Author Affiliations
Abstract
Background: Intraoperative squash cytology is an important diagnostic tool for rapid and accurate diagnosis of central nervous system tumors. It has been proven to play a vital role in determining further surgical management of patients undergoing surgery. It can be ascertained as an alternative method to frozen sections in resource-limited settings. The objective of this study was to evaluate the value of squash smear cytology in rapid intra-operative diagnosis of central nervous system tumors and its correlation with final histopathological diagnosis.
Methodology: This was a cross-sectional, descriptive study over a one-year duration. A total of 57 patients with nervous system tumors who underwent surgery at the Department of Neurosurgery, Tribhuvan University Teaching Hospital (TUTH) were included in the study. Intra-operatively, the biopsy was sent in isotonic saline; the unfixed tissue was crushed with slides to obtain smears. Cytological evaluation was performed and correlated with histopathology diagnosis as the gold standard.
Results: Out of 57 cases, 46 cases (80.7%) showed complete correlation between squash cytology and histopathology diagnosis. The age-wise evaluation showed an increase in frequency in the elderly, with a peak incidence in the fifth decade. Glial tumors were the most common (42%), followed by meningioma (21%) and schwannoma (19%). Among glial tumors, Glioblastoma was the most common.
Conclusion: Squash smear cytology is a reliable and rapid diagnostic tool, and it can assist in intra-operative decision-making and diagnosis of intracranial tumors in resource-limited settings.
Keywords
Squash smear, Central nervous system, Intracranial tumors, Resource-limited settings, Rapid diagnostic tool.
Introduction
The annual incidence of tumors of the central nervous system (CNS) ranges from 10-17 per 100,000 persons for intracranial tumors and 1-2 per 1,00,000 persons for intraspinal tumors. Out of these, half to three-quarters are primary tumors, and the rest are metastatic.[1] CNS tumors include gliomas, neuronal tumors, meningioma, peripheral nerve sheath tumors, lymphoma, germ cell tumors, and metastatic tumors.[2] A diverse variety of benign cysts exist in the CNS, including dermoid cysts, epidermoid cysts, Rathke’s cleft cysts, and other epithelial cysts.[3]
CNS lesions are one of the challenging domains for both neurosurgeons and pathologists. Making a diagnosis of CNS lesions is difficult based on clinical and radiological findings alone. Cytological and/ or histological diagnosis is required for confirmation and proper management.[4] In this regard, squash cytology smears (SCS) prepared from small biopsy intra-operatively seem to be very helpful to make a prudent decision on further management.[5] For rapid intra-operative diagnosis, there are many diagnostic modalities in addition to frozen section, such as squash and touch or imprint smears. SCS plays a vital role when tissue availability is limited, though frozen section is superior in providing both the histological patterns and cytomorphologic details. SCS is useful in the examination of the intercellular matrix, the evaluation of discohesive cells, and in inflammatory CNS lesions.[6] Compared with frozen sections, this technique has the dual advantage of better cytological details and requires less tissue; at the same time, it avoids freezing artifacts commonly encountered during frozen section preparation.[7] CNS tissue is especially prone to such artifacts, and rapid freezing is of utmost importance, which is also difficult due to the high lipid content of the tissue. Therefore, this problem can be avoided by the squash technique.[8]
The application of smear techniques as a means of obtaining rapid diagnosis for neurosurgical biopsies was first advocated by Dr. Eisenhardt and Dr. Cushing in the USA in the early 1930s.[9] The smear technique has been extensively used for the last 40 years in centers in the UK for the examination of neurological biopsies obtained from needle or stereotactic biopsies.[8] Badt et al. performed the crush method for rapid diagnosis of CNS lesions in Germany using a supravital stain, and it correlated well with histological diagnosis. However, the disadvantage of the stain was that its preparation was not permanent, meaning the slides could not be retained for extended periods for subsequent review.[10-12] For routine squash cytology examination, hematoxylin and eosin (H&E), Giemsa, and Papanicolaou (PAP) stains have been variably used to provide better cytological details.[13]
The strength of SCS is that it is simple, rapid, and effective. It provides good cellular details, allows tissue to be preserved for paraffin embedding, and requires minimal equipment and technical manpower. In addition, it can also be carried out at the operative site. One of the most important advantages is that even very tiny specimens can be sufficient for smear preparation, which is extremely important in surgical procedures involving intracranial lesions. It also provides excellent teaching material from a cytopathologist’s point of view.[14]
Advantages and pitfalls of squash smear cytology: Intra-operative smears of neurological specimens permit rapid and accurate diagnosis if a systematic approach is implemented in the analytical process.[15] To employ these techniques effectively, advantages and pitfalls should be properly understood, besides having knowledge of clinical and radiological information.
Common advantages
- Economy of time, technical equipment, and a limited amount of tissue: Turnover of the specimen is reduced because it takes only 5-10 minutes to prepare the smear, and staining can also be performed in a short time. Technical expertise and a cryostat needed for frozen section are bypassed.[2,16]
- Good cellular morphology: This method also provides an opportunity to study the cells that have not changed due to fixation and cutting. The usual shrinkage and distortion of the cells are thus avoided. Astrocytic processes in astrocytoma, nuclear details in certain tumors like pituitary adenoma, and lymphomas are better studied in squash cytology.[4,8]
- Preservation of some forms of architecture: In squash smears, besides better cellular details, the relationship of the various structures in the tumor as a whole is well preserved. For instance, the formation of whorls and masses of cells in meningioma, or the perivascular radiation of the cells in ependymoma, are commonly seen.[17-19]
- Bony lesions, which are not amenable to frozen sections, can be scraped and smeared on a slide. This can reveal enough information to allow initial treatment while awaiting decalcification and paraffin-embedded sections.[20]
Limitations
- Reliability on the elasticity of the specimen: Firm lesions like nerve sheath tumors are very difficult to smear due to the tough nature of the tissue. Smearing is also difficult in cystic and calcified lesions. Moreover, the thickness of smears cannot be controlled. Astrocytic tumors are relatively smeared well on the slide.[16]
- Grading: Grading of neoplasms in cytological preparations cannot be accurately done. For example, in the case of gliomas, overlapping vessels may be confused with endothelial proliferation and can be misinterpreted as high-grade tumors.[21]
- Sampling error: In some instances, when samples are taken from normal brain parenchyma or necrotic portions, a definite opinion on squash smear examination may not be possible. So, sampling from multiple tissue fragments should be taken into consideration.[22,23]
- Poor preservation of histologic architecture: In squash smears, the overall histological pattern and characteristic features are not elucidated well. For example, there is a loss of features like pseudopallisading necrosis in GBM.[22,24]
- Vascular lesion: Due to hemorrhagic nature and low cellular yield, vascular lesions can be misdiagnosed or labeled as inadequate.[25]
Cytologic features of different CNS tumors in squash cytology
- Normal brain tissue in squash cytology: The normal brain parenchyma appears as a poorly cellular smear with scattered elongated cells or naked small nuclei. The presence of more glial components and neuronal cell bodies from grey matter, along with the capillary bed with pericytes and histiocytes, may also be noted.[26]
- Reactive lesions: Reactive astrocytes possess abundant cytoplasm than normal cells. When compared to neoplastic cells, they show more prominent, numerous, long, and symmetric processes and a few hyperchromatic and lobulated nuclei in the absence of mitotic figures and progressive atypia.[27] The neoplastic astrocytes vary in stain intensity and show predominance of a single cell type, whereas reactive brain tissue contains a mixture of cells in the smear.[2]
- Astrocytic tumors:
- Pilocytic astrocytoma: Low-grade astrocytic tumors (pilocytic astrocytoma) are moderately cellular with minimal anisocytosis, having fine to coarse granular chromatin and inconspicuous nucleoli: the cytoplasm is scanty and shows variable processes with fibrillary background.[6]The tumor shows bipolar ‘hair’ cells with elongated, coarse cytoplasmic processes and bland, oval nuclei. Rosenthal fibers, eosinophilic granular bodies, and hyalinized vessels are usual findings.[4] Stellate cells and bare tumor nuclei are also present. Background may be myxoid. Nuclear pleomorphism, vascular proliferation, perivascular pseudo-rosettes, microcalcification, and hemosiderin-laden macrophages are infrequent features. Mitoses and necrosis are absent.[28]
- Anaplastic astrocytoma: The WHO has distinguished two high-grade astrocytic tumors: anaplastic astrocytoma (grade III) and glioblastoma (grade IV). Squash smear of both these entities shows greater nuclear pleomorphism, coarser chromatin, thicker glial processes, increased mitotic figures, and vascular proliferation. However, tumor grading in SCS is challenging because anaplastic astrocytoma and glioblastoma exhibit a continuum of features ranging from low to high grade.[4]
- Glioblastoma: SCS of glioblastoma are highly cellular and are characterized by a greater degree of nuclear pleomorphism, binucleation or multinucleation, eccentrically placed nuclei, coarser chromatin, and frequently prominent nucleoli. They also display a greater cellularity on a denser and fibrillary background. Additionally, there is endothelial proliferation and necrosis.[29] The clusters of tumor cells are interconnected with each other by thickened glial strands.[4]
- Oligodendroglioma: SCS of oligodendrogliomas typically has relatively rounded, smoother, and monomorphic nuclei. The differences in nuclei decrease with increasing histologic grade. Their nuclei have relatively bland chromatin. Mini-gemistocytes and some glial strands on high power help ascertain the diagnosis of oligodendroglioma. Perinuclear halos that give oligodendroglioma the fried egg appearance on histological sections are an artifact of fixation and not present on smears. Calcification, when present, may be a helpful feature.[4,8]
- Ependymoma: Ependymoma shows perivascular pseudo-rosettes or true rosettes, and the tumor cells are less cohesive at the periphery of fragments with unipolar cytoplasmic processes. The background is fibrillary, except in myxopapillary ependymoma. Metachromatic material is seen in myxopapillary ependymoma around vessels and in globi. The nucleus is round with stippled chromatin, and 1-2 small nucleoli can be seen. Grade III ependymomas show pleomorphism, mitosis, microvascular proliferation, and necrosis. Myxoid stroma is better appreciated by toluidine blue staining.[4,30]
- Meningioma: Tumor cells of meningioma are arranged in islands, whorls, or lobules. The nuclei are typically monotonous and bland, and their cellular aggregates are syncytial and interconnected. A minority of meningiomas display psammoma bodies. [4] In the subclass of meningioma, peculiar features often overlap, and subtyping yields poor correlation. In malignant meningiomas, the cytologic features are pleomorphic, like those of metastatic carcinoma, making diagnosis difficult.[31]
- Nerve sheath tumors: Unlike meningiomas, smears are much less useful for evaluating nerve sheath tumors. Due to a dense collagenous matrix, tumor cells appear as a thick clump with no cellular monolayer in cytologic preparation. Schwannomas having significant Antoni A areas show ‘snapped’ or ‘frayed rope’ appearance at the edges of clumps of tumor cells. There are small strands of collagen sticking out of the edges. There is a swarming of nuclei within bundles of fine filaments. The nuclei are oval to elongated and have fine chromatin and a smooth nuclear membrane. The differentiation of schwannoma from neurofibroma on squash cytology is difficult, and correlation with clinical features and radiological findings may assist in accurate diagnosis.[4]
- Craniopharyngioma: Tumor displays solid sheets of squamous epithelial cells with basaloid cells at the edges. They are monotonous, and a glob of wet keratin may be seen.[4]
- Lymphomas: An intra-operative smear provides key and unique information about lymphoma. In a squash smear, an even gradient of cellular density, monomorphic tumor cells with a lack of cellular cohesion on a background showing numerous tingible body macrophages and lymphoglandular bodies are the characteristic features of primary CNS lymphoma.[32] Typical discohesive lymphoma cells with lymphoglandular bodies without fibrillary background on squash smears would help to identify the lesion as hematopoietic origin.[33]
- Chordoma: At lower power, the myxoid matrix of chordoma imparts a basophilic hue on the background. The cell of this tumor exhibits epithelial properties characterized by distinct cell borders with abundant cytoplasm and adhesiveness to each other. The nuclei are monotonous and bland. Pleomorphism, anaplasia, and hyperchromasia may be minimal. The presence of relatively bland epithelial cells arranged in sheets and cords and set in a thick, myxoid matrix with a variable number of vacuoles is a key guiding feature.[4]
- Pituitary adenoma: The typical cytological features are epithelial cords and sheets with dyscohesive ends, acini, and papillary formations. Cytoplasmic content, depending on functional status, is variably clear, with vacuolation or eosinophilic bodies and occasional paranuclear bodies. Mild cellular pleomorphism and binucleated forms are common.[4,34] Pituitary adenomas possess distinct cytologic features and require distinction from normal pituitary tissues, germinomas, meningiomas, craniopharyngiomas, and oligodendrogliomas.[34]
- Medulloblastoma: The cells appear mostly discohesive with scant cytoplasm, and easily identified Homer-Wright rosettes are usually seen.[35] Similarly, the hallmark of medulloblastoma is small, round, monomorphic cells with hyperchromatic nuclei and a high nuclear cytoplasmic ratio. Crush artefacts and apoptotic bodies can also be identified[4,35]
- Mixed glioma: In cases of mixed gliomas, SCS interpretation and impression are difficult due to the predominance of one of the histological types, thereby causing inaccurate diagnosis. This shortcoming was noticed by Kini, et al., who performed a study on 100 intra-operative biopsies.[36] The smaller component of mixed glial tumor may be missed, especially if they are the less numerous oligodendroglia cells, partly due to sampling error but also because of the dense fibrillary background of the astrocytic component. Differentiation of astrocytic from oligodendroglia elements is of relevance during stereotactic biopsies. Due to prompt fixation, the diagnostic oligodendroglial artifacts from formalin fixation are not appreciated in squash smears, which may lead to a misinterpretation.[36]
- Metastatic tumor: The most common tumors to metastasize to the brain include adenocarcinomas, especially from the lung and breast, small cell or neuroendocrine-type carcinomas, renal cell carcinoma, and melanoma. The common cytological findings of metastatic tumors are the presence of pleomorphic cells, vascular proliferation, and necrosis. However, these tumors impart the cytological features of their site of origin.[4] The vascular features of metastatic brain tumors may mimic those of glioblastomas, thereby leading to an inaccurate diagnosis in SCS.[37]
Objectives:
- To evaluate the value of squash smear cytology in rapid intra-operative diagnosis of central nervous system (CNS) tumors and its correlation with final histological diagnosis.
- To evaluate the CNS tumors in relation to site, age, and sex.
- To compare the accuracy of squash cytological diagnosis of CNS tumors with that of histopathological diagnosis.
- To establish the role of squash cytology in the rapid assessment of CNS tumors in a limited resource setting.
Methodology
It was a cross-sectional (Descriptive) study over one year (December 1, 2015, to November 30, 2016). All patients who underwent operations for CNS mass lesions, including those with intra-operative squash cytology and post-operative histopathological samples received at the Department of Pathology, were included. Cases of squash cytology without a histopathological sample were excluded.
Specimen collection: Intra-operative specimens were received in normal saline. Squash smears were prepared by placing the tissue on the dry slide and crushing it using another glass slide, followed by fixation in 95% propanol for 10 minutes, and then processed for Giemsa and pap stain. The post-operative specimens received in 10% formalin were processed for histopathological examination; sections were stained with H&E stain.
Data collection and management: Specimen of intra-operative and post-operative CNS tumors received at the Department of Pathology was taken for study, after informed consent. A brief history, along with radiological findings, was taken from the requisition form and filled in a Proforma structured for this study. All the data were entered into Microsoft Excel XP. The results were then analyzed by IBM SPSS-20. Approval for the research was obtained from the IRC, Institute of Medicine, Tribhuvan University.
Results
During a period of one year of study, a total of 58 squash cytology specimens were received for intra-operative evaluation. One case was excluded from the study because the biopsy specimen was not available for histopathological evaluation. Out of 57 patients evaluated, the mean age was 39 years, with ages ranging from 4 to 73 years. A slight male predominance was observed, the Male: Female ratio being 1.1:1. The frequency of CNS tumors increased with age, and a peak was seen at 41-50 years (22.8%). Thereafter, the frequency declined to only 15.8% in the sixth decade of life. In most cases, 94.8% were seen in individuals up to 60 years of age. There were only three cases above 60 years of age (Figure 1).
In the evaluation of the location, the number of cases in the brain exceeded the spinal cord. In the brain, Glioblastoma was most common (22.8%) with the frontal lobe (61%) as the most common location, followed by the temporal lobe (23%). Similarly, only 13 cases (23%) were seen in the spinal cord, of which Schwannoma was the most common tumor (77%). The frontal lobe was the most common location, constituting 36.8%, followed by the temporal lobe. Neuroepithelial tumors constituted the most common type of tumor, followed by meningeal tumors and tumors of cranial/spinal nerves. Among Neuroepithelial tumors, Glioblastoma was the most common (Table 1).
In the evaluation of the CNS tumor as per WHO grading, Grade I CNS tumors were found to be the most common (34 cases, 59.6%). Among the grade I tumors, meningioma was the most common type (44%), followed by schwannoma (11%) and pilocytic astrocytoma (10.5%). Grade IV tumor (Glioblastoma) was the second most common tumor, followed by grade II and III tumors, respectively. In our study, test performance metrics of squash cytology to diagnose malignant tumor were Sensitivity-86.3%, Specificity-88.5%, PPV-82.6%, NPV-91.8%.
Out of 57 cases, 46 cases were correctly diagnosed in both squash cytology and histopathological examination, the concordance being 80.7% (Table 2). All 12 cases diagnosed as meningioma in squash cytology showed complete concordance in histopathological diagnosis (100% concordance). The lowest concordance was noted in pilocytic astrocytoma and oligodendroglioma, with concordance rates of 66.6% each. One of the cases of anaplastic oligodendroglioma is included within the category of oligodendroglioma. Eleven cases were discordant.
| S.No. | Squash cytology diagnosis | No. | Histopathology diagnosis | No. |
| 1. | Glioblastoma | 13 | Glioblastoma | 11 |
| Pilocytic astrocytoma | 01 | |||
| Anaplastic oligodendroglioma |
01 | |||
| 2. | Pilocytic astrocytoma | 04 | Pilocytic astrocytoma | 04 |
| 3. | Oligodendroglioma | 03 | Oligodendroglioma | 02 |
| Glioblastoma | 01 | |||
| 4. | Ependymoma | 03 | Ependymoma | 02 |
| Schwannoma | 01 | |||
| 5. | Myxopapillary ependymoma | 01 | Myxopapillary ependymoma | 01 |
| 6. | Meningothelial meningioma | 05 | Meningothelial meningioma | 05 |
| 7. | Transitional meningioma | 07 | Transitional meningioma
Meningothelial meningioma Psammomatous |
04
02 01 |
| 8. | Schwannoma | 08 | Schwannoma | 08 |
| 9. | Craniopharyngioma | 02 | Craniopharyngioma | 01 |
| Pilocytic astrocytoma | 01 | |||
| 10. | Neurofibroma | 02 | Neurofibroma | 01 |
| Schwannoma | 01 | |||
| 11. | Non-Hodgkin lymphoma | 01 | Non-Hodgkin lymphoma | 01 |
| 12. | Chordoma | 01 | Chordoma | 01 |
| 13. | Pituitary adenoma | 01 | Pituitary adenoma | 01 |
| 14. | Central neurocytoma | 01 | Central neurocytoma | 01 |
| 15. | Metastasis | 01 | Glioblastoma | 01 |
| 16. | Blood only | 01 | Cavernous hemangioma | 01 |
| 17. | Necrosis and occasional plasma cells | 01 | Plasmacytoma | 01 |
| 18. | Few ganglion cells and neurons in the fibrillary stroma | 01 | Anaplastic gangliocytoma | 01 |
| 19. | Few spindle cells | 01 | Schwannoma | 01 |
| Total | 57 | Total | 57 |
Table 1: Comparison of squash cytology diagnosis with corresponding histopathology
|
S.No.
|
Histopathology diagnosis | Number of cases | Concordant squash cytology diagnosis (Frequency) |
Concordance (%) |
| 1. | Glioblastoma | 13 | 11 | 84.6 |
| 2. | Pilocytic astrocytoma | 06 | 04 | 66.6 |
| 3. | Oligodendroglioma | 03 | 02 | 66.6 |
| 4. | Ependymoma | 03 | 03 | 100 |
| 5. | Meningioma | 12 | 12 | 100 |
| 6. | Schwannoma | 11 | 08 | 81.8 |
| 7. | Craniopharyngioma | 01 | 01 | 100 |
| 8. | NHL | 01 | 01 | 100 |
| 9. | Chordoma | 01 | 01 | 100 |
| 10. | Central Neurocytoma | 01 | 01 | 100 |
| 11. | Pituitary adenoma | 01 | 01 | 100 |
| 12.
|
Neurofibroma
Plasmacytoma Cavernous haemangioma Anaplastic gangliocytoma |
01
01 01 01 |
01
00 00 00 |
100
0 0 0 |
| Total | 57 | 46 | 80.7% |
Table 2: Concordance between squash cytology and histopathology
The widest age distribution was observed in grade I tumors, followed by grade IV tumors. Grade I tumors range from the first to the ninth decade, with a peak incidence in the fifth decade, and grade IV tumors range from the first to the eighth decade, with a peak incidence at the sixth decade. The age distribution of grade II tumors was from the first to the fourth decade. The narrowest age distribution was in grade III tumors, ranging from the third to the fifth decade only (Figure 1).
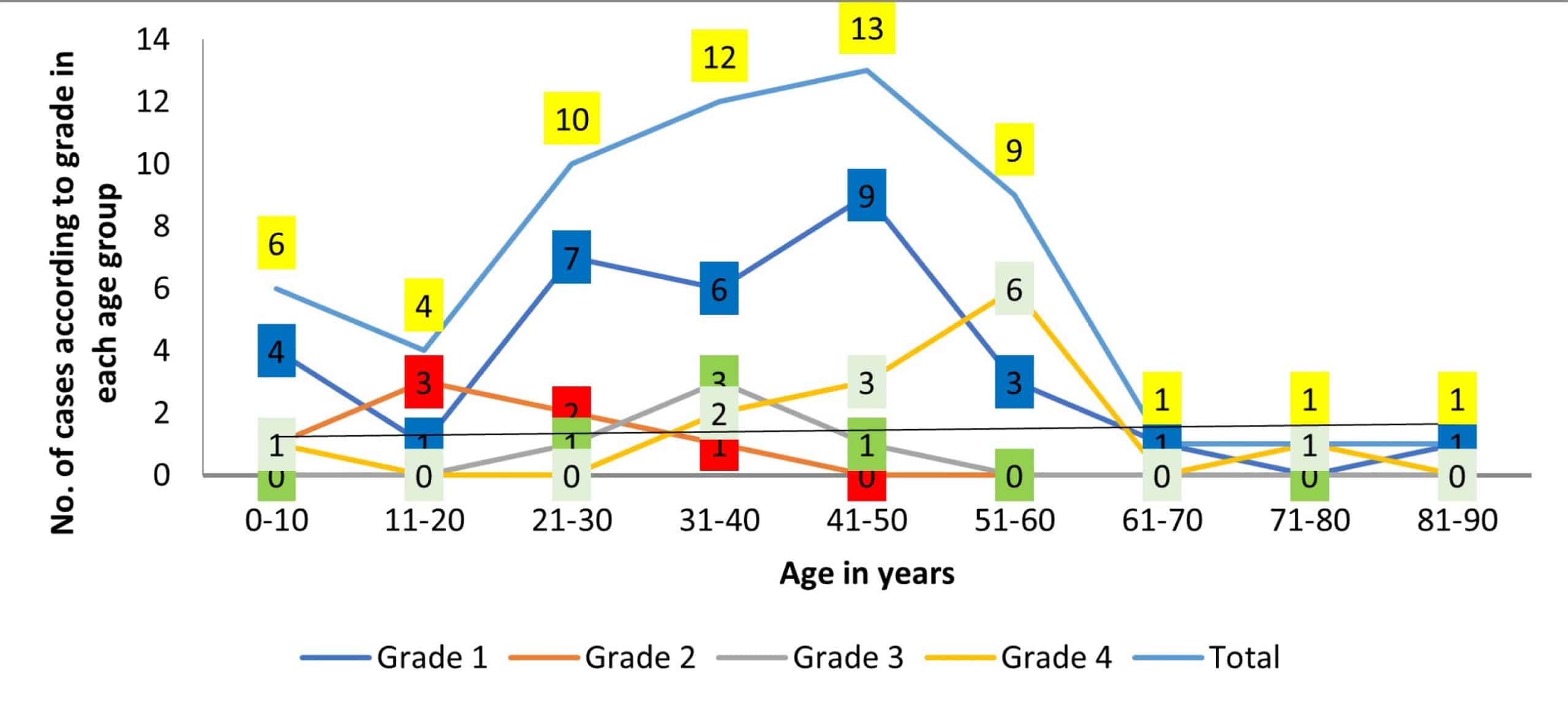
Figure 1: Age-wise distribution according to various grades of CNS tumors
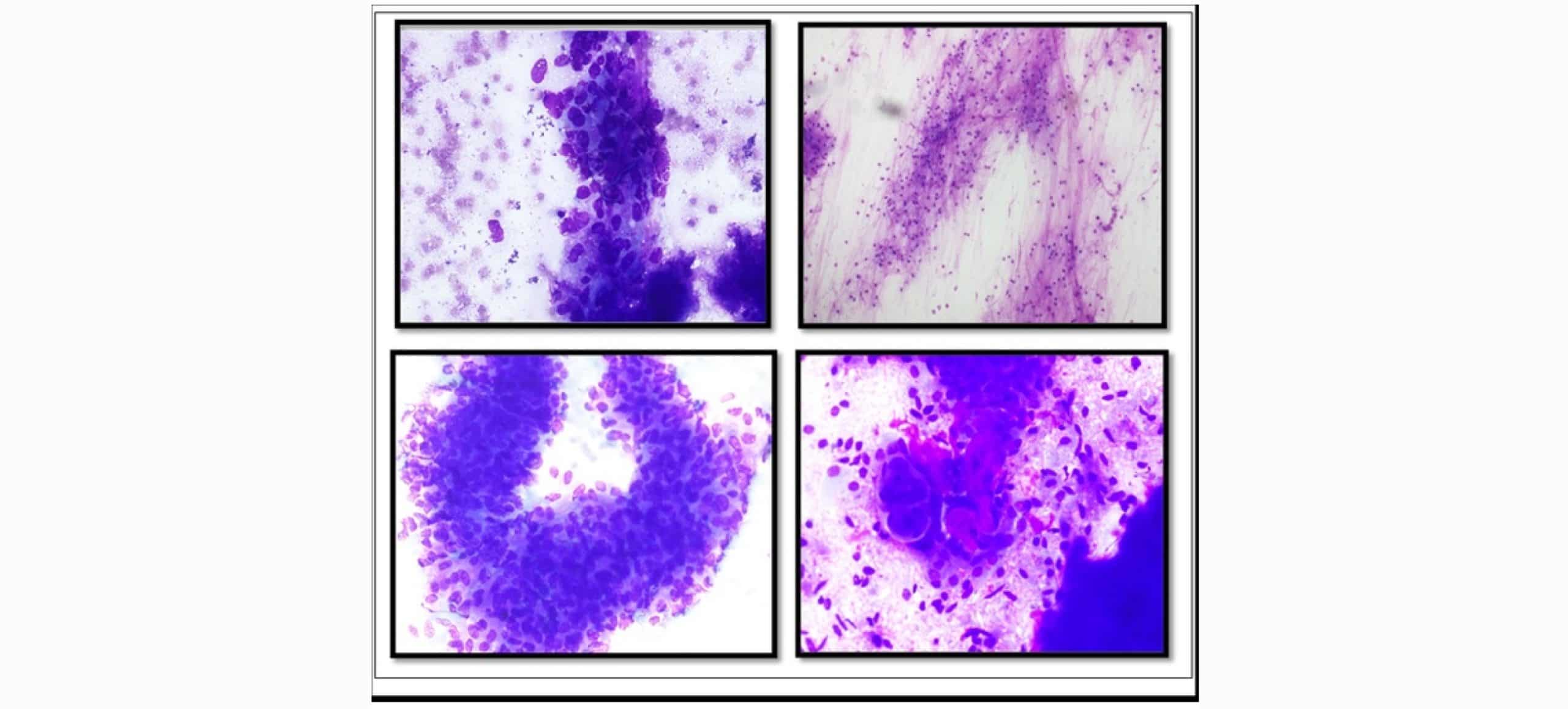
Figure 2: A. Glioblastoma showing sausage-shaped pleomorphic tumor cells. (Giemsa, 40X). B. Pilocytic astrocytoma showing piloid cells with long hair-like processes (Giemsa, 20X). C. Ependymoma showing tumor cells arranged in papillary architecture of epithelioid-like and sparse glial processes (Giemsa, 40X). D. Meningioma showing syncytial cells, slightly spindly, with indistinct boundaries, with a whirling pattern (Giemsa, 40X)
Discussion
Intraoperative diagnosis of CNS lesions plays a crucial role in making decisions on optimal operative management. In this regard, squash cytology provides rapid intraoperative diagnosis. A lot of studies and research have been done to prove the efficacy of squash cytology to ascertain the diagnosis of CNS lesions, and the results have shown that squash smear preparation is a fairly accurate, simple, and reliable tool for rapid intra-operative diagnosis of central nervous system lesions.[7,8,16,38,39] In this study, the age of the patients ranged from 4 years to 83 years with a slight male predilection (1.1:1). (Figure 1) An epidemiological study done by Fisher and Rickert showed that CNS tumors are more common in males compared to females, the ratio ranging from 1.1 to 3.5:1.[40,41] A lot of studies on CNS tumors have shown that the incidence of CNS tumors is slightly higher in males than in females.[23,25]
The frequency of CNS tumors increased with increasing age, with a peak in the fourth decade and then declined thereafter (Fig. 1). The widest age distribution was seen for grade I tumors, ranging from the first to the ninth decade, with a peak at the fifth decade of age. Grade IV tumor (glioblastoma) also showed a wide age distribution ranging from the first to the eighth decade of life, with a peak at the sixth decade. Grade II and III tumors were seen in the first to fourth decade and third to fifth decade of life, respectively. In a retrospective study of Arora et al, which included the cases of the past eight years, the peak incidence of CNS tumors was in the age group of 75-79 years.[42] A study by Govindaraman et al showed that the peak incidence of brain tumors was observed in individuals aged 31–40 years.[43] Walker et al in the United States, included 157 hospitals, showed that the rates of primary intracranial neoplasm increase steadily with advancing age.[44] The frequency of CNS tumors in our study was 5.3% in the first decade and 12.3% in the second decade of life. Several studies have shown that the incidence of CNS tumors is now increasing in younger age groups.[45-48]
In this study, most of the tumors (77%) were seen within the brain, followed by the spinal cord. Out of 44 brain tumors, the most common location was the frontal lobe (36.8%), followed by the frontoparietal lobe and the posterior fossa. This was similar to a study done by Ahmed et al in Karachi, in which the frontal lobe comprised 49.2%.[49] In a study done by Nanarng et al, the cerebral hemisphere was the most common site (66.6%), followed by the spinal cord (13.3%), with the frontoparietal lobe as the predominant site within the brain.[25] In our study, the frequency of CNS tumors in the frontal and frontoparietal lobes was recorded separately, and the frontoparietal lobe was the third most common site. However, 42.1% of tumors were seen in the frontal and frontoparietal lobes when combined.
Previous studies on CNS tumors have shown that the diagnostic accuracy of squash cytology ranges from 75% to 95%.[50,51,44] In a retrospective study conducted by Roessler et al, over a period of 15 years among 4,172 patients, complete correlation with the final diagnosis was achieved with a mean of 89.8%.[50] In a study by Govindaraman, the overall diagnostic accuracy was 90.6%.[43] Likewise, a study done by Bhardwaj in 70 cases showed that the accuracy of squash cytology was 98.5%.[52] Bajaj et al. also evaluated 72 cases of CNS neoplasm, and the overall diagnostic accuracy of squash was 98.6%.[53] In our study, the diagnostic accuracy of squash cytology was 80.7%(Table 2). A concordance rate of 100% was seen in 21 cases (36.8%). There were 12 cases of meningioma, two cases of ependymoma, and one case each of craniopharyngioma, non-Hodgkin lymphoma (NHL), chordoma, central neurocytoma, pituitary adenoma, and neurofibroma, which showed 100% concordance. However, such a high concordance was not seen (except for meningioma) when the number of cases increased, as in 13 cases of glioblastoma and eight cases of schwannoma, which showed 84.6% and 81.8% of concordance, respectively. (Table 2) In this study, the sensitivity and specificity of squash cytology were 86.3% and 88.5% respectively, which matched the various previous studies with a sensitivity of squash cytology ranging from 88-98%.[54,8]
In the present study, glial tumors constituted the most common neoplasm (42.0%), of which glioblastoma was the most common, 22.8%of total cases, and 54% of glial tumors only.(Table1) These findings are similar to the study done by Govindaraman et al, which showed glial tumor (33.3%) as the most common, followed by meningioma (24%) and schwannoma (18.6%).[43] In a study with a large sample size (44,497 cases), which was done by Crocetti in Europe, similar finding was observed (glioblastoma: 57.6% of all cases).[46] Ohgaki and Kleiheus, in their study, also showed that glioblastoma was the most frequent and most aggressive brain tumor, with the frontal lobe being the most preferred location.[55]
In this study, the male-to-female ratio for glioblastoma was just 0.8:1. However, several other studies have shown that males are slightly more affected than females, which is consistent with studies done by Fleury A et al and others.[56-58] Most of the cases (11 of 13) of tumors were seen in the fourth to sixth decades of life (Figure 1). Only one case each was seen in the first and seventh decades of life. Squash smears of 11 cases of glioblastoma offered complete correlation with histology (Table 2). They showed moderate to markedly cellular smear in a paucifibrillary background. Nuclear atypia was marked (Figure 2A). Endothelial proliferation and necrosis were also noted. Mitotic figures and giant cells were also seen in some smears. Among the two discordant cases, one was diagnosed as a pilocytic astrocytoma and another as an anaplastic oligodendroglioma in histopathology (Table 1). In the case of pilocytic astrocytoma, the glial background was interpreted as necrosis, and thick-walled blood vessels, along with a few aggregates of atypical-looking cells, were specified in squash cytology, leading to the diagnosis of high-grade glioma. Rosenthal fibers and eosinophilic granular bodies were also not seen. Some cases of pilocytic astrocytoma showing thick poloidal background and some pleomorphic or multinucleated cells with thick blood vessels, but without Rosenthal fibers or eosinophilic granular bodies, could be diagnosed as high-grade gliomas in squash cytology.[4] Rao et al also described a case of pilocytic astrocytoma, which was overgraded and misdiagnosed as glioblastoma in squash cytology due to vascularity and nuclear atypia.[59]
Meningioma was the second most common tumor in this study, comprising 12 cases, and the concordance was 100% (Table 2). However, regarding subtypes, only meningothelial meningioma showed 100% concordance, whereas, among seven cases diagnosed as transitional meningioma in squash cytology, only four cases showed concordance, while two cases were meningothelial meningioma and one case was psammomatous meningioma in histopathology. A squash cytology study done by Ali et al in 107 cases of meningioma concluded that typing and grading showed inaccurate results.[60] However, in a study done by Dumitrescu, it was also reported that all cases of meningioma showed complete correlation between squash and histopathological diagnosis.[61]
Schwannoma was the third most common type of tumor in this study. Out of 11 cases of histopathologically confirmed schwannoma, eight cases were correctly diagnosed as schwannoma in squash cytology. Out of the remaining three cases, one was diagnosed as ependymoma and another as neurofibroma in squash cytology (Table 1). The third case showed blood and a few spindled cells in squash smears, and no definite opinion was given. This might be due to the tough nature of the tissue, which offered resistance in smearing with tissue fragments attached to the slide.[4] In a study by Martinez et al, it was stated that some cases of schwannoma yield low cellularity in squash preparation, and hence render the diagnosis inadequate in cytological examination.[62]
Out of six cases of confirmed pilocytic astrocytoma in this study, four cases were correctly diagnosed in squash cytology (Table 1). One case was cytologically diagnosed as craniopharyngioma and another as glioblastoma. In the case of craniopharyngioma, smears showed possible reactive changes and abundant glial processes, leading to an inaccurate diagnosis of pilocytic astrocytoma. A poorly sampled tumor of craniopharyngioma could show substantial reactive and inflammatory changes, leading to differential diagnosis of pilocytic astrocytoma.[4] Out of four cases of ependymoma diagnosed in squash cytology, three cases showed concordance with histopathological diagnosis (Table 1 and Table 2). One of the cases was diagnosed as a schwannoma in cytology. In squash preparation, smears were moderately cellular with ependymal cells arranged around vessels having a prominent fibrillary background. The cells exhibited mild pleomorphism with round to elongated nuclei having speckled chromatin. Some of the cases showed signet ring and plasmacytoid cells as well. Perivascular pseudo-rosettes were also seen. Manasa et al had also stated that perivascular pseudo-rosettes were more readily presented in a squash smear in the case of ependymoma.[30] Schwannoma also shows elongated or spindled cells in a rather less cellular smear with the presence of blood vessels in hypocellular areas, with attached tumor cells mimicking perivascular pseudo-rosette, thereby falsely resembling ependymoma.[4]
Three cases of oligodendroglioma were identified, of which only two cases were concordant with histopathological diagnosis (Table 1 and Table 2). One of the cases was diagnosed as glioblastoma in histopathology. Oligodendrogliomas look like astrocytic tumors but can be distinguished by virtue of their rounder, smoother, and more monotonous nuclei than the elongated and folded nuclear membranes of the latter. Likewise, nuclei are more sausage-shaped and pleomorphic in glioblastoma, which can help to distinguish oligodendroglioma from glioblastoma.[4] Among the two cases of neurofibroma, one was confirmed on histopathological evaluation, while the other was diagnosed as schwannoma (Table 1 and Table 2). Squash smear of neurofibroma revealed low cellularity with spindle-shaped cells having wavy nuclei and a myxoid background. So, the proper diagnosis was offered in one of the cases. The presence of spindled-shaped cells in squash smears could misguide the pathologist towards schwannoma and hence should be examined thoroughly. Martinez et al. showed 100% concordance and stated relatively low cellularity as a clue to schwannoma.[62]
Craniopharyngioma was diagnosed in two cases. One was confirmed histopathologically, while another was diagnosed as pilocytic astrocytoma (Table 1 and Table 2). Squash smear showed sheets of squamous cells in a background consisting of keratinous debris. So, a correct diagnosis was offered in this case. In the study done by Kini et al, besides these features, the background also showed cholesterol crystals, calcified debris, foreign body-type giant cells, and histiocytes, which were not observed in our case.[36] One case each of NHL, pituitary adenoma, chordoma, and central neurocytoma diagnosed in squash cytology showed complete concordance with histopathology diagnosis (Table 1 and Table 2). However, given just one case each, the results could not be generalized.
Recommendation: Squash cytology findings, if interpreted in an algorithm together with the clinical picture and imaging findings, will help to reach an accurate and rapid diagnosis of intracranial tumors. Due to its simplicity, cost-effectiveness, and rapid diagnostic yield, squash cytology can be used as an alternative to frozen section in a resource-limited setting. However, we recommend further study with a large number of cases to establish the definite role and efficacy of squash cytology.
Conclusion
Squash cytology is a reliable, fairly accurate, and rapid tool in the intraoperative diagnosis of CNS tumors. This study supported the feasibility of squash cytology for intra-operative diagnosis and as an alternative to frozen section. In this study, the concordance rate between squash cytology and histopathology was as high as 80 to 100%. There was 100% accuracy of squash cytology in the case of meningioma and ependymoma, followed by glioblastoma and schwannoma. Peak age was in the fifth decade, and grade I tumors were most common, followed by grade IV, and both showed wide age distribution, ranging from the first to the ninth decade for the former and up to the eighth decade for the latter. Glial tumors were the most common nervous system tumors, and glioblastoma constituted slightly more than 50% of them. Tumors of the brain exceeded the spinal cord, and the frontal lobe was the most common location.
References
- Arora RS, Alston RD, Eden TO, Estlin EJ, Moran A, Birch JM. Age-incidence patterns of primary CNS tumors in children, adolescents, and adults in England. Neuro Oncol. 2009;11(4):403-413. doi:10.1215/15228517-2008-097
PubMed | Crossref | Google Scholar - Gray W, Kocjan G. Diagnostic Cytopathology E-Book: Expert Consult: Online and Print. 3rd ed, rev. Elsevier Health Sciences; 2010.
Diagnostic Cytopathology - Aker FV, Hakan T, Karadereler S, Erkan M. Accuracy and diagnostic yield of stereotactic biopsy in the diagnosis of brain masses: comparison of results of biopsy and resected surgical specimens. Neuropathology. 2005;25(3):207-213. doi:10.1111/j.1440-1789.2005.00634.x
PubMed | Crossref | Google Scholar - Joseph JT. Regional tumors. In: Joseph JT, Pine J, McGough J, Dougherty B, Rivera B, Panetta A, et al., editors. Diagnostic Neuropathology Smears. 1st ed. Walsworth Publishing Co; 2007:1-234.
Diagnostic Neuropathology Smears - Firlik KS, Martinez AJ, Lunsford LD. Use of cytological preparations for the intraoperative diagnosis of stereotactically obtained brain biopsies: a 19-year experience and survey of neuropathologists. J Neurosurg. 1999;91(3):454-458. doi:10.3171/jns.1999.91.3.0454
PubMed | Crossref | Google Scholar - Pawar N, Deshpande K, Surase S, D’Costa G, Balgi S, Goel A. Evaluation of the squash smear technique in the rapid diagnosis of central nervous system tumors: A cytomorphological study. Internet J Pathol. 2009;11(1).
Evaluation of the Squash Smear Technique in the Rapid Diagnosis of Central Nervous System Tumors: A… - Beuvon F, Varlet P, Fallet-Bianco C, Daumas-Duport C. The “smears” technique for the extemporaneous examination: diagnostic contribution to neurosurgical pathology. Ann Pathol. 2000;20(5):499-506.
The “smears” technique for the extemporaneous examination: diagnostic contribution to neurosurgical… - Sharma S, Deb P. Intraoperative neurocytology of primary central nervous system neoplasia: A simplified and practical diagnostic approach. J Cytol. 2011;28(4):147-158. doi:10.4103/0970-9371.86339
PubMed | Crossref | Google Scholar - Eisenhardt L, Cushing H. Diagnosis of Intracranial Tumors by Supravital Technique. Am J Pathol. 1930;6(5):541-552.7.
Diagnosis of Intracranial Tumors by Supravital Technique - Roessler K, Dietrich W, Kitz K. High diagnostic accuracy of cytologic smears of central nervous system tumors. A 15-year experience based on 4,172 patients. Acta Cytol. 2002;46(4):667-674. doi:10.1159/000326973
PubMed | Crossref | Google Scholar - Mitra S, Kumar M, Sharma V, Mukhopadhyay D. Squash preparation: A reliable diagnostic tool in the intraoperative diagnosis of central nervous system tumors. J Cytol. 2010;27(3):81-85. doi:10.4103/0970-9371.71870
PubMed | Crossref | Google Scholar - Berkeley BB, Adams JH, Doyle D, Graham DI, Harper CG. The smear technique in the diagnosis of neurosurgical biopsies. N Z Med J. 1978;87(603):12-15.
The smear technique in the diagnosis of neurosurgical biopsies - Agrawal M, Chandrakar SK, Lokwani D, Purohit MR. Squash cytology in neurosurgical practice: a useful method in resource-limited setting with lack of frozen section facility. J Clin Diagn Res. 2014;8(10):FC09-C12. doi:10.7860/JCDR/2014/10142.4921
PubMed | Crossref | Google Scholar - Kumar V, Abbas AK, Aster JC, editors. Robbins & Cotran Pathologic Basis of Disease. 9th ed. Elsevier; 2014.
Robbins & Cotran Pathologic Basis of Disease - Panth R. A systematic approach to cytological evaluation of central nervous system tumour. J Pathol Nepal. 2011;1(2):136-141. doi:10.3126/jpn.v1i2.5409
Crossref | Google Scholar - Jaiswal S, Vij M, Jaiswal AK, Behari S. Intraoperative squash cytology of central nervous system lesions: a single center study of 326 cases. Diagn Cytopathol. 2012;40(2):104-112. doi:10.1002/dc.21506
PubMed | Crossref | Google Scholar - Kamatchi V, Babu NA, Sankari SL, Rajesh E. Imprint cytology. J Pharm Bioallied Sci. 2015;7(Suppl 1):S207-208. doi:10.4103/0975-7406.155905
PubMed | Crossref | Google Scholar - Khamechian T, Alizargar J, Mazoochi T. The value of touch preparation for rapid diagnosis of brain tumors as an intraoperative consultation. Iran J Med Sci. 2012;37(2):105-111.
The Value of Touch Preparation for Rapid Diagnosis of Brain Tumors as an Intraoperative Consultation - Khamechian T, Alizargar J, Mazoochi T. The value of touch preparation for rapid diagnosis of brain tumors as an intraoperative consultation. Iran J Med Sci. 2012;37(2):471-476.
The Value of Touch Preparation for Rapid Diagnosis of Brain Tumors as an Intraoperative Consultation - Folkerth RD. Smears and frozen sections in the intraoperative diagnosis of central nervous system lesions. Neurosurg Clin N Am. 1994;5(1):1-18.
Smears and frozen sections in the intraoperative diagnosis of central nervous system lesions - Das R, Choudhury S, Mallik NC. Diagnostic accuracy of squash cytology in central nervous system tumours. J Evid Based Med Healthc. 2018;5(44):3062-3065. doi:10.18410/jebmh/2018/625
Crossref - Collaço LM, Tani E, Lindblom I, Skoog L. Stereotactic biopsy and cytological diagnosis of solid and cystic intracranial lesions. Cytopathology. 2003;14(3):131-135. doi:10.1046/j.1365-2303.2003.00046.x
PubMed | Crossref | Google Scholar - Hernandez O, Zagzag D, Kelly P, Golfinos J, Levine PH. Cytological diagnosis of cystic brain tumors: a retrospective study of 88 cases. Diagn Cytopathol. 2004;31(4):221-228. doi:10.1002/dc.20112
PubMed | Crossref | Google Scholar - Kresak JL, Rivera-Zengotita M, Foss RM, Yachnis AT. CNS intraoperative consultation: a survival guide for non-neuropathologists. Methods Mol Biol. 2014;1180:369-376. doi:10.1007/978-1-4939-1050-2_22
PubMed | Crossref | Google Scholar - Nanarng V, Jacob S, Mahapatra D, Mathew JE. Intraoperative diagnosis of central nervous system lesions: Comparison of squash smear, touch imprint, and frozen section. J Cytol. 2015;32(3):153-158. doi:10.4103/0970-9371.168835
PubMed | Crossref | Google Scholar - Mouriquand C, Benabid AL, Breyton M. Stereotaxic cytology of brain tumors. Review of an eight-year experience. Acta Cytol. 1987;31(6):756-764.
Stereotaxic cytology of brain tumors. Review of an eight-year experience - Di Stefano D, Scucchi LF, Cosentino L, Bosman C, Vecchione A. Intraoperative diagnosis of nervous system lesions. Acta Cytol. 1998;42(2):346-356. doi:10.1159/000331614
PubMed | Crossref | Google Scholar - Teo JG, Ng HK. Cytodiagnosis of pilocytic astrocytoma in smear preparations. Acta Cytol. 1998;42(3):673-678. doi:10.1159/000331825
PubMed | Crossref | Google Scholar - Khalid A, Haque AU. Touch impression cytology versus Frozen section as intraoperative consultation diagnosis. Int J Pathol. 2018:63-70.
Touch impression cytology versus Frozen section as intraoperative consultation diagnosis - Manasa PL, Uppin MS, Sundaram C. Analysis of squash smear cytology of ependymomas. Acta Cytol. 2012;56(2):183-188. doi:10.1159/000336146
PubMed | Crossref | Google Scholar - Kobayashi S. Meningioma, neurilemmoma and astrocytoma specimens obtained with the squash method for cytodiagnosis. A cytologic and immunochemical study. Acta Cytol. 1993;37(6):913-922.
Meningioma, neurilemmoma and astrocytoma specimens obtained with the squash method for cytodiagnosi… - Bhuyan P. Primary CNS lymphoma- a squash diagnosis. J Cytol Histol. 2011;2(6):1-3. doi:10.4172/2157-7099.1000127
Crossref | Google Scholar - Myageri A, Vandana UG, Melkundi S, Patil PB, Kulkarni P. Squash cytology of primary central nervous system lymphoma in an immunocompetent patient. Asian J Neurosurg. 2013;8(4):195-198. doi:10.4103/1793-5482.125673
PubMed | Crossref | Google Scholar - Ng HK. Smears in the diagnosis of pituitary adenomas. Acta Cytol. 1998;42(3):614-618. doi:10.1159/000331816
PubMed | Crossref | Google Scholar - Adesina AM. Intraoperative consultation in the diagnosis of pediatric brain tumors. Arch Pathol Lab Med. 2005;129(12):1653-1660. doi:10.5858/2005-129-1653-ICITDO
PubMed | Crossref | Google Scholar - Kini JR, Jeyraj V, Jayaprakash CS, Indira S, Naik CN. Intraoperative consultation and smear cytology in the diagnosis of brain tumours. Kathmandu Univ Med J (KUMJ). 2008;6(24):453-457. doi:10.3126/kumj.v6i4.1734
PubMed | Crossref | Google Scholar - Gi T, Sato Y, Tokumitsu T, et al. Microvascular proliferation of brain metastases mimics glioblastomas in squash cytology. Cytopathology. 2017;28(3):228-234. doi:10.1111/cyt.12405
PubMed | Crossref | Google Scholar - Sharma N, Misra V, Singh PA, Gupta SK, Debnath S, Nautiya A. Comparative efficacy of imprint and squash cytology in diagnosing lesions of the central nervous system. Asian Pac J Cancer Prev. 2011;12(7):1693-1696.
Comparative efficacy of imprint and squash cytology in diagnosing lesions of the central nervous sy… - Krishnani N, Kumari N, Behari S, Rana C, Gupta P. Intraoperative squash cytology: accuracy and impact on immediate surgical management of central nervous system tumours. Cytopathology. 2012;23(5):308-314. doi:10.1111/j.1365-2303.2011.00905.x
PubMed | Crossref | Google Scholar - Fisher JL, Schwartzbaum JA, Wrensch M, Wiemels JL. Epidemiology of brain tumors. Neurol Clin. 2007;25(4):867-vii. doi:10.1016/j.ncl.2007.07.002
PubMed | Crossref | Google Scholar - Rickert CH, Paulus W. Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Childs Nerv Syst. 2001;17(9):503-511. doi:10.1007/s003810100496
PubMed | Crossref | Google Scholar - Porter KR, McCarthy BJ, Freels S, Kim Y, Davis FG. Prevalence estimates for primary brain tumors in the United States by age, gender, behavior, and histology. Neuro Oncol. 2010;12(6):520-527. doi:10.1093/neuonc/nop066
PubMed | Crossref | Google Scholar - Govindaraman PK, Arumugam N, Ramasamy C, Prakasam G. Role of squash smear in intraoperative consultation of central nervous system tumors. J Sci Soc. 2017;44(1):7-14. doi:10.4103/jss.JSS_36_16
Crossref | Google Scholar - Walker AE, Robins M, Weinfeld FD. Epidemiology of brain tumors: the national survey of intracranial neoplasms. Neurology. 1985;35(2):219-226. doi:10.1212/wnl.35.2.219
PubMed | Crossref | Google Scholar - Farwell JR, Dohrmann GJ, Flannery JT. Central nervous system tumors in children. Cancer. 1977;40(6):3123-3132. doi:10.1002/1097-0142(197712)40:6<3123::aid-cncr2820400656>3.0.co;2-6
PubMed | Crossref | Google Scholar - Crocetti E, Trama A, Stiller C, et al. Epidemiology of glial and non-glial brain tumours in Europe. Eur J Cancer. 2012;48(10):1532-1542. doi:10.1016/j.ejca.2011.12.013
PubMed | Crossref | Google Scholar - Birch JM, Alston RD, Kelsey AM, Quinn MJ, Babb P, McNally RJ. Classification and incidence of cancers in adolescents and young adults in England 1979-1997. Br J Cancer. 2002;87(11):1267-1274. doi:10.1038/sj.bjc.6600647
PubMed | Crossref | Google Scholar - Kaderali Z, Lamberti-Pasculli M, Rutka JT. The changing epidemiology of paediatric brain tumours: a review from the Hospital for Sick Children. Childs Nerv Syst. 2009;25(7):787-793. doi:10.1007/s00381-008-0771-9
PubMed | Crossref | Google Scholar - Ahmed Z, Muzaffar S, Kayani N, Pervez S, Husainy AS, Hasan SH. Histological pattern of central nervous system neoplasms. J Pak Med Assoc. 2001;51(4):154-157.
Histological pattern of central nervous system neoplasms - Mitra S, Kumar M, Sharma V, Mukhopadhyay D. Squash preparation: A reliable diagnostic tool in the intraoperative diagnosis of central nervous system tumors. J Cytol. 2010;27(3):81-85. doi:10.4103/0970-9371.71870
PubMed | Crossref | Google Scholar - Guarda LA. Intraoperative cytologic diagnosis: evaluation of 370 consecutive intraoperative cytologies. Diagn Cytopathol. 1990;6(4):235-242. doi:10.1002/dc.2840060403
PubMed | Crossref | Google Scholar - Bhardwaj K, Kriplani D, Bhake A, Bhardwaj K. Study of intraoperative squash cytology of intracranial and spinal cord tumors. Int J Res Med Sci. 2015;3(11):3101-3108. doi:10.18203/2320-6012.ijrms20151144
Crossref - Philip SA, Bai EL, Padmaja GJV, Kumari S. Analysis of Intraoperative Squash Cytology of Central Nervous System Lesions and its Correlation with Immunohistopathology and Radiology. J Cytol. 2023;40(1):1-4. doi:10.4103/joc.joc_70_22
PubMed | Crossref | Google Scholar - Patil SS, Kudrimoti JK, Agarwal RD, Jadhav MV, Chuge A. Utility of squash smear cytology in intraoperative diagnosis of central nervous system tumors. J Cytol. 2016;33(4):205-209. doi:10.4103/0970-9371.190442
PubMed | Crossref | Google Scholar - Ohgaki H, Kleihues P. The definition of primary and secondary glioblastoma. Clin Cancer Res. 2013;19(4):764-772. doi:10.1158/1078-0432.CCR-12-3002
PubMed | Crossref | Google Scholar - Fleury A, Menegoz F, Grosclaude P, et al. Descriptive epidemiology of cerebral gliomas in France. Cancer. 1997;79(6):1195-1202. doi:10.1002/(sici)1097-0142(19970315)79:6<1195::aid-cncr19>3.0.co;2-v
PubMed | Crossref | Google Scholar - Ohgaki H, Dessen P, Jourde B, et al. Genetic pathways to glioblastoma: a population-based study. Cancer Res. 2004;64(19):6892-6899. doi:10.1158/0008-5472.CAN-04-1337
PubMed | Crossref | Google Scholar - Hess KR, Broglio KR, Bondy ML. Adult glioma incidence trends in the United States, 1977-2000. Cancer. 2004;101(10):2293-2299. doi:10.1002/cncr.20621
PubMed | Crossref | Google Scholar - Rao S, Rajkumar A, Ehtesham MD, Duvuru P. Challenges in neurosurgical intraoperative consultation. Neurol India. 2009;57(4):464-468. doi:10.4103/0028-3886.55598
PubMed | Crossref | Google Scholar - Ali S, Nassar A, Siddiqui MT. Crush preparations of meningiomas: can grading be accomplished? Diagn Cytopathol. 2008;36(11):827-831. doi:10.1002/dc.20929
PubMed | Crossref | Google Scholar - Dumitrescu G, Sava A, Ciobanu D, et al. Intracranial meningiomas: correlations between intraoperative consultation and histopathological diagnosis. Romanian Neurosurgery. 2012.
Intracranial meningiomas: correlations between intraoperative consultation and histopathological di… - Martinez AJ, Pollack I, Hall WA, Lunsford LD. Touch preparations in the rapid intraoperative diagnosis of central nervous system lesions. A comparison with frozen sections and paraffin-embedded sections. Mod Pathol. 1988;1(5):378-384.
Touch preparations in the rapid intraoperative diagnosis of central nervous system lesions. A compa…
Acknowledgments
None
Funding
None
Author Information
Corresponding Author:
Shankar Kafle
Department of Pathology
Maharajgunj Medical Campus, Kathmandu, Nepal
Email: shankar.kafle@mmc.tu.edu.np
Co-Authors:
Bibek KC, Jha A, Manandhar U
Department of Pathology
Maharajgunj Medical Campus, Kathmandu, Nepal
Pradhanang A
Department of Neurosurgery
Maharajgunj Medical Campus, Kathmandu, Nepal
Authors Contributions
KC B was responsible for conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles, as well as preparing the original draft. Pradhanang A and Manandhar U supervised the work, ensuring its accuracy and completeness. Jha A and Kafle S contributed through review and editing to refine the manuscript.
Ethical Approval
Approval of research was taken from the institutional research committee (IRC), Institute of Medicine, Tribhuvan University {Reference no: 116(6-11-E)2/074/075}. Written consent was taken from each patient/patient’s party to use data, image and any other information related to this research.
Conflict of Interest Statement
The authors declare no conflict of interest.
Guarantor
Dr Shankar Kafle served as the guarantor of the work.
DOI
Cite this Article
Bibek KC, Jha A, Manandhar U, Kafle S, Pradhanang A. Diagnostic Utility of Intraoperative Squash Cytology in Central Nervous System Tumors with Histopathological Correlation.medtigo J Neurol Psychiatr. 2025;2(4):e3084241. doi:10.63096/medtigo3084241 Crossref









