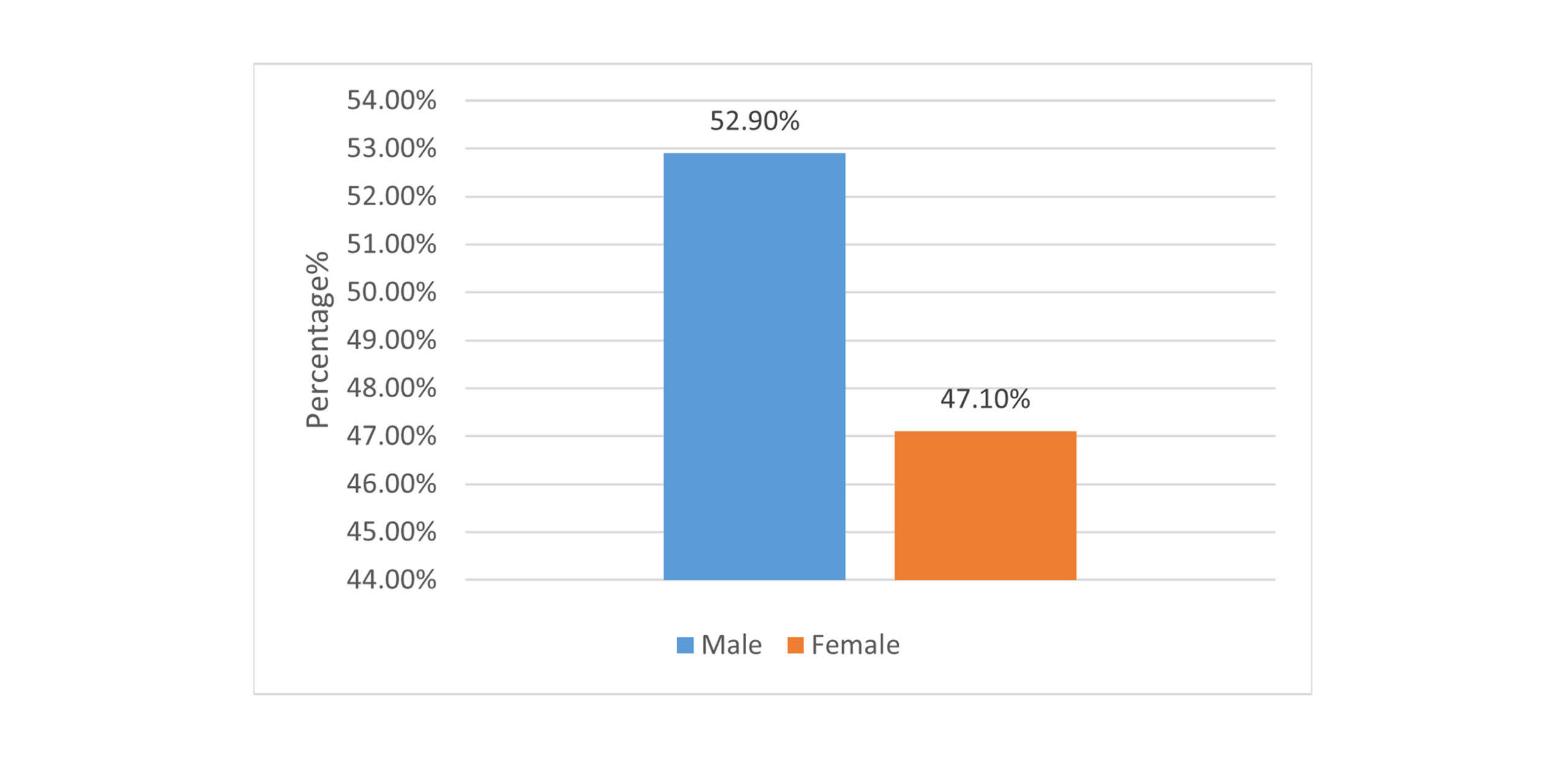
Figure 2: Gender distribution of study cohort (n = 60)
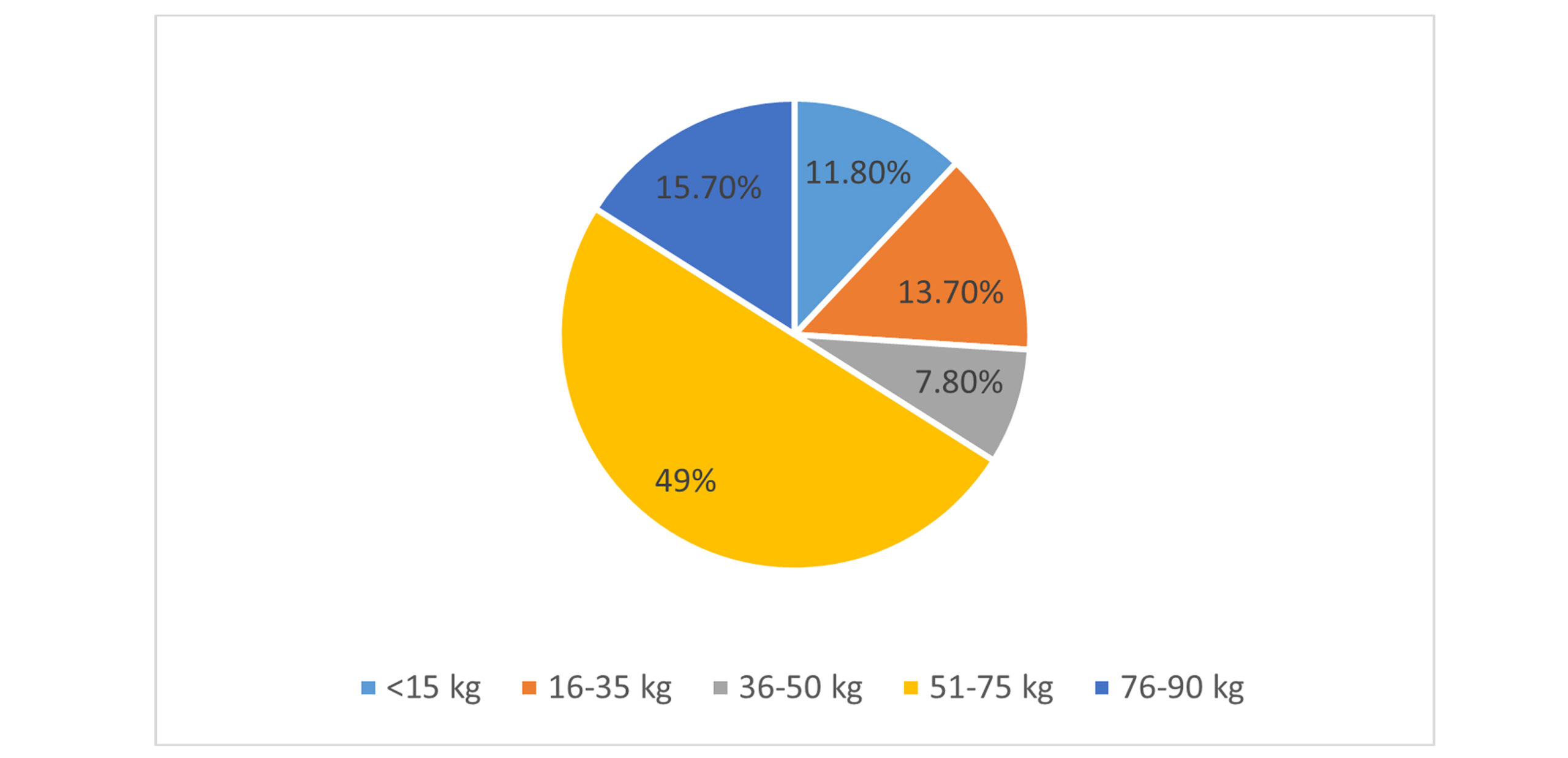
Figure 3: Weight categories in study population (n = 60)
Clinical characteristics of the study cohort: 15 patients in the cohort have a family history of epilepsy. Only one person has epilepsy due to the use of drugs. Most of the participants (n = 27) have been suffering from epilepsy for more than 10 years. 1/4 of the participants (n = 15) experience more than 1 seizure per day. Twenty-four patients are experiencing a severe type of seizure, and 19 are suffering from a mild type.
| Variable | Category | Frequency |
| Family history |
Yes No Unsure |
15 38 7 |
| Root cause |
Brain injury or trauma Alcohol intake or drug use Genetic factors Metabolic disorders Congenital brain malfunctions |
18 1 15 12 14 |
| Duration of epilepsy |
Less than 1 year 1-5 years 6-10 years More than 10 years |
7 12 14 27 |
| Current seizure frequency |
Less than one seizure per month 1-2 seizures per month 1-2 seizures per week 1 seizure per day More than one seizure per day |
7 13 15 10 15 |
| Severity of seizure |
Mild Moderate Severe Variable |
19 13 24 4 |
| Current AED therapy | Yes No |
60 0 |
| Monotherapy /combination therapy Monotherapy/ combination therapy |
Monotherapy
Combination therapy |
33 27 |
| Duration of therapy | Less than 1 year 1-3 years 3-6 years More than 6 years |
7 8 16 29 |
Table 2: Clinical characteristics of the study cohort (N = 60)
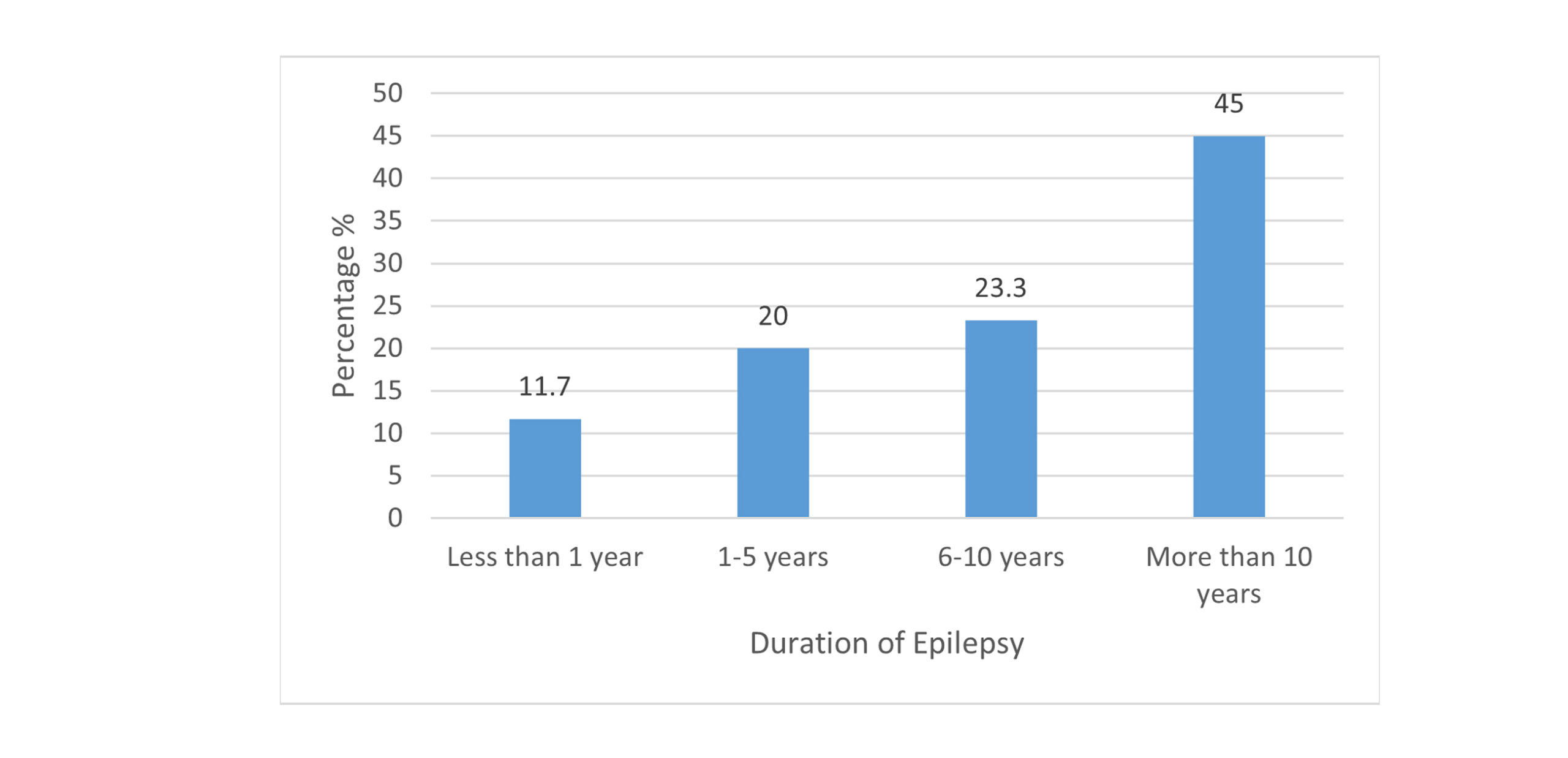
Figure 4: Duration history of epilepsy in selected patients
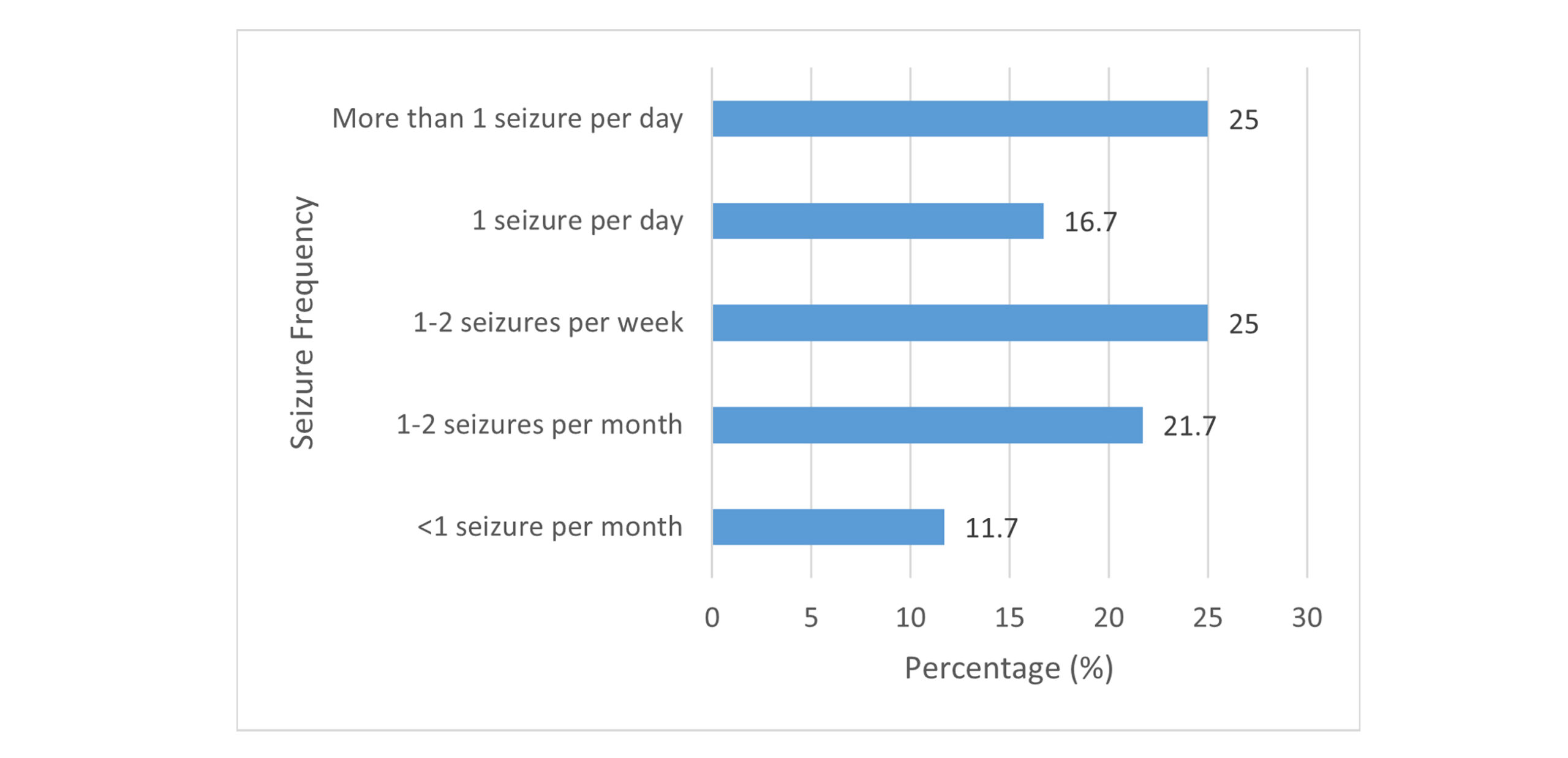
Figure 5: Current seizure frequency in selected patients
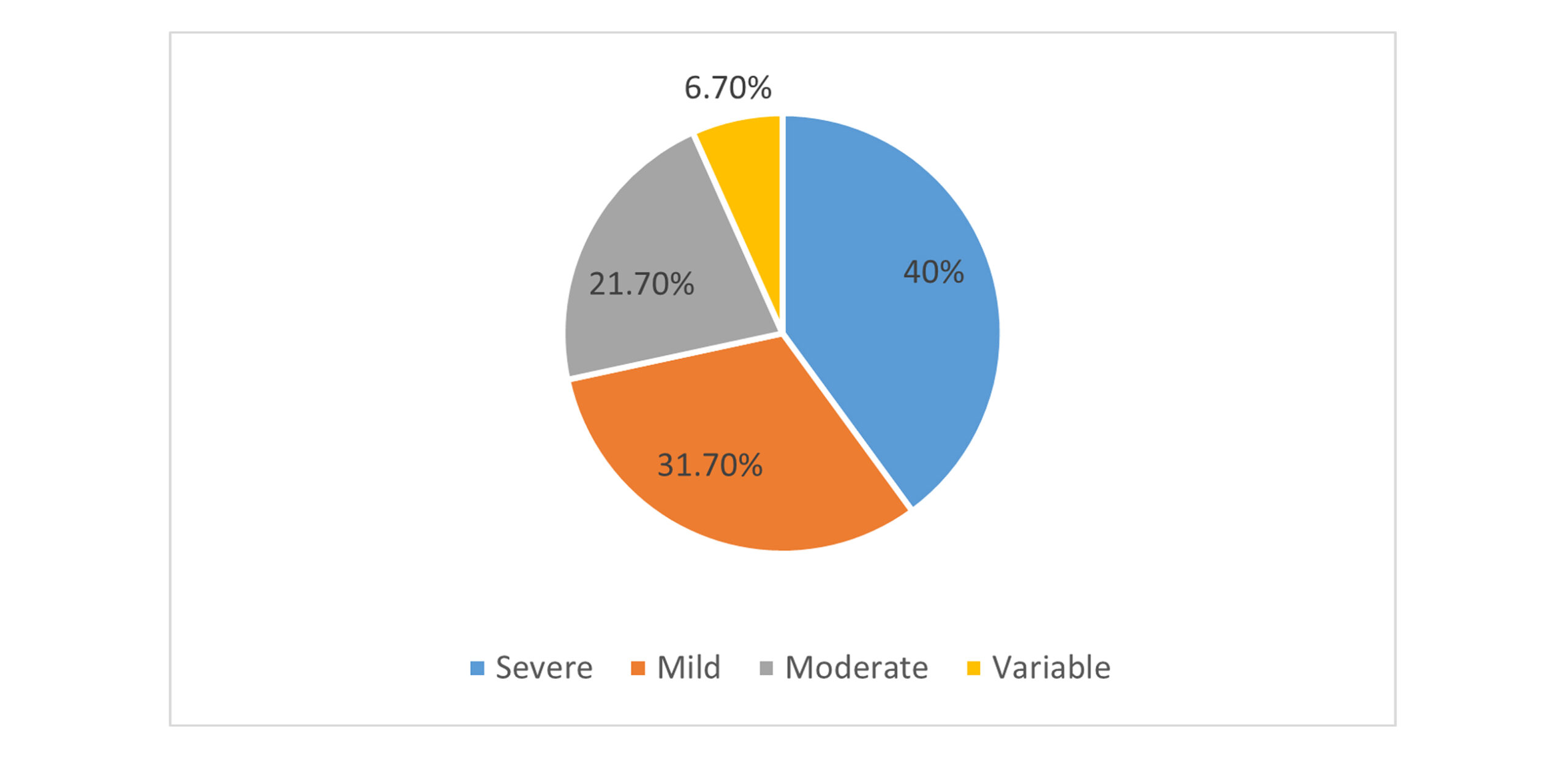
Figure 6: Categories of severity of seizures in selected patients
Therapeutic regimen of study participants: Most participants (55%, n = 33) are on monotherapy, while (45%, n = 27) receive combination therapy. The most prescribed drug is carbamazepine in patients who are on monotherapy (n=11). Zonisamide and clobazam are the drugs that are mostly prescribed together in patients on polytherapy (n=9). Divalproex sodium is the drug that is mostly prescribed in combination with other AEDs. The drugs prescribed to patients in polytherapy, and monotherapy are given below in table 3.
| AEDs prescribed | Frequency | Percent | Valid percent | Cumulative percent |
| Carbamazepine | 11 | 18.3 | 18.3 | 18.3 |
| Carbamazepine +brivaracetam | 2 | 3.3 | 3.3 | 21.7 |
| Carbamazepine + divalproex sodium | 2 | 3.3 | 3.3 | 25.0 |
| Oxcarbazepine + divalproex sodium | 1 | 1.7 | 1.7 | 26.7 |
| Clobazam | 4 | 6.7 | 6.7 | 33.3 |
| Clobazam + zonisamide | 9 | 15.0 | 15.0 | 48.3 |
| Divalproex sodium + levetiracetam | 1 | 1.7 | 1.7 | 50.0 |
| Divalproex sodium | 5 | 8.3 | 8.3 | 58.3 |
| Divalproex sodium + carbamazepine + levetiracetam | 5 | 8.3 | 8.3 | 66.7 |
| Divalproex sodium +levetiracetam | 4 | 6.7 | 6.7 | 73.3 |
| Gabapentin | 2 | 3.3 | 3.3 | 76.7 |
| Lamotrigene | 1 | 1.7 | 1.7 | 78.3 |
| Levetiracetam | 4 | 6.7 | 6.7 | 85.0 |
| Levetiracetam + divalproex sodium + clonazepam | 2 | 3.3 | 3.3 | 88.3 |
| Levetiracetam + oxcarbazepine | 1 | 1.7 | 1.7 | 90 |
| Zonisamide | 6 | 10 | 10 | 100.0 |
| Total | 60 | 100.0 | 100.0 | – |
Table 3: Pharmacological agents prescribed for epileptic patients along with their respective frequencies
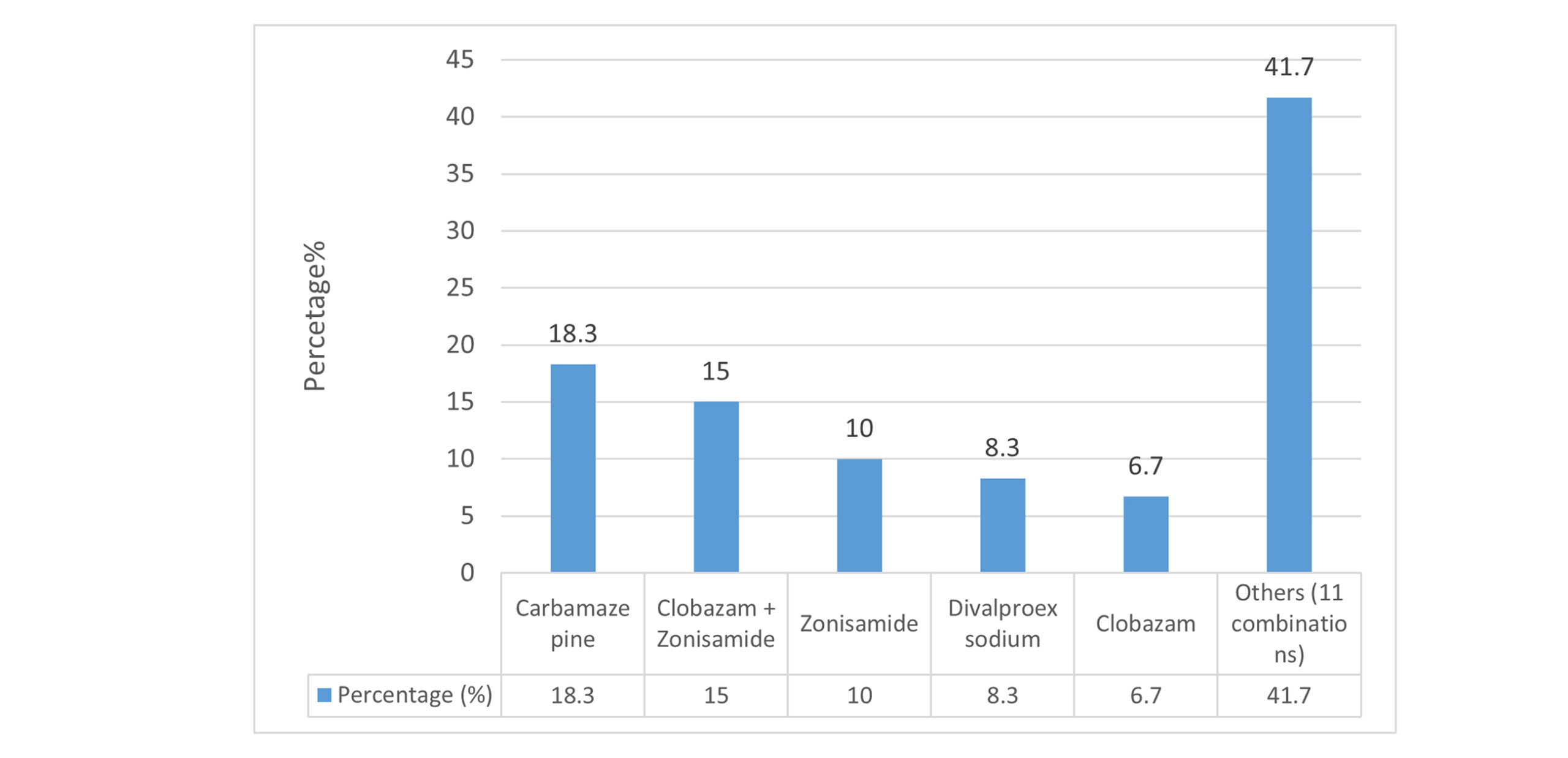
Figure 7: Most frequently prescribed AEDs to selected patients
Incidence of side effects: Among all the side effects, neurological side effects were predominant, with 62% reporting dizziness (44% moderate severity) and 58% experiencing fatigue or drowsiness (52% mild severity). Gastrointestinal effects included nausea/vomiting (34%) and constipation (28%). Musculoskeletal complaints, such as muscle weakness (68%), were frequently reported, while severe allergic reactions (e.g., difficulty breathing) occurred in 12% of participants.
| Side effect/variable | Yes (n) | Incidence (%) |
| Dizziness | 37 | 61.7% |
| Fatigue / Drowsiness | 35 | 58.3% |
| Nausea / Vomiting | 20 | 33.3% |
| Constipation | 17 | 28.3% |
| Musculoskeletal complaints (muscle weakness) | 41 | 68.3% |
| Severe allergic reactions (e.g., difficulty breathing) | 7 | 11.7% |
Table 4: Incidence of the most common side effects reported
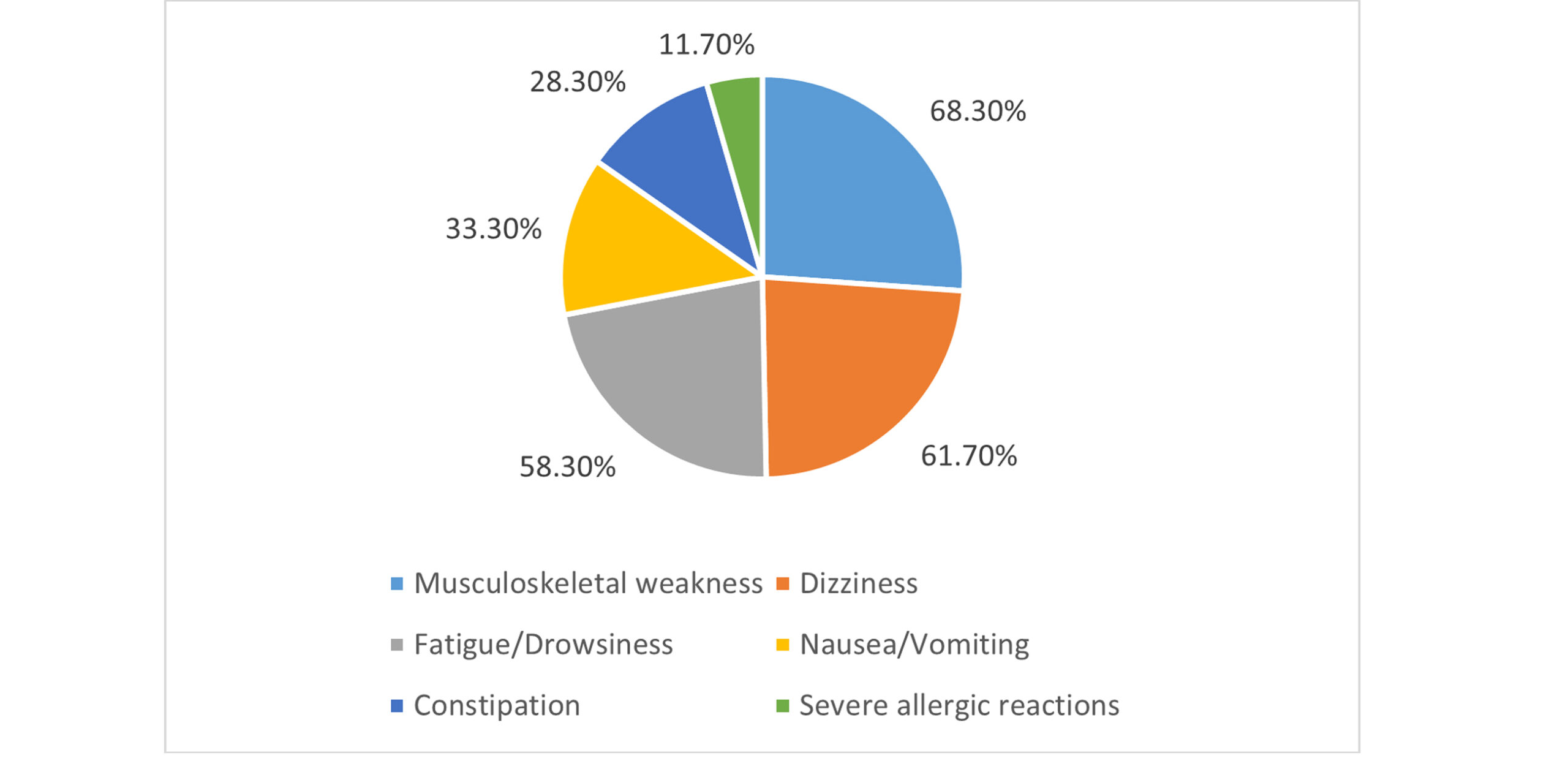
Figure 8: Incidence of common adverse events reported by study participants
Prevalence of adverse effects in monotherapy vs. combination therapy: Table 5 summarizes the comparative analysis of the prevalence of the adverse effects associated with monotherapy vs combination therapy in the context of epilepsy management. The data indicates that the combination therapy is associated with markedly higher frequencies of dizziness (82% compared to 58%), weight gain (64% vs 38%), and severe allergic reactions (18% in contrast 6%) when juxtaposed with monotherapy, with each comparison yielding statistically significant p-values.
| Adverse effect | Monotherapy (%) | Combination therapy (%) | p-value |
| Dizziness | 58 | 82 | 0.012 |
| Weightgain | 38 | 64 | 0.015 |
| Severe allergic reactions | 6 | 18 | 0.032 |
Table 5: Prevalence of adverse effects in monotherapy vs. combination therapy
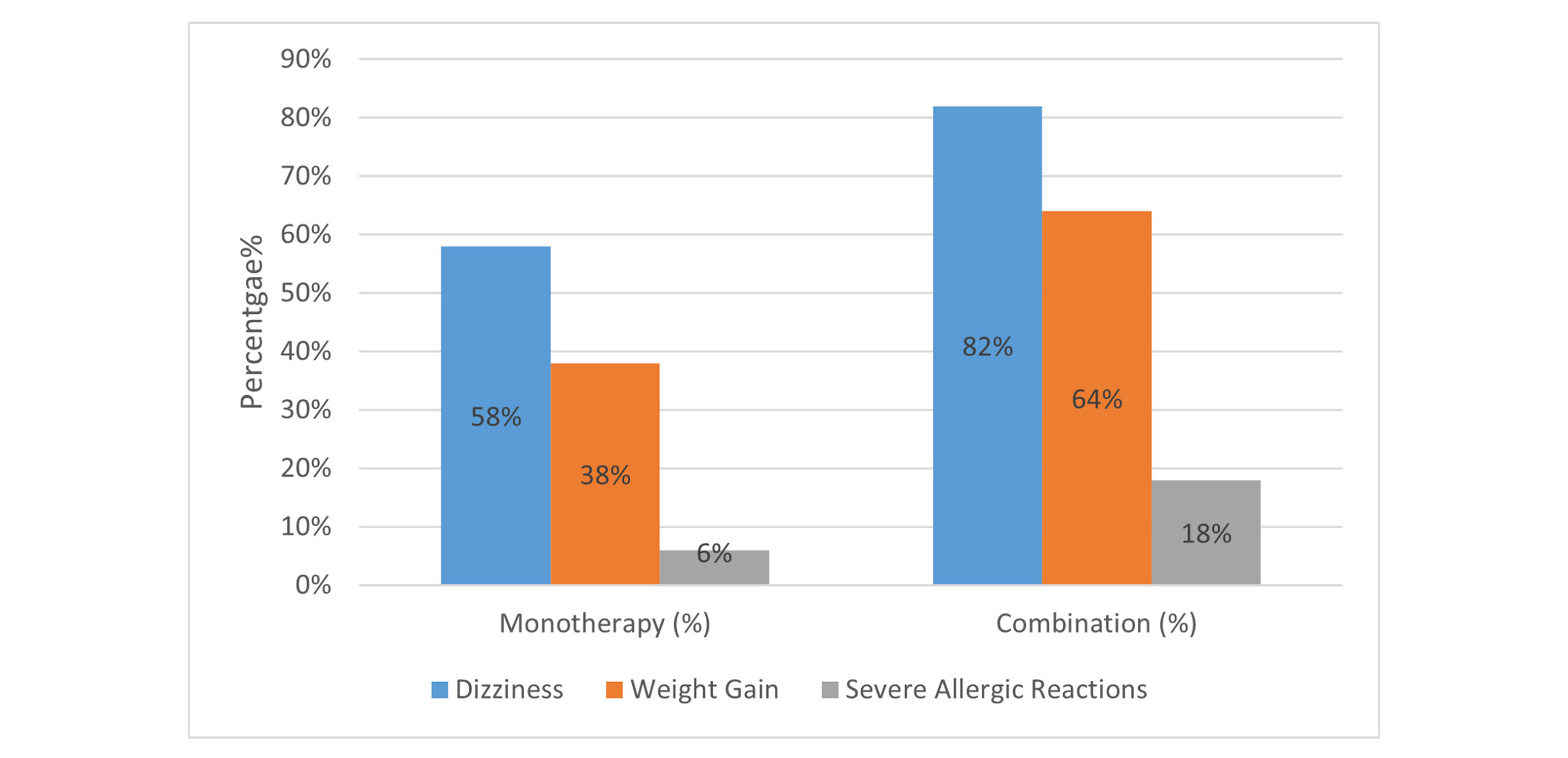
Figure 9: Association of prevalence of adverse effects in monotherapy vs. combination therapy
Association between various clinical variables: Table 6 summarizes the outcomes of Chi-square tests conducted to investigate associations among clinical variables. It delineates a significant correlation between the type of AED and weight gain, gender, and dizziness, as well as other clinical factors such as employment status and seizure frequency. However, certain variables, including education level and age group, did not yield statistically significant associations.
| Variable/Question | Chi-square (χ²) | Degrees of freedom | p-value | Significance (α=0.05) |
| Is there an association between AED type (mono/combination) and weight gain? | 6.82 | 1 | 0.009 | Significant |
| Is there an association between gender and dizziness? | 4.15 | 1 | 0.042 | Significant |
| Is there an association between AED type and severe allergic reactions? | 8.24 | 1 | 0.002 | Significant |
| Does employment status correlate with seizure frequency? | 7.89 | 3 | 0.048 | Significant |
| Is the family history of epilepsy linked to genetic root causes? | 9.32 | 2 | 0.009 | Significant |
| Does therapy duration correlate with side effect severity? | 10.45 | 3 | 0.015 | Significant |
| Is there an association between education level and AED discontinuation? | 4.56 | 2 | 0.102 | Not Significant |
| Does marital status correlate with cognitive issues? | 2.34 | 2 | 0.310 | Not Significant |
| Is the age group associated with AED type (mono/combination)? | 5.12 | 4 | 0.163 | Not Significant |
| Are musculoskeletal effects linked to AED type? | 3.78 | 1 | 0.052 | Marginal |
Table 6: Summary of chi-square tests for associations between clinical variables
Association between demographic factors and treatment variables: Table 7 summarizes key statistical analyses about the association between demographic and treatment variables with ADEs of AEDs. Chi-square tests revealed significant associations between gender and dizziness as a side effect. Females reported dizziness more frequently than males (χ² = 6.24, p = 0.012). Combination therapy was linked to higher fatigue rates compared to monotherapy (χ² = 4.87, p = 0.027). The Mann-Whitney U test indicated a significant gender-based difference in seizure severity. Males experienced moderate episodes of seizures, whereas females reported severe episodes (U = 1023, p = 0.043). The Kruskal-Wallis test demonstrated that longer AED therapy duration correlated with increased dizziness severity (H (3) = 9.67, p = 0.022). Logistic regression identified severe dizziness (odds ratio [OR] = 3.1, p = 0.004) and age >45 years (OR = 2.4, p = 0.018) as significant predictors of AED discontinuation. Subgroup analysis highlighted that combination therapy users had higher rates of weight gain (64% vs. 38%, χ² = 5.92, p = 0.015) and severe allergic reactions (18% vs. 6%, Fisher’s exact test, p = 0.032) as compared to monotherapy. All of this underscores the need for tailored monitoring in high-risk groups.
| Test applied | Sample question | Test statistic | p-value | Significance (α = 0.05) | Effect size / Notes |
| Chi-square test | Is dizziness associated with gender? | χ² = 6.24, df = 1 | 0.012 | Significant | Cramer’s V ≈ 0.22 (small–moderate) |
| Is fatigue associated with therapy type (mono vs. combination)? | χ² = 4.87, df = 1 | 0.027 | Significant | Combination therapy → higher fatigue | |
| Is weight gain associated with therapy type? | χ² = 5.92, df = 1 | 0.015 | Significant | Combination: 64% vs. 38% | |
| Fisher’s exact test | Are severe allergic reactions associated with therapy type? | Fisher’s Exact Test | 0.032 | Significant | Combination: 18% vs. 6% |
| Mann-Whitney U test | Do males and females differ in seizure severity? | U = 1023 | 0.043 | Significant | Males: Median = Moderate; Females: Median = Severe |
| Kruskal-Wallis test | Does therapy duration affect dizziness severity? | H(3) = 9.67 | 0.022 | Significant | Longer therapy → higher dizziness severity |
| Logistic regression | Predictors of AED discontinuation: Severe dizziness | OR = 3.1 | 0.004 | Significant | Strong predictor |
| Predictors of AED discontinuation: Age >45 years | OR = 2.4 | 0.018 | Significant | Older adults are more likely to discontinue | |
| Predictors of severe allergic reactions: Combination therapy | OR = 3.8 | 0.002 | Significant | Nagelkerke R² = 0.31 | |
| Predictors of severe allergic reactions: Age >45 years | OR = 2.2 | 0.028 | Significant | — |
Table 7: Summary of key statistical findings for adverse effects of anti-epileptic drugs
Discussion
This study depicts that neurological side effects predominate the safety profile of anti-epileptic medicines (AEDs). The observed prevalence of central nervous system (CNS) adverse effects, such as dizziness (62%) and fatigue or drowsiness (58%), is consistent with global literature. This includes studies indicating that 74% of patients experience dizziness and 5–7% report fatigue. Our results showed that in comparison to monotherapy, combination therapy was associated with increased rates of side effects. These included fatigue (χ² = 4.87, p = 0.027), weight gain (64% vs. 38%, χ² = 5.92, p = 0.015), and severe allergic reactions (18% vs. 6%, p = 0.032). The increased risk linked to polytherapy further supports existing evidence.[26,27] All of this highlights the combination of regimens as a modifiable risk factor. Drug-specific profiles align with previous findings, including phenytoin-related gum hyperplasia and valproate-related adverse drug reactions (ADRs).[24,28] Our statistical studies revealed gender as a significant risk factor. Females reported dizziness more often than males (χ² = 6.24, p = 0.012), and had more severe seizures (U = 1023, p = 0.043). These align with studies indicating improved seizure control in females.[29] However, this is inconsistent with the literature, which does not include gender-based analyses.
Previous research connects polytherapy and age to reduced tolerability of AEDS.[25,26] This is also consistent with our results, which identify age above 45 years (OR = 2.4, p = 0.018) and severe dizziness (OR = 3.1, p = 0.004) as predictors of discontinuation. Furthermore, our analysis showed that in addition to gastrointestinal concerns such as nausea/vomiting and constipation, musculoskeletal problems, especially muscle weakness, were very common. This notably high incidence of muscle weakness (68%) in our cohort contrasts with standard CNS-focused adverse event profiles reported in the literature (e.g., 80% CNS effects).[24] This may indicate variations in regional prescribing practices or differences in the methodologies used for symptom assessment.
Limitations: The small sample size (n=60) makes it hard to generalize, especially when it comes to rare events like severe allergies. The single-center design could cause selection bias, and the cross-sectional technique makes it hard to figure out what causes AEDs and AEs. Patient-reported data may not be accurate because of recall bias in self-reported symptoms. This problem has been seen in several studies as well.[24,25]
Future perspectives: Future research should focus on larger-scale cohort studies that are multi-center, and there must be longitudinal follow-up of at least 2-5 years. This primarily allows monitoring of ADEs over time.[24,29] For severe ADEs, genetic and pharmacogenomic biomarkers must be investigated, which also facilitates personalized therapy and helps in the selection of such AEDs. Epilepsy is prevalent among children globally, yet there is a scarcity of data on the safety of these AEDS in pediatric patients. Hence, further studies in this demographic are necessary. Research must incorporate patient-reported outcomes, including cognitive function and mood disorders, alongside clinical adverse event data to evaluate quality of life comprehensively. Comparative studies on newer antiepileptic drugs (e.g., lacosamide, levetiracetam) in relation to older agents are necessary, as their representation in existing literature is insufficient.[30]
Conclusion
In conclusion, this study shows that neurological and musculoskeletal AEs are a big problem for people with epilepsy, especially women, older adults, and people who are on multiple medications. While it is consistent with evidence from around the world on the toxicity of central nervous system (CNS) – dominated AEDs, it stresses the importance of combination therapy in increasing the chances of AEs and points out useful signs that treatment should be stopped. The results show that personalized AED selection and tailored AE monitoring procedures are especially important for high-risk groups. To improve the effectiveness of epilepsy therapy and patient safety, we need to conduct extensive research. Also, we need more focused pediatric safety assessments and pharmacogenomic techniques.
References
- Chabolla DR. Characteristics of the epilepsies. Mayo Clin Proc. 2002;77(9):981-990. doi:10.4065/77.9.981
PubMed | Crossref | Google Scholar - Jenrow KA, Elisevich K. Pathophysiology of epilepsy. In: Understanding Epilepsy: A Study Guide for the Boards. 2019:1-18.
Pathophysiology of epilepsy - Penderis J. Pathophysiology of epileptic seizures. In Pract. 2014;36:3-9. doi:10.1136/inp.g5098
Crossref | Google Scholar - Jefferys J, Sanders JW, Walker MC, Smalls JE. Basic mechanisms of epilepsy. In: Epilepsy 2003: From Synapse to Society. A Practical Guide to Epilepsy; 2004.
Basic mechanisms of epilepsy - Singh G, Sander JW. The global burden of epilepsy report: Implications for low- and middle-income countries. Epilepsy Behav. 2020;105:106949. doi:10.1016/j.yebeh.2020.106949
PubMed | Crossref | Google Scholar - Centers for Disease Control and Prevention. Epilepsy Facts and Stats. 2024.
Epilepsy Facts and Stats - Park KM, Kim SE, Lee BI, et al. Top 100 cited articles on epilepsy and status epilepticus: A bibliometric analysis. J Clin Neurosci. 2017;42:12-18. doi:10.1016/j.jocn.2017.02.065
PubMed | Crossref | Google Scholar - F. Orooj, R. Siddiqui, A. Ahmad, and M. Siddiqui. Epilepsy in Pakistan: Frequency and common epilepsy types in a tertiary care hospital. J Neurol Sci. 2021;429:119094. doi: 10.1016/j.jns.2021.119094
Crossref | Google Scholar - Khatri IA, Iannaccone ST, Ilyas MS, Abdullah M, Saleem S. Epidemiology of epilepsy in Pakistan: review of literature. J Pak Med Assoc. 2003;53(12):594-597.
Epidemiology of epilepsy in Pakistan: review of literature - Aziz H, Ali SM, Frances P, Khan MI, Hasan KZ. Epilepsy in Pakistan: a population-based epidemiologic study. Epilepsia. 1994;35(5):950-958. doi:10.1111/j.1528-1157.1994.tb02539.x
PubMed | Crossref | Google Scholar - Siddiqui F, Sultan T, Mustafa S, Siddiqui S, Ali S, Malik A, Sajjad Z, Barech S, Jooma R. Epilepsy in Pakistan: national guidelines for clinicians. Pak J Neurol Sci. 2015;10(3):Article 11.
Epilepsy in pakistan: national guidelines for clinicians - Perucca E. An introduction to antiepileptic drugs. Epilepsia. 2005;46 Suppl 4:31-37. doi:10.1111/j.1528-1167.2005.463007.x
PubMed | Crossref | Google Scholar - Garnett WR. Antiepileptic drug treatment: outcomes and adherence. Pharmacotherapy. 2000;20(8 Pt 2):191S-199. doi:10.1592/phco.20.12.191s.35250
PubMed | Crossref | Google Scholar - Sander JW. The use of antiepileptic drugs–principles and practice. Epilepsia. 2004;45 Suppl 6:28-34. doi:10.1111/j.0013-9580.2004.455005.x
PubMed | Crossref | Google Scholar - LaRoche SM, Helmers SL. The New Antiepileptic Drugs: Scientific Review. JAMA. 2004;291(5):605–614. doi:10.1001/jama.291.5.605
Crossref | Google Scholar - Sarhan EM, Walker MC, Selai C. Evidence for efficacy of combination of antiepileptic drugs in treatment of epilepsy. J Neurol Res. 2015;5(6):267‑276. doi:10.14740/jnr356w
Crossref | Google Scholar - Bootsma HP, Ricker L, Hekster YA, et al. The impact of side effects on long-term retention in three new antiepileptic drugs. Seizure. 2009;18(5):327-331. doi:10.1016/j.seizure.2008.11.006
PubMed | Crossref | Google Scholar - Lhatoo SD, Wong IC, Polizzi G, Sander JW. Long-term retention rates of lamotrigine, gabapentin, and topiramate in chronic epilepsy. Epilepsia. 2000;41(12):1592-1596. doi:10.1111/j.1499-1654.2000.001592.x
PubMed | Crossref | Google Scholar - Carpay JA, Aldenkamp AP, van Donselaar CA. Complaints associated with the use of antiepileptic drugs: results from a community-based study. Seizure. 2005;14(3):198-206. doi:10.1016/j.seizure.2005.01.008
PubMed | Crossref | Google Scholar - Beghi E; et al. Efficacy and tolerability of the new antiepileptic drugs. Epilepsy & Behaviour. 2004;5(6):267-276. doi:10.1016/S1474-4422(04)00882-8
Crossref | Google Scholar - Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomised controlled trial. Lancet. 2007;369(9566):1000-1015. doi:10.1016/S0140-6736(07)60460-7
PubMed | Crossref | Google Scholar - Dichter MA, Brodie MJ. New antiepileptic drugs. N Engl J Med. 1996;334(24):1583-1590. doi:10.1056/NEJM199606133342407
PubMed | Crossref | Google Scholar - Brodie MJ, Richens A, Yuen AW. Double-blind comparison of lamotrigine and carbamazepine in newly diagnosed epilepsy. UK Lamotrigine/Carbamazepine Monotherapy Trial Group. Lancet. 1995;345(8948):476-479. doi:10.1016/s0140-6736(95)90581-2
PubMed | Crossref | Google Scholar - Mahatme MS, Gupta V, Hiware SK, Hardas MM. A questionnaire-based study of adverse drug effects of antiepileptic drugs in a tertiary care hospital in Central India. Int J Basic Clin Pharmacol. 2018;7(12):2336-2341. doi:10.18203/2319-2003.ijbcp20184842
Crossref | Google Scholar - Ayalew MB, Muche EA. Patient reported adverse events among epileptic patients taking antiepileptic drugs. SAGE Open Med. 2018;6:2050312118772471. doi:10.1177/2050312118772471
PubMed | Crossref | Google Scholar - Carpay JA, Aldenkamp AP, van Donselaar CA. Complaints associated with the use of antiepileptic drugs: results from a community-based study. Seizure. 2005;14(3):198-206. doi:10.1016/j.seizure.2005.01.008
PubMed | Crossref | Google Scholar - Chaplin JE, Yepez Lasso R, Shorvon SD, Floyd M. National general practice study of epilepsy: the social and psychological effects of a recent diagnosis of epilepsy. BMJ. 1992;304(6839):1416-1418. doi:10.1136/bmj.304.6839.1416
PubMed | Crossref | Google Scholar - Maqbool M, Dugassa D, Fekadu G. Adverse drug reactions of antiepileptic drugs in the neurology department of a tertiary care hospital, Srinagar, Jammu & Kashmir, India. Arch Neurosci. 2021;8:e112364. doi:10.5812/ans.112364
Crossref | Google Scholar - Ali S, Fatima M. Comparison of anti-epileptic drugs in terms of treatment outcomes, adverse effects and quality of life. J Clin Diagn Res. 2019;13:FC01-07. doi:10.7860/JCDR/2019/41895.13228
Crossref | Google Scholar - Magar Y, Hiray R, Ghongane B. Utilization pattern of antiepileptic drugs and their adverse effects in a tertiary healthcare and teaching hospital. Int J Basic Clin Pharmacol. 2018;8(1):39-46. doi:10.18203/2319-2003.ijbcp20185119
Crossref | Google Scholar
Acknowledgments
The authors declare that there are no acknowledgments to disclose.
Funding
The authors received no financial support.
Author Information
Corresponding Author:
Hamid Tariq Shah
Department of Pharmacy
Quaid-i-Azam University, Islamabad, Pakistan
Email: hamidtariq283@gmail.coma
Co-Authors:
Aneesa Anjum, Adan Minal Asif, Amna Arshad, Waqas Siddiqe, Uswa Mansoor, Sahifa Ali, Soma Akbar
Department of Pharmacy
Quaid-i-Azam University, Islamabad, Pakistan
Authors Contributions
All authors contributed to the conception, design, data collection, statistical analysis, literature review, and writing of this manuscript. All authors read and approved the final version.
Ethical Approval
Ethical approval was obtained in the form of a permission letter from the respective bodies of Pakistan Institute of Medical Sciences (PIMS) and Pakistan Ordinance Factory Hospital (POF). Patients were informed of the study’s objectives, and informed consent was obtained.
Conflict of Interest Statement
The authors declare no conflict of interest.
Guarantor
Hamid Tariq Shah is the guarantor of the article and takes responsibility for the integrity of the work as a whole.
DOI
Cite this Article
Shah HT, Anjum A, Asif AM, et al. Evaluation of Adverse Drug Effects of Anti-Epileptic Drugs in Tertiary Care Hospitals. medtigo J Pharmacol. 2026;3(1):e3061311. doi:10.63096/medtigo3061311 Crossref









