Author Affiliations
Abstract
Delirium is a frequent and serious complication in hospitalized and critically ill patients, especially among older adults, leading to higher morbidity, mortality, and healthcare costs.
This systematic review included five recent studies assessing pharmacologic and non-pharmacologic interventions for delirium prevention, monitoring, and management. Eligible studies involved adult or elderly hospitalized or intensive care unit (ICU) patients, with outcomes on delirium incidence, severity, or duration.
The dynamic delirium (DyDel) program, a structured nurse- and family-led intervention, significantly reduced delirium incidence (5.6% vs 14.8%, p=0.0492) and duration. Suvorexant showed no significant overall reduction but decreased hyperactive/mixed subtypes (p=0.04). Delirium trajectory analyses revealed distinct clinical outcomes, with some patterns predicting higher mortality. Consistent pain, agitation, and delirium (PAD) monitoring improved clinical outcomes in multiple studies.
Evidence supports structured monitoring, early risk identification, and multidisciplinary, patient-centered interventions as effective strategies to reduce delirium burden. Combining pharmacologic and non-pharmacologic approaches, especially through standardized monitoring and individualized care, can improve delirium outcomes in high-risk hospitalized populations.
Keywords
Delirium management, Frailty, Postoperative delirium, Intensive care unit, Interventions, Delirium monitoring compliance.
Introduction
Delirium is an acute disturbance in attention and awareness, affecting up to 75% of intensive care unit (ICU) patients and a substantial proportion of hospitalized older adults. It is associated with poor clinical outcomes, including prolonged hospital stays, cognitive decline, institutionalization, and increased mortality, creating a significant healthcare and economic burden. The condition’s complex and varied causes make it hard to diagnose and treat, and existing trials have been largely ineffective.[1-6]
Delirium affects 29-64% of hospitalized older adults and is linked to worse outcomes. Although pain is common in hospitalized older adults (38-84%), its role in delirium is less understood. Pain and delirium share complex, bidirectional links involving depression and sleep disruption. Pain may trigger delirium via acute cerebral stress, catecholamine release, and inflammatory responses.[7-14]
The condition is multifactorial, involving predisposing factors such as advanced age, dementia, and severe illness, and precipitating factors including infection, surgery, and medication effects. Accurate and timely identification is challenging, and effective prevention and management require both pharmacologic and non-pharmacologic strategies.[15-20]
Delirium is marked by sudden disturbances in attention and awareness, requiring tailored nursing care to prevent adverse outcomes like falls or injuries. Despite evidence-based guidelines, delirious patients often receive inadequate or inappropriate care, such as restraints or poor hydration, which can worsen their condition.[21-25]
This systematic review examines recent evidence on interventions aimed at preventing, monitoring, and managing delirium in hospitalized and critically ill patients, with a focus on structured monitoring protocols, pharmacologic treatments, and individualized care approaches.
Methodology
A systematic search was conducted in PubMed for studies published between January 1, 2020, and December 31, 2025. The search focused on the prevention, monitoring, and management of delirium, with emphasis on randomized controlled trials (RCTs), clinical trials, and observational studies involving adult and elderly hospitalized or ICU patients.
The initial search yielded 1,842 records. Prior to screening, 1,430 records were removed for various reasons, including duplicates and ineligibility based on title or preliminary filters. The remaining 412 records were subjected to title and abstract screening, of which 150 were excluded for not meeting the inclusion criteria. A total of 262 reports were sought for full-text retrieval, while 204 could not be retrieved due to access issues or unavailability.
A total of 58 full-text articles were assessed for eligibility. Of these, 53 articles were excluded for reasons including insufficient outcome data, unclear delirium definitions or diagnostic tools, or poor methodological quality. Ultimately, five studies met all inclusion criteria and were included in the final synthesis.
Inclusion criteria:
- Studies involving adult or elderly hospitalized patients
- Studies that focused on prevention, monitoring, or management of delirium
- Reports with clear outcome measures related to delirium incidence, severity, duration, or clinical outcomes.
Exclusion criteria:
- Books, commentaries, editorials, letters, documents, and book chapters
- Articles published in languages other than English
- Animal studies and in vitro (laboratory) studies
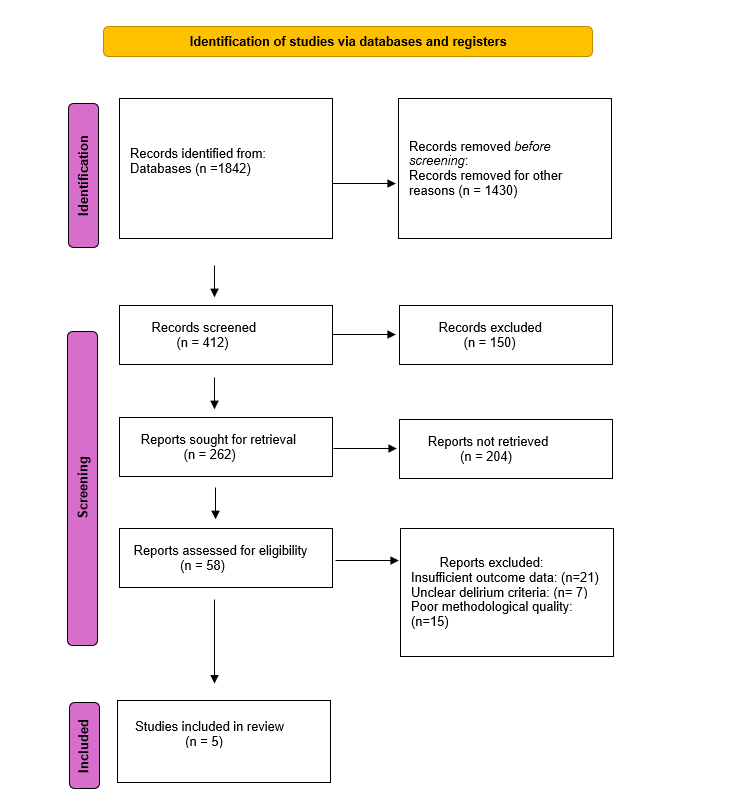
Figure 1. PRISMA flow diagram
Results
Effectiveness of intervention on delirium incidence and clinical outcomes in ICU patients
In the study by Gómez Tovar, LO. (NCT05172583), A double-masked, phase III randomized controlled trial was conducted in the ICU of a Colombian university hospital. Adults (>18 years), admitted within 24 hours, delirium-free, and accompanied by a family member were eligible. Patients with Richmond Agitation-Sedation Scale (RASS) scores of +4 or -5, or with cognitive/neurological disorders, were excluded. Using Hahai and Khurshid’s formula (α=0.05, β=0.20), assuming 30% delirium in controls vs. 12% in intervention and accounting for 10% attrition, 213 patients were enrolled (71 intervention, 142 control) with a 1:2 allocation via concealed block randomization.
The intervention group received DyDel, a non-pharmacological delirium prevention program incorporating the ABCDEF bundle, ICU humanization guidelines, and patient preferences (e.g., magazines, games, audio-visual tools). Interventions were RASS-adjusted and delivered face-to-face by a nurse and family member during every ICU shift. Components were mandatory or optional per patient preference.
The control group received standard ICU care: orientation, scheduled family visits, daily physiotherapy, and dexmedetomidine sedation. Primary outcomes were delirium incidence and duration (CAM-ICU). Secondary outcomes included ICU stay length, mechanical ventilation days, restraint use, and pain scores. A blinded nurse assessed outcomes each shift. Intention-to-treat analysis was performed with p ≤ 0.05 using SPSS v28.
Data were collected from August 2021 to February 2022. Of 213 ICU patients recruited within 24 hours of admission, 71 were assigned to the intervention group and 142 to the control group. Patients who died or remained in the ICU for less than 24 hours were excluded. Baseline characteristics were similar between groups: mean age was 60.3 ± 15.2 years, 59.6% were male, and cardiovascular disease was the leading diagnosis (74.6%). High blood pressure (51.2%) and diabetes (27.7%) were common comorbidities.
There were no significant differences in pharmacologic treatments or device use; fentanyl and morphine were commonly administered, and urinary and central venous catheters were frequently used.
Delirium incidence was significantly lower in the intervention group (5.6%) compared to the control group (14.8%) (p=0.0492), with all intervention cases being hypoactive. Delirium days were fewer in the intervention group (0.07 ± 0.31 vs. 0.34 ± 1.28; p=0.016). Relative risk was 0.38 (p=0.0455), with an absolute risk reduction of 9.1% and a number needed to treat (NNT) of 11.
Secondary outcomes favoured the intervention group, with significantly more days of low pain intensity (Visual Analogue Scale (VAS) <4: p=0.002; Campbell <3: p=0.015), longer time to delirium onset (p=0.038), and shorter ICU stay risk (Hazard ratios: HR=2.90, p=0.0255).[26]
Clinical outcomes of suvorexant for delirium prevention in older adults
In a double-blind, placebo-controlled phase 3 randomized trial by Hatta K et al., conducted at 50 hospitals in Japan (Oct 2020-Dec 2022), patients aged 65-90 hospitalized for acute illness or elective surgery under general anaesthesia were enrolled if at high delirium risk due to mild cognitive impairment, prior delirium, or both. Exclusion criteria included current delirium (DSM-5) or (Delirium Rating Scale–Revised–98: DRS-R-98 ≥14.5), use of psychotropic drugs, or neurological conditions such as epilepsy, Parkinson’s disease, or moderate/severe dementia.
Eligible participants were randomized 1:1 to receive either nightly suvorexant 15 mg or placebo for 5-7 days, stratified by age (<75 or ≥75) and hospitalization type. Screening occurred within 14 days before surgery or 24 hours post-admission. Daily delirium assessments (DSM-5, DRS-R-98) were conducted by trained raters. Trial medication was stopped upon delirium onset. Hypnotics and psychotropics were prohibited during assessment, and nonpharmacologic prevention strategies were applied as needed.
The primary outcome was delirium incidence (DSM-5). Secondary outcomes included severity and incidence based on DRS-R-98. Exploratory analysis addressed hyperactive and mixed subtypes, while sleep quality was assessed via the Richards-Campbell Sleep Questionnaire. A modified intent-to-treat analysis included all treated patients with ≥1 DSM-5 evaluation. Miettinen-Nurminen methods, Cox regression, and Kaplan-Meier analysis (time to delirium) were used. Safety was monitored through adverse events, labs, and vital signs.
Of 207 randomized participants, 203 received treatment: 101 in the suvorexant group and 102 in the placebo group. The mean age was 81.5 years (suvorexant) and 82.0 years (placebo). Treatment discontinuation occurred in 22.8% (suvorexant) and 30.4% (placebo), mainly due to delirium. Baseline characteristics were similar: 91.1% were aged ≥75, 93.1% had mild cognitive impairment or dementia, and 18.7% had prior delirium. Hospitalization was for elective surgery (62.6%) or acute illness (37.4%). Mean treatment duration was 5.8 days (suvorexant) and 5.5 days (placebo).
Delirium incidence (DSM-5) was lower with suvorexant (16.8%) versus placebo (26.5%), though not statistically significant (difference -8.7%, 95% confidence interval: CI -20.1% to 2.6%, p=.13). Time-to-event rates were 2.5 vs 4.1 per 100 person-days (HR 0.62, 95% CI 0.34-1.15, p=.13). A post hoc analysis showed a significant reduction in hyperactive/mixed subtypes with suvorexant (10.9% vs 21.6%; difference -10.7%, 95% CI-21.2% to -0.6%, p=.04). Hypoactive delirium rates were similar but varied by hospitalization type. DRS-R-98 secondary outcomes favoured suvorexant, though not significantly.
Safety outcomes were comparable. One death (urosepsis) in the suvorexant group was deemed unrelated. Adverse events were similar, with slightly more falls (6.9% vs 2.9%) and hallucinations (3.0% vs 1.0%) in the suvorexant group.[27]
Distinct delirium trajectories and their association with clinical outcomes
In a secondary analysis by Lindroth H et al., deidentified prospective data from two RCTs (Pharmacologic Management of Delirium (PMD) and de-PMD, NCT00842608) involving ICU patients were used. The dataset included 551 adult patients (≥18 years) from three Indianapolis hospitals diagnosed with delirium using CAM-ICU. After excluding 20 patients without post-randomization delirium severity data, 531 patients remained, contributing 4,438 CAM-ICU-7 assessments over 7 days. Patients were excluded for language barriers, sensory issues, substance intoxication, severe mental illness, or pregnancy.
Delirium severity was measured daily with CAM-ICU-7 (score range 0-7), with coma (RASS -4 or -5) assigned a score of 7. After July 1, 2010, assessments were conducted twice daily. Demographics, comorbidities (Charlson Comorbidity Index), illness severity of Acute Physiology and Chronic Health Evaluation II (APACHE II), and functional/cognitive baseline were collected via electronic health records (EHRs) or caregiver reports. Daily adverse events and interventions were also recorded.
Group-based trajectory modelling (Proc Traj, SAS v9.4) was used to model delirium severity patterns, assuming a zero-inflated Poisson distribution. Model selection relied on Bayesian information criteria, posterior probability, and classification odds. Sensitivity analyses treated attrition due to death (n=20) or discharge (n=134) as missing at random (MAR). Least Absolute Shrinkage and Selection Operation (LASSO) regression and multinomial modelling, with Bonferroni correction, were used to predict outcomes. About 34% of assessments were missing, largely due to discharge or clinical care, and were addressed in the model assumptions.
A total of 531 participants with delirium were included, with a mean age of 60 years, 55% female, and 73% on mechanical ventilation. Five distinct delirium trajectories were identified: mild-brief (17%), severe-rapid recovers (16%), mild-accelerating (7%), severe-slow recovers (22%), and severe-nonrecovers (40%). Differences in baseline cognition, Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE), Instrumental Activities of Daily Living (iADL), race (African American), and ventilation status significantly varied across trajectories. African Americans were nearly three times more likely to follow the severe-nonrecovers trajectory (odds ratio: OR 2.8, 95% CI 1.6-5.0).
Clinical indicators such as bladder catheter use, restraint days, mechanical ventilation days, and unsafe behaviour were significantly more frequent in the severe-nonrecovers group compared to the mild-brief group. Mean CAM-ICU-7 scores ranged from 0.5 (mild-brief) to 5.9 (severe-nonrecovering), with significant variation in RASS scores and symptom fluctuations.
Hospital outcomes varied: mild-brief had the lowest mortality (30-day: 3%), while mild-accelerating had the highest (30-day: 31%), followed by severe-nonrecovers (17%). Length of stay was longest in severe-slow recovers and severe-nonrecovers. Discharge to home was highest in mild-brief (62%) and lowest in mild-accelerating (20%).
Logistic regression showed that trajectory membership, particularly mild-accelerating (OR 9.39), significantly predicted 30-day mortality. LASSO modelling confirmed that trajectory, age, comorbidities, illness severity, and functional status were significant mortality predictors (receiver operating characteristic: ROC=0.78). Sensitivity analyses supported the robustness of the five-trajectory model, with consistent membership distribution and trajectory-mortality associations.[28]
Clinical correlates and prognostic implications of delirium course trajectories in critical care
Deffland M et al. conducted a retrospective cohort study at Charité-Universitätsmedizin Berlin, approved by the local ethics committee (EA 2/092/14), with informed consent waived. Data were extracted (June 2017-August 2018) for ICU admissions from January to December 2016. Adult ICU patients invoiced by Diagnosis-Related Group (DRG) and not enrolled in PAD-related trials were included. Exclusions were non-adults, multiple ICU ward admissions, deceased patients, and cases lacking intermediate ICU Day documentation or feasible CAM-ICU monitoring. Data were anonymized during export.
Clinical and economic outcomes were derived from COPRA and SAP systems. Primary outcomes included ICU and hospital length of stay (LoS), duration of mechanical ventilation, case mix per day, and profit per day. The key predictor was adherence to PAD monitoring-sedation (RASS ≥ -3), delirium (CAM-ICU), and pain (VAS/BPS based on sedation/delirium status). Adherence was defined as the percentage of ICU shifts monitored, excluding admission/discharge days; 100% adherence indicated “achieved.”
Covariates included age and APACHE II score (≤10, 11-20, >20). Analyses combined classical statistics (SPSS v24) and machine learning (RStudio v1.1.419). The Boruta algorithm identified relevant predictors, and the Maximal Information Coefficient (MIC) assessed association strength. This dual approach enhanced predictor detection and minimized overfitting, improving generalizability.
A total of 1323 patients were included for pain and agitation/sedation monitoring, and 1266 for delirium monitoring, with discrepancies due to inadequate RASS data. The median age was 68 years; 84.4% underwent surgery, and 60% were male. Median ICU and hospital LoS were 4 and 12 days, respectively; median mechanical ventilation lasted 32 hours. Monitoring adherence medians were 91.7% for pain, 90.4% for agitation/sedation, and 100% for delirium. Median economic values were 0.44 case mix points/day and €11.05 profit/day. Median APACHE II score was 14.
Two-group analyses revealed that adherence to pain, sedation, and delirium monitoring was associated with significantly improved clinical outcomes (shorter hospital/ICU LoS and ventilation duration) across all APACHE groups. However, economic outcomes were generally worse with monitoring adherence. Exceptions were found in specific APACHE strata but were not statistically significant.
Regression analysis showed that higher APACHE II scores were consistently linked to increased hospital/ICU LoS and ventilation duration. Pain monitoring adherence reduced ICU LoS, and age was inversely related to ventilation duration. Delirium monitoring adherence decreased the case mix per day.
Machine learning found pain monitoring as the strongest predictor of clinical outcomes, while APACHE II best predicted profit per day. Associations between PAD monitoring and ICU LoS were strongest, while other associations were weaker (MIC < 0.2).[29]
Clinical and economic effects of pain, agitation, and delirium monitoring compliance
In Kanno M. study, this single-center prospective observational study included patients aged ≥65 years undergoing urological surgery under general or spinal anaesthesia at Yokohama City University Medical Center (April–September 2019). Exclusion criteria were ICU admission, impaired consciousness (Japan Coma Scale 100-300), language or cognitive limitations, preoperative delirium, or missing data.
Delirium and subsyndromal delirium (SSD) were assessed using the Japanese version of the CAM, validated for nurse use. Assessments occurred once preoperatively and thrice daily for three postoperative days. Nurses received standardized training based on the Short CAM Training Manual to ensure consistent evaluations.
POD was defined by CAM criteria: acute onset, inattention, and either disorganized thinking or altered consciousness. SSD was defined by one or more CAM criteria without a full POD diagnosis. The primary outcome was the combined incidence of POD and SSD.
Independent variables included age, sex, activities of daily living (ADL), comorbidity (Charlson Comorbidity Index), dementia, anaesthesia type, surgical details, lab values, and use of physical restraints or bed sensors. The Comprehensive Geriatric Assessment-7 (CGA7) was the key variable.
Binary logistic regression was used to identify predictors of POD/SSD, with variables showing p<0.2 in univariate analysis included in multivariate models. Analyses were conducted using SPSS v26 with significance set at p<0.05.
We included 1323 patients for pain and agitation/sedation monitoring and 1266 for delirium monitoring. The difference resulted from patients with insufficient RASS data, particularly those with a RASS of -3 or lower, who could not be assessed for delirium. The median patient age was 68 years; 60% were male, and 84.4% underwent surgery. Median ICU and hospital LoS were 4 and 12 days, respectively, with a median of 32 hours of mechanical ventilation. Median adherence to monitoring was 91.7% for pain, 90.4% for agitation/sedation, and 100% for delirium. Median economic outcomes were 0.44 case mix points per day and €11.05 profit per day. The median APACHE II score was 14.
Two-group analysis showed that adherence to PAD monitoring was associated with shorter ICU/hospital LoS and ventilation duration across all APACHE strata. However, economic outcomes were generally worse with monitoring adherence. Exceptions lacked statistical significance. Regression models showed that higher APACHE II scores predicted worse clinical outcomes. Adherence to pain monitoring was linked to reduced ICU LoS, while age was inversely related to ventilation duration. Delirium monitoring was associated with a lower case mix/day. Machine learning identified pain monitoring adherence as the strongest predictor of clinical outcomes and APACHE II as the best predictor of economic outcomes.[30]
| Study | Design & Population | Groups / Interventions | Primary Outcomes | Key Findings |
| Gómez Tovar LO | RCT, double-masked, phase III (N=213; ICU patients >18 yrs; Colombia) | DyDel program vs. standard ICU care (incl. dexmedetomidine) | Delirium incidence and duration (CAM-ICU) | Decreased delirium: 5.6% vs 14.8%, p=0.0492; decreased delirium days: 0.07 vs 0.34, p=0.016; NNT=11; lower ICU LoS, lower pain, delayed onset |
| Hatta K et al. | RCT, DBPC, phase III (N=203; older adults 65–90 yrs; Japan) | Suvorexant 15 mg vs. placebo for 5–7 nights | Delirium incidence (DSM-5) | Decreased delirium: 16.8% vs 26.5%, p=0.13 (NS); lower hyperactive/mixed: 10.9% vs 21.6%, p=0.04; Similar adverse events |
| Lindroth H et al. | Secondary analysis of 2 RCTs (N=531 ICU patients with delirium; US) | 5 delirium severity trajectories (CAM-ICU-7 based) | Delirium trajectory patterns and outcomes | Severe-nonrecovers: increased severity, increased LoS, increased mortality (17%); Mild-brief: decreased mortality (3%), increased discharge home (62%) |
| Deffland M et al. | Retrospective cohort (N=1323 PAD-monitored ICU patients; Germany) | Adherence to PAD monitoring vs. non-adherence | ICU/hospital LoS, ventilation, economic outcomes | PAD adherence decreased LoS, ventilation; Pain monitoring decreased ICU LoS; Delirium monitoring decreased case mix/day; APACHE II increased poor outcomes |
| Kanno M et al. | Prospective observational (N=1323; elderly ≥65 yrs postop urological surgery; Japan) | PAD monitoring adherence | POD + SSD incidence, LoS, ventilation | Monitoring lower LoS, ventilation; Pain monitoring is the strongest clinical predictor; Delirium monitoring has a lower case mix/day |
Table 1. Effectiveness of interventions on delirium in ICU and hospitalized patients
Discussion
The multifaceted nature of delirium, especially in older adults and ICU patients, remains a pressing clinical challenge. This compilation of studies reinforces the need for robust assessment tools and targeted interventions to mitigate its incidence and consequences. The Gómez Tovar study demonstrated that a nurse- and family-led, non-pharmacological ICU intervention significantly reduced delirium incidence (5.6% vs 14.8%, p=0.0492), duration, and pain levels, highlighting the value of personalized, human-centered care. Meanwhile, Hatta et al.’s trial of suvorexant in high-risk older adults showed a non-significant but clinically relevant reduction in delirium incidence, with post hoc analysis indicating a significant effect on hyperactive/mixed subtypes (p=0.04).
Trajectory analyses by Lindroth and Deffland further clarified delirium’s variable progression. Lindroth identified five distinct delirium courses, with the “mild-accelerating” group showing the highest mortality (31%), while “mild-brief” had the lowest (3%). African American patients and those on ventilation were disproportionately represented in worse trajectories, emphasizing disparities and the need for tailored interventions. Deffland’s analysis supported the link between consistent pain, sedation, and delirium (PAD) monitoring and better clinical outcomes, though economic benefits were less clear.
Finally, Kanno’s observational study confirmed that PAD monitoring adherence correlated with shorter ICU/hospital stays, supporting the clinical utility of structured assessments. Despite varying methodologies and settings, all studies stress the need for early risk identification, standard monitoring, and personalized prevention strategies to reduce delirium burden and improve outcomes across diverse patient populations.
Conclusion
Delirium remains a common and serious complication in hospitalized and critically ill patients. The reviewed studies demonstrate that structured, multidisciplinary approaches such as the DyDel program, targeted pharmacologic therapy, and consistent PAD monitoring can meaningfully reduce delirium incidence, duration, and related complications. Early identification, standardized assessment, and patient-centered prevention strategies are essential to improving outcomes and reducing the healthcare burden associated with delirium.
References
- Ormseth CH, LaHue SC, Oldham MA, Josephson SA, Whitaker E, Douglas VC. Predisposing and Precipitating Factors Associated With Delirium: A Systematic Review. JAMA Netw Open. 2023;6(1):e2249950. doi:10.1001/jamanetworkopen.2022.49950
PubMed | Crossref| Google Scholar - Gracie TJ, Caufield-Noll C, Wang NY, Sieber FE. The Association of Preoperative Frailty and Postoperative Delirium: A Meta-analysis. Anesth Analg. 2021;133(2):314-323. doi:10.1213/ANE.0000000000005609
PubMed | Crossref | Google Scholar - Zietlow KE, Wong S, Heflin MT, et al. Geriatric Preoperative Optimization: A Review. Am J Med. 2022;135(1):39-48. doi:10.1016/j.amjmed.2021.07.028 PubMed | Crossref | Google Scholar
- Noah AM, Almghairbi D, Evley R, Moppett IK. Preoperative inflammatory mediators and postoperative delirium: systematic review and meta-analysis. Br J Anaesth. 2021;127(3):424-434. doi:10.1016/j.bja.2021.04.033
PubMed | Crossref | Google Scholar - Komici K, Fantini C, Santulli G, et al. The role of diabetes mellitus on delirium onset: a systematic review and meta-analysis. Cardiovasc Diabetol. 2025;24(1):216. doi:10.1186/s12933-025-02782-w
PubMed | Crossref | Google Scholar - Ghezzi ES, Greaves D, Boord MS, et al. How do predisposing factors differ between delirium motor subtypes? A systematic review and meta-analysis. Age Ageing. 2022;51(9):afac200. doi:10.1093/ageing/afac200 PubMed | Crossref | Google Scholar
- Kalvas LB, Harrison TM. State of the science in pediatric ICU delirium: An integrative review. Res Nurs Health. 2020;43(4):341-355. doi:10.1002/nur.22054
PubMed | Crossref | Google Scholar - Eost-Telling C, McNally L, Yang Y, et al. The association between delirium and falls in older adults in the community: a systematic review and meta-analysis. Age Ageing. 2024;53(12):afae270. doi:10.1093/ageing/afae270 PubMed | Crossref | Google Scholar
- Sist L, Pezzolati M, Ugenti NV, et al. Prioritization Patterns of Nurses in the Management of a Patient With Delirium: Results of a Q-Methodology Study. Res Nurs Health. 2025;48(2):257-270. doi:10.1002/nur.22449 PubMed | Crossref | Google Scholar
- White N, Bazo-Alvarez JC, Koopmans M, West E, Sampson EL. Understanding the association between pain and delirium in older hospital inpatients: systematic review and meta-analysis. Age Ageing. 2024;53(4):afae073. doi:10.1093/ageing/afae073
PubMed | Crossref | Google Scholar - Earwaker M, Anderson N, Egli V. Developing nursing interventions in Paediatric Emergence Delirium: a scoping review. Contemp Nurse. 2024;60(1):82-95. doi:10.1080/10376178.2024.2319852
PubMed | Crossref | Google Scholar - Dong B, Yu D, Jiang L, Liu M, Li J. Incidence and risk factors for postoperative delirium after head and neck cancer surgery: an updated meta-analysis. BMC Neurol. 2023;23(1):371. doi:10.1186/s12883-023-03418-w
PubMed | Crossref | Google Scholar - Richardson SJ, Cropp AD, Ellis SW, Gibbon J, Sayer AA, Witham MD. The interrelationship between multiple long-term conditions (MLTC) and delirium: a scoping review. Age Ageing. 2024;53(7):afae120. doi:10.1093/ageing/afae120
PubMed | Crossref | Google Scholar - Liu J, Xu S, Wang J, et al. Prevalence of cognitive frailty among older adults in China: a systematic review and meta-analysis. BMJ Open. 2023;13(4):e066630. doi:10.1136/bmjopen-2022-066630
PubMed | Crossref | Google Scholar - Zazzara MB, Ornago AM, Cocchi C, Serafini E, Bellelli G, Onder G. A pandemic of delirium: an updated systematic review and meta-analysis of occurrence of delirium in older adults with COVID-19. Eur Geriatr Med. 2024;15(2):397-406. doi:10.1007/s41999-023-00906-7
PubMed | Crossref | Google Scholar - Skains RM, Hayes JM, Selman K, et al. Emergency Department Programs to Support Medication Safety in Older Adults: A Systematic Review and Meta-Analysis. JAMA Netw Open. 2025;8(3):e250814. doi:10.1001/jamanetworkopen.2025.0814
PubMed | Crossref | Google Scholar - Mukminin MA, Yeh TH, Lin HC, Rohmah I, Chiu HY. Global prevalence and risk factors of delirium among patients following acute stroke: A systematic review and meta-analysis. J Stroke Cerebrovasc Dis. 2025;34(2):108221. doi:10.1016/j.jstrokecerebrovasdis.2024.108221
PubMed | Crossref | Google Scholar - Andreas M, Piechotta V, Skoetz N, et al. Interventions for palliative symptom control in COVID-19 patients. Cochrane Database Syst Rev. 2021;8(8):CD015061. doi:10.1002/14651858.CD015061
PubMed | Crossref | Google Scholar - Zhou B, Yu DD, Xu X, Wang J, Li J. Association of preoperative neutrophil-to-lymphocyte ratio with the risk of postoperative delirium in elderly patients undergoing noncardiac surgery: a systematic review and meta-analysis. Psychogeriatrics. 2024;24(4):993-1003. doi:10.1111/psyg.13138
PubMed | Crossref | Google Scholar - Houghton JSM, Nickinson ATO, Bridgwood B, et al. Prevalence of Cognitive Impairment in Individuals with Vascular Surgical Pathology: A Systematic Review and Meta-Analysis. Eur J Vasc Endovasc Surg. 2021;61(4):664-674. doi:10.1016/j.ejvs.2020.12.016
PubMed | Crossref | Google Scholar - Marcantonio ER. Delirium in Hospitalized Older Adults. N Engl J Med. 2017;377(15):1456-1466. doi:10.1056/NEJMcp1605501
PubMed | Crossref | Google Scholar - Oh-Park M, Chen P, Romel-Nichols V, Hreha K, Boukrina O, Barrett AM. Delirium Screening and Management in Inpatient Rehabilitation Facilities. Am J Phys Med Rehabil. 2018;97(10):754-762. doi:10.1097/PHM.0000000000000962
PubMed | Crossref | Google Scholar - Gonin P, Beysard N, Yersin B, Carron PN. Excited Delirium: A Systematic Review. Acad Emerg Med. 2018;25(5):552-565. doi:10.1111/acem.13330
PubMed | Crossref | Google Scholar - Ghezzi ES, Greaves D, Boord MS, et al. How do predisposing factors differ between delirium motor subtypes? A systematic review and meta-analysis. Age Ageing. 2022;51(9):afac200. doi:10.1093/ageing/afac200 PubMed | Crossref | Google Scholar
- Eost-Telling C, McNally L, Yang Y, et al. The association between delirium and falls in older adults in the community: a systematic review and meta-analysis. Age Ageing. 2024;53(12):afae270. doi:10.1093/ageing/afae270 PubMed | Crossref | Google Scholar
- Gómez Tovar LO, Henao Castaño AM. Dynamic delirium – Nursing intervention to reduce delirium in patients critically Ill, a randomized control trial. Intensive Crit Care Nurs. 2024;83:103691. doi:10.1016/j.iccn.2024.103691 PubMed | Crossref | Google Scholar
- Hatta K, Kishi Y, Wada K, et al. Suvorexant for Reduction of Delirium in Older Adults After Hospitalization: A Randomized Clinical Trial. JAMA Netw Open. 2024;7(8):e2427691. doi:10.1001/jamanetworkopen.2024.27691
PubMed | Crossref | Google Scholar - Lindroth H, Khan BA, Carpenter JS, et al. Delirium Severity Trajectories and Outcomes in ICU Patients. Defining a Dynamic Symptom Phenotype. Ann Am Thorac Soc. 2020;17(9):1094-1103. doi:10.1513/AnnalsATS.201910-764OC
PubMed | Crossref | Google Scholar - Deffland M, Spies C, Weiss B, et al. Effects of pain, sedation and delirium monitoring on clinical and economic outcome: A retrospective study. PLoS One. 2020;15(9):e0234801. doi:10.1371/journal.pone.0234801 PubMed | Crossref | Google Scholar
- Kanno M, Doi M, Kubota K, Kanoya Y. Risk factors for postoperative delirium and subsyndromal delirium in older patients in the surgical ward: A prospective observational study. PLoS One. 2021;16(8):e0255607. doi:10.1371/journal.pone.0255607
PubMed | Crossref | Google Scholar
Acknowledgments
Not applicable
Funding
Not applicable
Author Information
Corresponding Author:
Samatha Ampeti
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Email: ampetisamatha9@gmail.com
Co-Authors:
Shubham Ravindra Sali, Mansi Srivastava, Raziya Begum Sheikh, Sonam Shashikala B V, Patel Nirali Kirankumar
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Authors Contributions
All authors contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles. They were collectively involved in the writing, original draft preparation, and writing – review & editing to refine the manuscript. Additionally, all authors participated in the supervision of the work, ensuring accuracy and completeness. The final manuscript was approved by all named authors for submission to the journal.
Ethical Approval
Not applicable
Conflict of Interest Statement
None
Guarantor
None
DOI
Cite this Article
Shubham RS, Samatha A, Mansi S, Raziya BS, Sonam SBV, Patel NK. Delirium in Hospitalized and ICU Patients: Systematic Review of Pharmacologic and Non-Pharmacologic Interventions. medtigo J Emerg Med. 2025;2(3):e3092238. doi:10.63096/medtigo3092238 Crossref









