Author Affiliations
Abstract
A 74-year-old African American woman with a medical history of rheumatoid arthritis (RA) and end-stage renal disease (ESRD) on hemodialysis presented with a sudden onset of multiple, firm, hyperpigmented skin plaques resembling eruptive keloids. Notably, many of these lesions appeared in areas without any preceding trauma, except for one longstanding keloid on her left elbow. The unusual distribution and rapid development of these lesions, combined with her significant renal dysfunction, raised concern for nephrogenic systemic fibrosis (NSF), a rare but serious fibrosing disorder linked to gadolinium-based contrast exposure in patients with compromised renal function. Review of her medical records revealed gadolinium exposure approximately three months prior during contrast-enhanced magnetic resonance imaging (MRI). Her symptoms began shortly thereafter, with progressively worsening skin thickening, tightening, and decreased mobility. Dermatology evaluation noted findings consistent with NSF rather than typical keloids. Despite supportive measures including physical therapy and topical treatments, her condition deteriorated rapidly, contributing to multisystem decline and ultimately leading to her death. This case illustrates an atypical presentation of NSF mimicking eruptive keloids and emphasizes the need for early recognition. It also highlights the importance of minimizing gadolinium use in patients with advanced kidney disease to prevent potentially fatal complications.
Keywords
Keloid, Renal failure, Nephrogenic systemic fibrosis, Gadolinium, Rheumatoid arthritis.
Introduction
Keloids are benign growths of dense, fibrous tissue resulting from an abnormal wound-healing response to injury.[1,2] This injury can stem from trauma, surgery, inflammation, or burns. While most keloids present within 3 months of the inciting event, some occur up to a year later.[2,3] Clinically, keloids manifest as firm, pruritic, and often painful nodules unlikely to regress spontaneously.[1-3] Unlike hypertrophic scars, keloids spread beyond the initial injury borders, invading adjacent normal skin. Common sites for keloid formation include the earlobes, shoulders, chest, back of the neck, and upper back.[1,3]
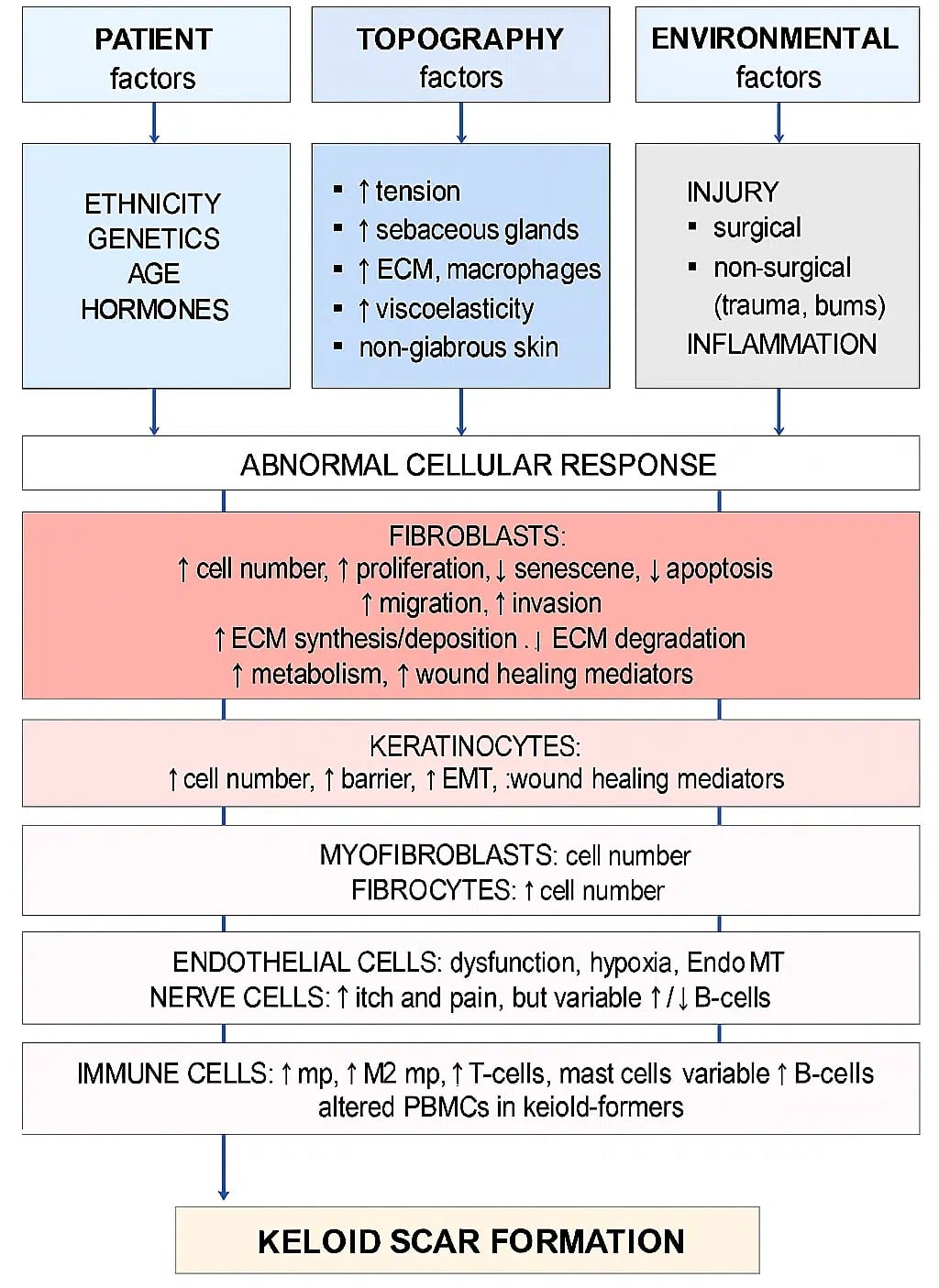
Figure 1: Pathogenesis of keloid formation
Keloid pathogenesis involves a complex interplay between environmental and genetic factors. Individuals with darker skin tones, particularly those of African, Asian, or Hispanic descent, are significantly more predisposed, with studies suggesting up to a 15-fold increased risk compared to individuals with lighter skin tones.[4,5] Common triggers include acne, folliculitis, injectable vaccinations, ear piercings, and surgical incisions, all of which can initiate abnormal wound-healing responses.[6] While most keloids develop within three months of the inciting injury, some may arise as late as a year afterward.[7] While the precise mechanisms remain under investigation, research highlights the roles of transforming growth factor beta (TGF-β), which stimulates growth and collagen production, and platelet-derived growth factor (PDGF), which promotes connective tissue proliferation, in keloid formation.[3,6]
Treatment options for keloids include silicone gel sheets, steroid injections, surgical excision, cryotherapy, laser removal, and radiotherapy. However, efficacy is variable, and recurrence is common even with intervention.[8] One important consideration in patients with end-stage renal disease is NSF, a rare, rapidly progressive disorder marked by extensive cutaneous and systemic fibrosis, including involvement of the diaphragm, heart, and skeletal muscles. NSF is strongly associated with prior exposure to gadolinium-based contrast agents, particularly in individuals with severely impaired renal function (estimated glomerular filtration rate (eGFR) <30 mL/min/1.73 m²). Since its first description in 2000, NSF has emerged as a significant clinical concern in both nephrology and dermatology due to its potentially irreversible course.[9]
Case Presentation
A 74-year-old African American woman with a 36-year history of RA, untreated for the first 20 years, presents with keloid formation. These keloids arise on old, healed scars and recent dialysis needle insertion sites. She had maintained good medication and dietary compliance during her four years on chronic intermittent hemodialysis (3 days/week), with adequate clearance. A renal biopsy revealed secondary amyloidosis as the etiology of her renal failure; she had no prior history of hypertension or substance abuse. While her family has a history of RA, none have experienced renal failure or require dialysis. She reported chronic polyarthritis affecting both small and large joints, along with significant early morning stiffness. She denied fever, weight loss, Raynaud’s phenomenon, skin rash, nodules, hair loss, or urinary symptoms. There’s no history of mouth ulcers, photosensitivity, or sicca syndrome.
Her dialysis access, a mature left upper arm fistula used with no complications for over three years, started to develop painful keloids at the needle sites, necessitating a switch to a tunneled internal jugular catheter. Weeks later, she developed a keloidal lesion on previously healed, flat scars from prior dialysis catheters (subclavian and internal jugular) and even a 25-year-old gallbladder surgery scar. She denies recent trauma or constrictive clothing at these sites.
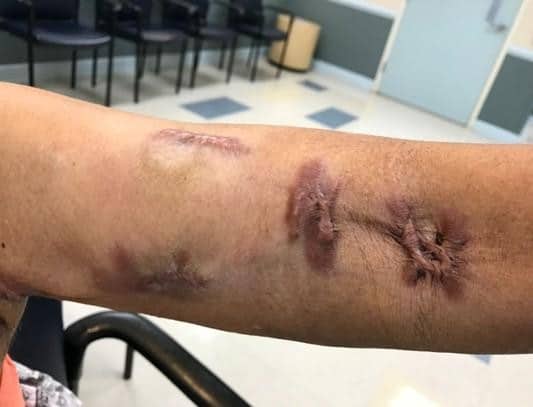
Figure 2: New keloidal scars on dialysis sites
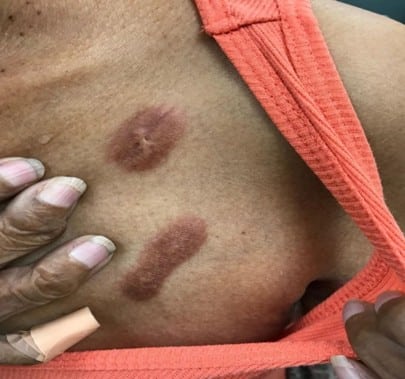
Figure 3: New keloid scar on an old, healed, flat scar of old catheter insertion sites
Physical examination revealed normal blood pressure (110/60) and pulse (78), with a BMI of 22. Multiple tender and swollen joints were present, primarily in the metacarpophalangeal (MCP) joints and knees. The skin examination revealed a 4 by 5 cm mildly tender pinkish red hyperpigmented firm plaque with an irregular border on the flexural side of the left elbow joint, a 2 by 3 cm pinkish red hyperpigmented tender firm plaque on the left upper chest, and RUQ of the abdomen. The other systems examined were unremarkable.
The patient does not produce urine, and laboratory evaluation showed creatinine of 5.6 mg/dl; blood urea nitrogen (BUN) of 52.4 mg/dl; albumin of 3.8 g/dl; erythrocyte sedimentation rate (ESR) of 42 mm/hour; C-reactive protein (CRP) ofu10g/l; and fasting blood sugar of 96 mg/dl. Her complete blood count, liver function tests, serum electrolytes, hemoglobin A1c, and complement levels were normal. Her antinuclear antibodies (ANA), anti-cyclic citrullinated peptide, virology profile of hepatitis B (HBV), human immunodeficiency virus (HIV), and hepatitis C (HCV) were all negative. Notably, three months prior, she underwent an MRI with Group 1 Gadolinium contrast at another hospital and required dialysis thereafter. Her medications (unchanged for two years) include nephrite, calcium acetate, methotrexate, prednisolone, folic acid, erythropoietin, and intravenous iron (administered during dialysis). The patient subsequently deteriorated and died.
Discussion
A keloid scar is a benign hyperproliferative growth of fibrous tissue from an abnormal healing response that develops after an injury or trauma to the skin, although they may also occur spontaneously.[3,6] Typically, keloids form within months of the initial injury. The delayed formation in this patient, appearing years after the initial trauma, is unusual and prompts further investigation into possible contributing factors. This atypical presentation is associated with a few syndromes, such as Noonan syndrome and Goeminne syndrome.[10] The association of these keloids with specific genetic syndromes suggests a potential link between mutations and keloid formation. In addition to these genetic syndromes, NSF, seen in patients with renal failure and a GFR < 30 mL/min/1.73 m^2, has been associated with fibrotic skin changes and keloid-like lesions.[11-13]
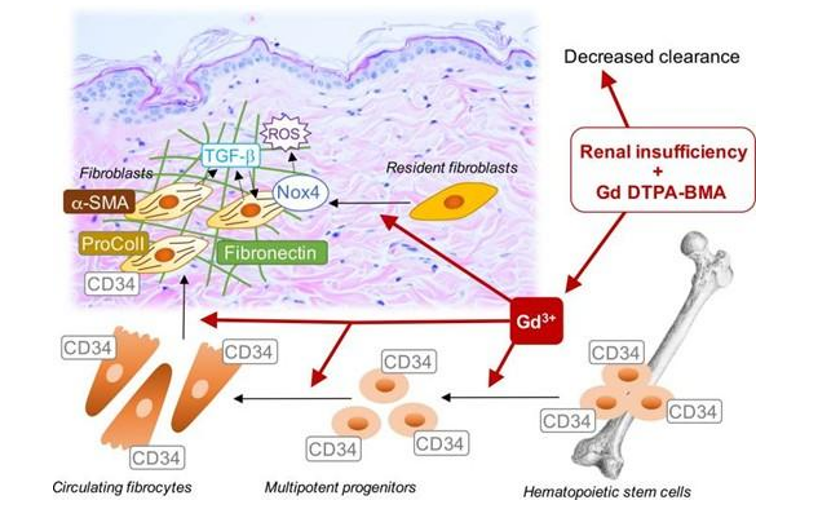
Figure 4: Possible molecular mechanism implicated in gadolinium-based contrast-induced fibrosis
NSF is characterized by progressive cutaneous and multi-organ fibrosis after exposure to gadolinium.[14] Additionally, uremia can alter wound healing processes. The exact cause of NSF is not fully understood, but it is believed to involve gadolinium, a contrast agent used in imaging scans. Gadolinium may break free from its chelate and interact with tissue receptors, stimulating the production of chemicals that promote fibrosis, leading to tissue thickening.[14] Patients with severe kidney dysfunction are at higher risk. In advanced kidney disease, gadolinium stays in the body longer. Some hypotheses suggest that gadolinium could trigger a fibrotic cycle, leading to excessive collagen deposition.[14,15] Various theories have been proposed, including the role of hepatitis C, immune system suppression, and the involvement of specific cells and chemicals in the development of NSF.
The pathogenic correlation between scarring in the setting of uremia is not well understood. Still, studies have shown that pathogenesis is multifactorial, most notably inflammatory and fibrotic pathways contributing to keloid formation.[16,17] Uremia can lead to chronic inflammation and vascular changes in the body due to the proliferation of fibroblasts and tissue ischemia, which leads to fibrosis.[17] Chronic inflammation triggers endothelial-to-mesenchymal transition (EMT), which promotes the formation of myofibroblasts and the continuous cycle of collagen synthesis and deposition in the extracellular matrix.[18] Additionally, vascular damage prevents adequate delivery of immune cells and growth factors to the skin, impairing proper healing ability.[18,19]
NSF is characterized by progressive cutaneous fibrosis with thickened, indurated, and woody plaques rather than classic raised keloids.[20] Unlike keloids, which typically arise from abnormal wound healing after trauma, NSF lesions often develop in areas without prior injury and may involve multi-organ fibrosis, including muscle and internal organs.[21,22] Differential diagnoses considered include hypertrophic scars, scleroderma, and cutaneous amyloidosis; however, the history of gadolinium exposure, chronic renal failure, lesion distribution, and rapid progression strongly support NSF as the favored diagnosis in this case.[23] Recognizing these clinical and histopathological distinctions is critical for appropriate diagnosis and management.
In this case, the patient’s prior exposure to gadolinium contrast may have altered her immune response and wound healing, potentially contributing to the unusual formation of keloid-like lesions on decades-old surgical scars. Additionally, her secondary amyloidosis from RA resulted in renal failure requiring chronic hemodialysis. The repeated needle insertions during dialysis likely caused chronic microtrauma and local inflammation, further predisposing her to keloid development. The patient’s clinical deterioration underscores the severe and progressive nature of her condition, reinforcing the suspicion of NSF as a significant contributing factor.
Conclusion
This case underscores the formation of keloids in a patient with end-stage renal disease and prior gadolinium exposure, emphasizing the importance of considering NSF in similar presentations. The progressive course of the patient’s condition highlights the critical need for increased vigilance and prompt management of potential complications associated with gadolinium exposure, especially in individuals with impaired renal function. Clinicians should exercise caution with the use of gadolinium-based contrast agents, especially in individuals with severe chronic kidney disease (eGFR <30 mL/min/1.73 m²), and consider alternative imaging modalities whenever possible to minimize the risk of NSF and its serious complications.
References
- Wang ZC, Zhao WY, Cao Y, et al. The Roles of Inflammation in Keloid and Hypertrophic Scars. Front Immunol. 2020;11:603187. doi:10.3389/fimmu.2020.603187 PubMed | Crossref | Google Scholar
- Shim J, Oh SJ, Yeo E, et al. Integrated Analysis of Single-Cell and Spatial Transcriptomics in Keloids: Highlights on Fibrovascular Interactions in Keloid Pathogenesis. J Invest Dermatol. 2022;142(8):2128-2139.e11. doi:10.1016/j.jid.2022.01.017 PubMed | Crossref | Google Scholar
- Hunasgi S, Koneru A, Vanishree M, Shamala R. Keloid: A case report and review of pathophysiology and differences between keloid and hypertrophic scars. J Oral Maxillofac Pathol. 2013;17(1):116-120. doi:10.4103/0973-029X.110701 PubMed | Crossref | Google Scholar
- Chen H, Hou K, Wu Y, Liu Z. Use of Adipose Stem Cells Against Hypertrophic Scarring or Keloid. Front Cell Dev Biol. 2022;9:823694. doi:10.3389/fcell.2021.823694 PubMed | Crossref | Google Scholar
- Marneros AG, Krieg T. Keloids–clinical diagnosis, pathogenesis, and treatment options. J Dtsch Dermatol Ges. 2004;2(11):905-913. doi:10.1046/j.1439-0353.2004.04077.x PubMed | Crossref | Google Scholar
- Robles DT, Berg D. Abnormal wound healing: keloids. Clin Dermatol. 2007;25(1):26-32. doi:10.1016/j.clindermatol.2006.09.009 PubMed | Crossref | Google Scholar
- Huang C, Liu L, You Z, Du Y, Ogawa R. Managing keloid scars: From radiation therapy to actual and potential drug deliveries. Int Wound J. 2019;16(3):852-859. doi:10.1111/iwj.13104 PubMed | Crossref | Google Scholar
- Ud-Din S, Bayat A. Strategic management of keloid disease in ethnic skin: a structured approach supported by the emerging literature. Br J Dermatol. 2013;169 Suppl 3:71-81. doi:10.1111/bjd.12588 PubMed | Crossref | Google Scholar
- Elmholdt TR, Olesen ABB, Jørgensen B, Kvist S, Skov L, Thomsen HS, Marckmann P, Pedersen M. Nephrogenic Systemic Fibrosis in Denmark – A Nationwide Investigation. PLoS One. 2013;8(12):e82037. doi:10.1371/journal.pone.0082037 PubMed | Crossref | Google Scholar
- Jfri A, Rajeh N, Karkashan E. A Case of Multiple Spontaneous Keloid Scars. Case Rep Dermatol. 2015;7(2):156-160. doi:10.1159/000437249 PubMed | Crossref | Google Scholar
- Shaheen A. Comprehensive review of keloid formation. J Clin Res Dermatol. 2017;4:1-18. doi:10.15226/2378-1726/4/5/00168 Crossref | Google Scholar
- Sadowski EA, Bennett LK, Chan MR, et al. Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology. 2007;243(1):148-157. doi:10.1148/radiol.2431062144 PubMed | Crossref | Google Scholar
- Marckmann P, Skov L, Rossen K, et al. Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol. 2006;17(9):2359–2362. doi:10.1681/ASN.2006050486 PubMed | Crossref | Google Scholar
- Wagner B, Drel V, Gorin Y. Pathophysiology of gadolinium-associated systemic fibrosis. Am J Physiol Renal Physiol. 2016;311(1):F1-11. doi:10.1152/ajprenal.00166.2016 PubMed | Crossref | Google Scholar
- Shamam YM, Hashmi MF, De Jesus O. Nephrogenic Systemic Fibrosis. eBook. StatPearls publishing; 2024. Nephrogenic Systemic Fibrosis
- Vaziri ND, Pahl MV, Crum A, Norris K. Effect of uremia on structure and function of immune system. J Ren Nutr. 2012;22(1):149-156. doi:10.1053/j.jrn.2011.10.020 PubMed | Crossref | Google Scholar
- Six I, Flissi N, Lenglet G, et al. Uremic Toxins and Vascular Dysfunction. Toxins (Basel). 2020;12(6):404. doi:10.3390/toxins12060404 PubMed | Crossref | Google Scholar
- Huang C, Ogawa R. The Vascular Involvement in Soft Tissue Fibrosis-Lessons Learned from Pathological Scarring. Int J Mol Sci. 2020;21(7):2542. doi:10.3390/ijms21072542 PubMed | Crossref | Google Scholar
- Ogawa R, Akaishi S. Endothelial dysfunction may play a key role in keloid and hypertrophic scar pathogenesis – Keloids and hypertrophic scars may be vascular disorders. Med Hypotheses. 2016;96:51-60. doi:10.1016/j.mehy.2016.09.024 PubMed | Crossref | Google Scholar
- Lunyera J, Mohottige D, Alexopoulos AS, et al. Risk for Nephrogenic Systemic Fibrosis After Exposure to Newer Gadolinium Agents: A Systematic Review. Ann Intern Med. 2020;173(2):110-119. doi:10.7326/M20-0299 PubMed | Crossref | Google Scholar
- Weinreb JC, Kuo PH. Nephrogenic systemic fibrosis. Magn Reson Imaging Clin N Am. 2009;17(1):159-167. doi:10.1016/j.mric.2009.01.003 PubMed | Crossref | Google Scholar
- Lomonte C, Basile C. Preoperative assessment and planning of haemodialysis vascular access. Clin Kidney J. 2015;8(3):278-281. doi:10.1093/ckj/sfv022 PubMed | Crossref | Google Scholar
- Kribben A, Witzke O, Hillen U, Barkhausen J, Daul AE, Erbel R. Nephrogenic systemic fibrosis: pathogenesis, diagnosis, and therapy. J Am Coll Cardiol. 2009;53(18):1621-1628. doi:10.1016/j.jacc.2008.12.061 PubMed | Crossref | Google Scholar
Acknowledgments
None
Funding
None
Author Information
Corresponding Author:
Miriam Michael
Department of Internal Medicine
Howard University, Washington, DC, USA
University of Maryland School of Medicine, Baltimore, USA
Email: michaelclarkefamily@gmail.com
Co-Authors:
Samrawit Zinabu, Arsema Zadu, Elizabeth Beyene, Mekdem Bisrat, Ahmad Mohammed, Huda Gasmelseed
Department of Internal Medicine
Howard University, Washington, DC, USA
Noah Wheaton, Jordan Young, Christian Wong, Courtney Squirewell, Bianca Campbell
College of Medicine
Howard University, Washington, DC, USA
Ayushi Sen
Department of Medicine
American University of Antigua, Osbourn, Antigua, Antigua and Barbuda
Authors Contributions
Samrawit Zinabu contributed to the conceptualization of the study and was responsible for writing the original and final versions of the document. Jordan Young, Ahmad Mohammed, Ayushi Sen, Bianca Cambell, Courtney Squirewell, Arsema Zadu, and Noah Wheaton contributed to writing the final document, editing references, and formatting. Elizabeth Beyene, Mekdem Bisrat, and Huda Gasmelseed assisted in editing the final document. Miriam Michael contributed to the conceptualization, edited the final document, and provided supervision throughout the project.
Informed Consent
Informed consent for publication of this case report and accompanying images was obtained from the patient. All efforts were made to ensure patient confidentiality and anonymity.
Conflict of Interest Statement
The authors declare no conflicts of interest.
Guarantor
None
DOI
Cite this Article
Zinabu S, Wheaton N, Young J, et al. Eruptive Keloids in a Patient on Dialysis: A Case of Suspected Nephrogenic Systemic Fibrosis. medtigo J Med. 2025;3(2):e30623229. doi:10.63096/medtigo30623229 Crossref









