Author Affiliations
Abstract
Charcot’s arthropathy is a degenerative condition resulting from a reduction in the sensory and proprioceptive innervation of the affected joint. Charcot’s arthropathy is a slowly advancing degeneration resulting from the breakdown of joints in the foot and ankle, along with the adjacent bony structures. Herein, we report the case of a middle-aged man who presented with 5-years history of type-2 diabetes mellitus, non-compliant with medications, and foot deformity, most likely has Charcot’s arthropathy. Cardiopulmonary examination was normal. Neurological examinations revealed loss of sensation in the left foot. X-ray demonstrated swelling, erythema, and deformity of the left foot. A small ulcer of about 1 cm (0.9 inches) is present on the plantar surface of the foot under the first metatarsal head. This clinical condition improved after successful non-surgical treatments.
Keywords
Charcot’s arthropathy, Foot, Diabetes mellitus, Deformity, Conservative treatments, Patient education
Introduction
Neuropathic arthropathy condition named for the French neurologist Jean-Martin Charcot (1825–1893), is brought on by a gradual loss of nerve function that eventually results in the foot and ankle being destroyed [1]. Within the broader diabetic community, the occurrence of Charcot foot is estimated to range from 0.1% to 7.5%. However, in individuals with diabetes displaying evident peripheral neuropathy, this prevalence can escalate to as much as 35% [2]. The likelihood of developing a Charcot foot is not influenced by whether an individual has type I or type II diabetes. At present, the accepted view is that the disease is mediated by an uncontrolled inflammatory process in the foot once it strikes a susceptible person. The manifestation of Charcot’s arthropathy is marked by a progressive occurrence of fractures and dislocations, primarily induced by inflammation. This inflammatory process also contributes to osteolysis[3]. Dysregulated autonomic nervous system also plays a role in the pathogenesis, leading to increased blood flow and bone resorption [4]. Diagnosing an active Charcot foot primarily relies on the patient’s history and clinical observations. However, to ensure accuracy, imaging should also be conducted[5].
To treat Charcot foot completely, the foot must become plantigrade (able to rest flat on the ground), so that the patient can walk safely in a diabetic shoe that is readily available on the market [6]. Protective weight bearing is required after an active episode, involving weight-bearing devices such as prescription shoes, boots, or braces. Surgical treatment is beneficial in CN cases refractory to offloading and immobilization or in the case of recalcitrant ulcers [7]. The aim of this study was to report the clinical manifestations, imaging study, and management of Charcot’s diabetic foot.
Case Presentation
A 42-year-old man presents at the physician’s office with complaints of swelling and numbness in his foot, which have progressively worsened over the past month. His medical history is notable for type 2 diabetes mellitus diagnosed five years ago, managed with metformin, although he admits to being non-compliant with his medications. He has a 20-year history of smoking half a pack of cigarettes daily and reports no alcohol consumption or illicit drug use. Vital signs are within normal limits, including a temperature of 37°C (98.9°F), blood pressure of 130/80 mm Hg, pulse rate of 77/min, and respiratory rate of 18/min. The cardiopulmonary examination reveals no abnormalities, but examination of the left foot shows swelling, erythema, and deformity. An X-ray of the foot (Figure 1) reveals degenerative changes with the fragmentation of articular surfaces of the joint leading to subluxation and deformity. There is a small 1 cm (0.9 inches) ulcer located on the plantar surface of the foot beneath the first metatarsal head. Neurological examination indicates a loss of sensation in the left foot, while reflexes remain intact. The tibial and dorsalis pedis pulses are normal. Pinprick test and monofilament testing assess sensitivity to pain, touch, and pressure.
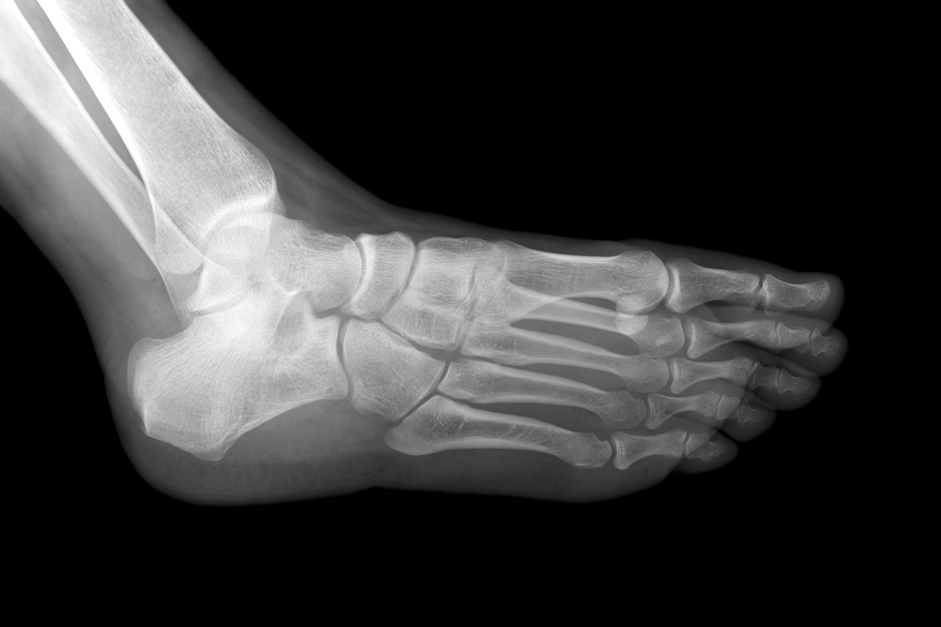
Figure 1: Left foot X-ray
Case Management
The patient received conservative treatment, and he was told to contact his primary care physician again if his symptoms persisted. Following that, he received care from several specialists, including a vascular surgeon, an endocrinologist, and his primary care physician for ongoing edema. Conservative treatments such as were used as the first line of treatment to address severe edema. After that, the patient was immobilized for 10 to 12 weeks with a non-weight-bearing cast. To prevent such incidents, the patient received education about the diabetic Charcot process and was instructed on how to wear appropriate footwear. His foot healed without incident, and no additional complications were observed. The swelling totally disappeared. The patient’s pain was controlled with a treatment regimen, especially bisphosphonates, and was seen every 3 to 4 months to monitor his overall pedal status. He still exercises caution in all that he does, and he constantly checks his feet for any negative changes.
Discussion
Diabetes affects approximately 30 million people in the United States, or 9% of the population. Diabetes-related foot issues are a serious health risk and a frequent reason for hospital stays [8]. Most foot issues experienced by diabetics are related to the disease’s damage to small blood vessels. Diabetic neuropathy can give rise to various significant foot complications, including Charcot arthropathy, also known as Charcot neuroarthropathy or simply Charcot foot. A disorder of the foot and ankle known as Charcot arthropathy, which can lead to severe deformities is brought on by an incapacity to detect injuries [8,9].
Tarsometatarsal (lisfranc) joints (type 1) are the most affected by diabetic Charcot arthropathy. Type 1 and type 2 joints of the hindfoot, type 3a and 3B joints of the ankle and posterior calcaneus, type 4 multiple regions, and type 5 forefoot joints are the next most common joint types [10]. Foot edema is the first indication of Charcot, and it can happen even in the absence of a visible wound. At first, the patient may notice that it’s difficult to fit into shoes.
The foot was examined by imaging techniques like X-rays, Computed tomography (CT) scan, MRI scan, Bone scan/indium scan. A definitive diagnosis requires a synovial fluid analysis or biopsy [4,5]. There have been reports of misdiagnoses, including gout, deep vein thrombosis, soft tissue injury, rheumatoid arthritis, and infections, delaying the diagnosis of Charcot arthropathy. Delays in diagnosis can have serious and incapacitating effects, like structural foot deformities [11]. Management includes total contact casting, reducing pressure, and preventing further joint destruction. The gold standard for treating diabetic foot ulcers is total contact casting. But to minimize complications such as iatrogenic ulceration or skin abrasions, proper training is needed for healthcare professionals [12].
Shoe-wear modifications and medications for pain control bisphosphonates, help to some extent. Because bisphosphonates can prevent osteoclastic bone resorption, they are typically used to treat disorders marked by abnormal turnover, particularly in the acute active phase of Charcot neuroarthropathy (CN) and occasionally in its chronic phases [13]. Normally, specialized footwear includes Oxford bluchers with a depth ranging from 0.6 to 1.3 cm more than standard shoes. This extra space is particularly beneficial when using insoles and foot orthoses, which are necessary in instances of foot deformities arising from Charcot arthropathy [14]. In cases of substantial foot deformity, adjustments may be needed to modify ready-made shoes accordingly. The shoes must be customized [15].
In severe cases, postoperative management with resection of bony prominences arthrodesis may correct the deformity. Patients who are refractory to these treatments require amputation [5]. Multiplane circular fixators, such as triple, pantalar, and talectomy with tibiocalcaneal or concurrent use of tibionavicular and calcaneocuboid arthrodesis, are the most often used primary arthrodesis techniques in the rearfoot/ankle CN [16]. Amputation poses a notable risk in the context of diabetic foot ulcers; however, the same level of risk is not typically associated with Charcot arthropathy unless the condition is further complicated by the presence of an ulcer [17]. Amputation of the lower limb is reserved for cases in which prior surgery proved unsuccessful due to unstable arthrodesis, recurrent ulceration, or infection [18].
Postgraduate Institute of Medical Education and Research (PGIMER) conducted phase-2 clinical trials in diabetic chronic CN patients for evaluating the efficacy of terparatide and examining the change in bone remodeling as measured by F18 SPECT CT, bone mineral density (BMD), and bone turnover markers (BTM). This randomized study was carried out in 10 diabetic patients with inactive Charcot’s foot who received teriparatide (20 µg) per day subcutaneously [19]. Weill Medical College of Cornell University carried out the cohort study in Qatari patients (enrolled 57 patients and Age: 30 to 90 years) with diabetic Charcot Foot for assessing pathophysiology by measuring complications of vascular, Charcot foot in monocyte epigenetics, kidney functioning, HBA1C gene analysis during follow-up period of 6months to 2years [20].
Recently published review article stated that to enhance adherence to the conservative Charcot treatment and prevent the long-term consequences, such as infection and ulceration, severe deformity, and amputation, it is imperative to educate patients and manage their expectations [21].
The current tertiary care center amputation rate for Charcot foot patients remains approximately 7% even with appropriate therapeutic support [22]. Diabetic Charcot foot patients have noticeably more severe cases of muscle edema and atrophy. There is no relationship between the severity of this disease and muscle atrophy. A flexor digitorum brevis muscle cross-sectional muscle area (CSA) of less than 139 mm2 in the hindfoot may be a marker of diabetic Charcot foot disease [23]. When Cerament was used, there was a significant increase in the percentage of functional limb salvage, infection clearance, and a reduction in the length of time patients required post-operative antibiotic therapy. Apart from the enhancement of their functional status, also noticed an apparently better chance of survival for the person suffering from diabetic foot disease [24].
Conclusion
This case illustrates that diabetic foot Charcot arthropathy may be associated with swelling, erythema, and deformity. Diagnosis will be further evaluated by using MRI, Bone scintigraphy in individuals with diabetes mellitus, and neuropathy. In severe conditions, surgical excision of bone and realignment arthrodesis may result in the successful salvage of a foot that might otherwise need to be amputated.
References
- Papanas N, Maltezos E. Etiology, pathophysiology, and classifications of the diabetic Charcot foot. Diabet Foot Ankle. 2013;4. doi:10.3402/dfa.v4i0.20872 PubMed | Crossref | Google Scholar
- Schoots IG, Slim FJ, Busch-Westbroek TE, Maas M. Neuro-osteoarthropathy of the foot radiologist: friend or foe? Semin Musculoskelet Radiol. 2010;14(3):365-376. doi:10.1055/s-0030-1254525 PubMed | Crossref | Google Scholar
- Jeffcoate WJ, Game F, Cavanagh PR. The role of proinflammatory cytokines in the cause of neuropathic osteoarthropathy (acute Charcot foot) in diabetes. Lancet. 2005;366(9502):2058-2061. doi:10.1016/S0140-6736(05)67029-8 PubMed | Crossref | Google Scholar
- Ergen FB, Sanverdi SE, Oznur A. Charcot foot in diabetes and an update on imaging. Diabet Foot Ankle. 2013;4. doi:10.3402/dfa.v4i0.21884 PubMed | Crossref | Google Scholar
- Molines L, Darmon P, Raccah D. Charcot’s foot: newest findings on its pathophysiology, diagnosis, and treatment. Diabetes Metab. 2010;36(4):251-255. doi:10.1016/j.diabet.2010.04.002 PubMed | Crossref | Google Scholar
- Wukich DK, Sung W. Charcot arthropathy of the foot and ankle: modern concepts and management review. J Diabetes Complications. 2009;23(6):409-426. doi:10.1016/j.jdiacomp.2008.09.004 PubMed | Crossref | Google Scholar
- Rogers LC, Frykberg RG, Armstrong DG, et al. The Charcot foot in diabetes. Diabetes Care. 2011;34(9):2123-2129. doi:10.2337/dc11-0844 PubMed | Crossref | Google Scholar
- Phillips A, Rice L. Identification of Charcot foot: A case report. Nephrol Nurs J. 2022;49(5):419-425. Identification of Charcot foot: A case report
- Burns PR, Monaco SJ. Revisional surgery of the diabetic Charcot foot and ankle. Clin Podiatr Med Surg. 2017;34(1):77-92. doi:10.1016/j.cpm.2016.07.009 PubMed | Crossref | Google Scholar
- Trepman E, Nihal A, Pinzur MS. Current topics review: Charcot neuroarthropathy of the foot and ankle. Foot Ankle Int. 2005;26(1):46-63. doi:10.1177/107110070502600109 PubMed | Crossref | Google Scholar
- Chantelau E. The perils of procrastination: effects of early vs. delayed detection and treatment of incipient Charcot fracture. Diabet Med. 2005;22(12):1707-1712. doi:10.1111/j.1464-5491.2005.01677.x PubMed | Crossref | Google Scholar
- Messenger G, Masoetsa R, Hussain I. A narrative review of the benefits and risks of total contact casts in the management of diabetic foot ulcers. J Am Coll Clin Wound Spec. 2018;9(1-3):19-23. doi:10.1016/j.jccw.2018.05.002 PubMed | Crossref | Google Scholar
- Park GY. Orthotic management for diabetic foot. J Korean Soc Prosthet Orthot. 2009;3:21-26. Orthotic management for diabetic foot
- Selby PL, Young MJ, Boulton AJ. Bisphosphonates: a new treatment for diabetic Charcot neuroarthropathy? Diabet Med. 1994;11(1):28-31. doi:10.1111/j.1464-5491.1994.tb00225.x PubMed | Crossref | Google Scholar
- Chang MC, Choo YJ, Park IS, Park MW, Kim DH. Orthotic approach to prevention and management of diabetic foot: A narrative review. World J Diabetes. 2022;13(11):912-920. doi:10.4239/wjd.v13.i11.912 PubMed | Crossref | Google Scholar
- Güven MF, Karabiber A, Kaynak G, Oğüt T. Conservative and surgical treatment of the chronic Charcot foot and ankle. Diabet Foot Ankle. 2013;4. doi:10.3402/dfa.v4i0.21177 PubMed | Crossref | Google Scholar
- Sohn MW, Stuck RM, Pinzur M, Lee TA, Budiman-Mak E. Lower-extremity amputation risk after Charcot arthropathy and diabetic foot ulcer. Diabetes Care. 2010;33(1):98-100. doi:10.2337/dc09-1497 PubMed | Crossref | Google Scholar
- Stone NC, Daniels TR. Midfoot and hindfoot arthrodeses in diabetic Charcot arthropathy. Can J Surg. 2000;43(6):449-455. Midfoot and hindfoot arthrodeses in diabetic Charcot arthropathy
- Efficacy of Teriparatide in Diabetic Inactive Charcot Neuroarthropathy of Foot. ClinicalTrials.gov Identifier: NCT02023411. 2013. Efficacy of Teriparatide in Diabetic Inactive Charcot Neuroarthropathy of Foot
- Genetic Contribution to the Pathophysiology of the Charcot Foot in Qatari Patients with Diabetes. ClinicalTrials.gov Identifier: NCT02316483. 2015. Genetic Contribution to the Pathophysiology of the Charcot Foot in Qatari Patients with Diabetes
- Schweitzer M, Rockhill S. Conservative management of Charcot neuroarthropathy. Clin Podiatr Med Surg. 2022;39(4):585-594. doi:10.1016/j.cpm.2022.05.005 PubMed | Crossref | Google Scholar
- Waibel FWA, Weber S, Selman F, et al. No difference in risk of amputation or frequency of surgical interventions between patients with diabetic and nondiabetic Charcot arthropathy. Clin Orthop Relat Res. 2023;481(8):1560-1568. doi:10.1097/CORR.0000000000002546 PubMed | Crossref | Google Scholar
- Berli MC, Azaiez N, Götschi T, et al. Muscle atrophy in diabetic patients with Charcot foot: a case-control study. Skeletal Radiol. 2023;52(9):1661-1668. doi:10.1007/s00256-023-04328-1 PubMed | Crossref | Google Scholar
- Kavarthapu V, Giddie J, Kommalapati V, Casey J, Bates M, Vas P. Evaluation of adjuvant antibiotic-loaded injectable bio-composite material in diabetic foot osteomyelitis and Charcot foot reconstruction. J Clin Med. 2023;12(9):3239 doi:10.3390/jcm12093239 PubMed | Crossref | Google Scholar
Acknowledgments
Not applicable
Funding
Not applicable
Author Information
Corresponding Author:
Raziya Begum Sheikh
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Email: raziya.pharma@gmail.com
Co-Authors:
Rupashi Mukhia
Department of Outcomes Research
Anesthesiology Institute, Cleveland Clinic, USA
Authors Contributions
All patient-related data were collected by Dr. Rupashi Mukhia. Raziya Begum Sheikh contributed to the writing of the manuscript, including the original draft preparation and subsequent review and editing to refine the final version.
Informed Consent
Not applicable
Conflict of Interest Statement
This case report is based on a clinical case published in the “Cases” section of medtigo.com. The authors declare no conflicts of interest related to this publication.
Guarantor
Not applicable
DOI
Cite this Article
Begum RS, Rupashi M. Diabetic Charcot’s Arthropathy of the Foot. medtigo J Med. 2023;1(4):e3062142. doi:10.63096/medtigo3062142 Crossref









