Author Affiliations
Abstract
Hailey–Hailey disease (HHD) is an infrequent blistering dermatosis initially documented in 1939 by the siblings Howard and Hugh Hailey, boasting an estimated incidence of 1 in 50,000. The mode of inheritance follows an autosomal dominant pattern with complete penetrance, though there exists variable expressivity among affected family members. Clinically, it typically manifests with flaccid vesicles and blisters on erythematous skin, leading to erosion, fissures, and vegetations. Commonly, maceration and superinfections ensue. Symmetric lesions tend to emerge in intertriginous regions, including retroauricular folds, lateral aspects of the neck, axillae, umbilicus, inguinal, and perianal areas. Assessing the efficacy and safety of treatments for HHD poses a challenge due to the absence of evidence-based guidelines and extensive clinical trials. Different treatment modalities are available for HHD, such as topical, systemic agents, and procedural therapies. This review described classical and emerging treatment options for HHD.
Keywords
Hailey–Hailey disease, ATP2C1 gene, Lesions, Oxidative stress, Remission, Treatment modalities.
Introduction
The Hailey brothers, dermatologists, Hugh Edward and William Howard, first described HHD, also known as familial benign chronic pemphigus or familial benign pemphigus, in 1939. It is a rare genetic disorder and consists of blisters and erosions that typically affect the neck, armpits, skin folds, and genitalia [1]. It is an ATP2C1 gene mutation-related autosomal dominant genodermatosis that affects keratinocyte adhesion by disrupting healthy skin. But as an autoimmune disease, pemphigus develops when the body’s immune system unintentionally targets healthy tissue [2,3]. The lack of targeted therapies and well-controlled clinical trials makes management of HHD challenging, even with advances in our understanding of its molecular basis. Furthermore, dermatologists find it difficult to manage HHD because of its persistent and recalcitrant nature, which significantly lowers a patient’s quality of life. This review focused on epidemiology, signs and symptoms, causes/pathogenesis, diagnosis and differential diagnosis, and treatment or management of HHD.
Epidemiology
HHD affects both males and females equally, with an estimated prevalence of 1 in 50,000 individuals in the general population [4]. Determining the actual prevalence is challenging, as HHD often goes misdiagnosed or undiagnosed. Although HHD typically manifests around puberty, some cases may not exhibit symptoms until the third or fourth decade of life [5]. The expression of HHD in affected family members varies in age and follows an autosomal dominant inheritance pattern with complete penetrance [6]. Familial benign pemphigus commonly appears in late adolescence or early adulthood, particularly in the 30s and 40s. Triggers and aggravating factors include menstruation, pregnancy, patch testing, skin infections, scabies, heat, and sweating. Sun exposure can either worsen or improve lesions, depending on the individual [7]. Additionally, certain drugs, such as efalizumab and nonsteroidal anti-inflammatory drugs (NSAIDs), have been linked to the onset of HHD [8].
Signs and Symptoms
Variability in the signs and severity of Hailey-Hailey disease is observed even among family members, with most cases having a family history of the disorder [9]. The primary presentation of HHD usually involves an erosive, blistering skin rash that predominantly affects the groin, neck, armpits, and chest [10], which is represented in Figure 1 [11]. Lesions in Hailey-Hailey disease may develop a yellow, crusty layer, and individuals may experience a sensation of burning or itching. The separation of lesions can lead to severe, cracked skin, and unpleasant odors may arise from secondary skin infections. HHD is characterized by relapsing and remitting skin lesions, meaning they resolve spontaneously but may occasionally
reappear [12]. The duration of an outbreak and the interval between the clearance and recurrence of lesions vary. Typically, lesions heal without leaving scars, but sun exposure, heat, friction, and injuries can exacerbate them [13]. Additionally, many patients exhibit asymptomatic longitudinal white bands in their fingernails. Skin lesions in HHD also carry a potential risk of giving rise to squamous cell carcinomas [14,15].
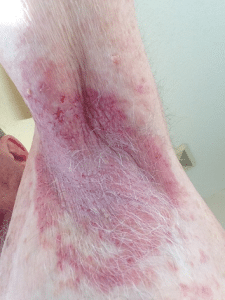
Figure 1: Mild, uninfected left axilla lesion in Hailey-Hailey
Pathogenesis
HHD stems from a mutation in the ATP2C1 gene located on the 3q21-q24 long region of chromosome 3. This gene encodes the Golgi apparatus’s human secretory pathway Ca2+/Mn2+-ATPase 1 proteins (hSPA1C). The hSPA1C protein acts as a calcium and magnesium transporter, leading to the deposition of calcium in the Golgi lumen. A malfunctioning calcium pump disrupts cell-to-cell adhesion, causing the affected cells to separate and resulting in skin damage known as acantholysis [16,17]. Decreased cellular Adenosine triphosphate (ATP) levels in turn impair actin organization and cell-to-cell adhesion [18]. Oxidative stress and certain messenger ribonucleic acids (mRNAs) may contribute to the pathophysiology of HHD. Oxidative stress increases the expression of miR-125b, while the keratinocytes of affected patients show
down-regulated levels of the proteins notch 1 and p63, which play roles in signal pathways leading to keratinocyte differentiation [19]. HHD follows an autosomal dominant inheritance pattern, indicating that a single mutated copy of the gene can cause the disorder. In cases of dominant genetic disorders, the altered gene can be inherited from either parent. Each pregnancy involving an affected parent carries a 50% chance that the child will inherit the mutated gene, and both men and women are equally at risk [19,20].
Several factors such as genetic inheritance, de novo mutations, genetic variability, and environmental factors may contribute to ATP2C1 gene mutations in HHD. If one parent carries the mutated gene, there is a 50% chance that each of their children will inherit the mutation and develop HHD. In some instances, individuals with HHD may experience a spontaneous mutation in the ATP2C1 gene not inherited from either parent. These de novo mutations can arise during the formation of reproductive cells or early in embryonic development. Different mutations in the ATP2C1 gene can lead to varying degrees of severity and clinical manifestations of HHD. The specific mutation and its location within the gene can impact how the protein functions, consequently influencing the disease’s severity. Specific environmental factors like heat, friction, and sweating can exacerbate skin lesions in individuals with HHD [3,6].
Differential Diagnosis
The diagnosis of HHD is recommended based on clinical morphology, lesion locations, family history, and histology displaying the characteristic appearance of the epidermis resembling a dilapidated brick wall [21]. The diagnostic process involves a comprehensive clinical assessment, a detailed patient history, identification of distinctive findings, and various specialized tests, including the surgical removal and microscopic examination (biopsy) of affected skin tissue. Biopsy results may reveal aberrant keratinization, the abnormal formation of keratin, and acantholysis, the breakdown of cell-to-cell adhesion. Blood tests do not detect antibodies in individuals with HHD, thereby eliminating the possibility of autoimmune diseases like pemphigus. Molecular genetic testing for variants in the ATP2C1 gene can confirm the diagnosis [16,20,21].
Direct immunofluorescence tests can help differentiate HHD from other conditions. Pemphigus vulgaris typically shows an intercellular pattern with IgG, while IgA pemphigus exhibits IgA. Paraneoplastic pemphigus may display granular deposits of IgG or IgM at the dermoepidermal junction, in addition to the distinctive intercellular pattern [22].
Immunological and autoimmune disorders, in particular other acantholytic dermatoses and intertriginous infections, are the primary differential diagnoses of HHD. Additional detail on the differential diagnosis is mentioned in Table 1.
| Diagnosis | Histology |
| HHD [14] | Generalized acantholysis
Clefts both intraepidermal and suprabasilar Dyskeratotic cells infrequently Absence of hyperkeratosis Follicle plugging absent Direct immunofluorescence in negative |
| Darier’s disease [21] | Less noticeable acantholysis
Diminutive suprabasilar clefts Clearly visible dyskeratotic cells Over keratosis Follicular blockage Direct immunofluorescence in negative |
| Erythrasma [23] | Gram-positive rods in the stratum corneum are stained with gram stain.
Mild perivascular infammatory infltrate Hyperkeratosis |
| Pemphigus vegetans [24] | Acantholysis suprabasilar
Acanthosis of the adnexal mucosa Blisters on the skin (intraepidermal) Direct immunofluorescence: IgG and C3 accumulation on the surface of epidermal keratinocytes |
| Inverse psoriasis [25] | Cutaneous hyperplasia
Rete ridge elongation and suprapapillary plate thinning Hypogranulosis Smooth parakeratosis |
| Galli–Galli disease [26] | Acantholysis suprabasilar, subtle
Dyskeratosis focal Extended rete ridges Increased pigmentation of basal keratinocytes Direct immunofluorescence in negative |
| Axillary granular parakeratosis [27] | Not acantholysis
The parakeratosis Stratum corneum that is thicker Granules of keratohyalin are retained Increased vascularity |
| Tinea cruris [28] | Hyphae or fungal spores within the stratum corneum stained with potassium hydroxide
Spongiosis that varies Inflammation in Dermis |
Table 1: Clinical differential diagnosis of HHD
Treatment for HHD
The treatment of HHD is tailored to address the specific symptoms manifested by each patient. The choice of treatment is influenced by factors such as the type and severity of the disease, the patient’s age, and overall health. Fortunately, in recent years, there have been developments in new therapeutic options for managing HHD.
General Precautions
Management of HHD involves reducing perspiration and minimizing friction, which can exacerbate the condition. Wearing light, loose clothing is recommended to mitigate these factors. It is advisable to avoid occlusive and adhesive dressings. Antiseptic cleansers, such as chlorhexidine and others, prove effective in treating and preventing secondary microbial infections associated with HHD [29].
Topical Therapy
Corticosteroids: The primary recommendation for first-line therapy in Hailey-Hailey disease is the use of topical corticosteroids (TCS). Early application of TCS can potentially prevent the development of lesions. A cross-sectional study by Burge et al. involving 58 patients showed a favorable response in 86% of cases with topical corticosteroids [30]. To minimize the risk of secondary infections, combinations with topical antimicrobial agents are often employed alongside TCS. If topical corticosteroids prove ineffective, intralesional steroids may be considered as an alternative option [31].
Calcineurin Inhibitors: For long-term control of inflammation with reduced adverse events or side effects compared to topical corticosteroids (TCS), alternative options are considered, particularly in intertriginous areas [32]. Cream Tacrolimus (0.1%) has demonstrated more effective penetration than cream pimecrolimus (1%), primarily due to vehicle characteristics [33]. Umar et al. suggested using intermittent therapy for breakthrough cases, employing clobetasol propionate 0.05% foam and switching every six weeks between daily tacrolimus and daily clobetasol foam [34]. In a half-side comparative study, two patients underwent a once-daily application of topical cyclosporine. The first patient’s results were contrasted with TCS, and the second patient’s with cleared in both patients [35].
5-Fluorouracil: Dammak A et al. demonstrated the use of topical 5-fluorouracil 5% cream for the treatment of groin-only HHD. The cream was applied three times a week for three months, followed by once a week for an additional three months. The patient achieved total remission after three months, and there were no relapses observed in the following year [36]. However, further case studies are necessary to explore the actual effectiveness and mode of action of this topical medication.
Vitamin D Analogs: The utilization of topical vitamin D analogs, such as calcitriol, in HHD patients has yielded variable results in terms of efficacy due to limited research. In two case reports, applying calcitriol ointment twice daily resulted in total clearance of skin lesions, with remission observed after three months of follow-up [37]. Another case study involved a split-lesion treatment comparing tacalcitol to betamethasone dipropionate ointment in the same patient, with greater improvement noted when using tacalcitol [38]. Further research is needed to better understand the effectiveness and optimal use of topical vitamin D analogs in HHD management.
Cadexomer Iodine: Topical Cadexomer Iodine possesses absorbent, antimicrobial, anti-inflammatory, and healing-promoting properties. Tang MBY et al. reported complete clearance of HHD within 10 months using this topical agent [39]. However, further research is necessary to establish the efficacy and suitability of topical Cadexomer Iodine as a therapeutic option for HHD.
Antibiotics: Patients with HHD may potentially benefit from gentamicin since 20% of pathogenic mutations in the disease result in an early stop codon that truncates the calcium ATPase [40]. Kellermayer et al. effectively demonstrated this in a single patient with an ATP2C1 mutation (1402C>T) resulting in a UGA premature stop mutation. The administration of Gentamicin 0.1% (with an unstated vehicle) for 7–10 days led to full resolution in the patient. They further validated these results in vitro using a yeast model with the same mutation, replacing gentamicin with paromomycin [41]. However, more research is needed to determine the broader applicability and effectiveness of gentamicin in treating HHD.
Oral Therapy
Includes systemic medications such as antimicrobials (Antibiotics, Dapsone, Terbinafine), retinoids, immunosuppressive drugs (Corticosteroids, Methotrexate, Thalidomide, Cyclosporine, Azathioprine), and biological drugs (Tumor Necrosis Factorα Inhibitors).
Antibiotics: Topical treatments are often combined with oral antibiotics in the management of HHD. In a case series, oral erythromycin or penicillin antibiotics showed improvement in 43% of 58 patients [30].
Persić-Vojinović et al. found that the combination of oral erythromycin and topical tacrolimus completely resolved disseminated HHD [42]. Le Saché-de P et al. reported that after taking doxycycline 100 mg daily for three months, five out of six patients experienced complete remission. Three out of these five patients remained symptom-free with a daily dose of 50–100 mg of doxycycline, one reported less frequent disease activity, and two experienced complete remission for five years [43]. Higashimae et al. documented that two HHD patients who received 200–300 mg of nicotinamide and minocycline daily showed partial recovery from their condition [44]. It’s important to note that while these reports show promise, further research is needed to establish the broader efficacy and safety of these oral antibiotic treatments for HHD.
Dapsone: Dapsone, known for its antibacterial and anti-inflammatory properties, has been used in the treatment of HHD. Dr. Bernett Johnson demonstrated the efficacy of oral dapsone in three patients. They initially received a dosage of 100–200 mg daily, which was later tapered to a maintenance dose of 50 mg daily. This treatment approach resulted in the resolution of pruritus and skin lesions in these patients [45]. Nevertheless, further research is essential to validate the effectiveness and safety of dapsone as a therapeutic option for HHD on a broader scale.
Terbinafine: While not commonly used, there have been reports of terbinafine being effective in treating HHD. Lew T et al. described a single case in which a patient had both HHD and Trichophyton rubrum. The patient took 250 mg of terbinafine daily for four weeks, leading to complete resolution of the disease. Remarkably, the effects persisted for eight months after discontinuing the medication [46]. Despite this promising case, additional research is necessary to establish the broader applicability and safety of terbinafine as a treatment for HHD.
Retinoids: Oral retinoids, such as isotretinoin, etretinate, and acitretin, are employed in the treatment of HHD. The mechanism of action is believed to involve the normalization of keratinocyte differentiation. Etretinate has demonstrated complete resolution of lesions in two additional cases at both low (25 mg/day) and high (60 mg/day) doses, with improvement observed between two and six weeks [47]. Sárdy M and Ruzicka T reported a case where a patient tapering off oral prednisolone and taking 30 mg of isotretinoin daily showed improvement. Clinical remission was achieved when isotretinoin was replaced with alitretinoin at the same dosage. Another patient, unresponsive to topical treatments or ablative laser therapy, experienced complete remission after a month of oral alitretinoin at a 30 mg/day dose. Both patients had very few relapses [48]. While these findings are promising, further research is required to establish the broader efficacy and safety of oral retinoids in HHD treatment.
Corticosteroids: Due to the worsening of HHD when oral corticosteroids are discontinued, they are rarely prescribed for short-term treatment. A retrospective study conducted by Benmously-Mlika R et al. involved 17 HHD patients taking prednisolone orally. After a 13-month follow-up, only one patient with severe HHD showed better results compared to the other patients [49]. This suggests that while oral corticosteroids may provide some benefit, their long-term use for HHD is limited, and alternative treatments with more sustained effectiveness are often explored.
Methotrexate: Methotrexate, extensively used in dermatology, has been shown to be effective in treating refractory HHD. Vilarinho C et al. demonstrated prolonged (15 mg per week) and complete (7.5 mg per week) reduction of the illness in refractory HHD patients [50]. This indicates that methotrexate can be a valuable therapeutic option for managing HHD, particularly in cases where other treatments have not been successful.
Thalidomide: Thalidomide may be considered a viable option for patients with severe HHD. Nanda KB et al. demonstrated that patients who did not show improvement with oral dapsone or intravenous corticosteroids achieved complete remission after taking 100 mg of thalidomide orally three times a day for a week. The dosage was subsequently reduced to 50 mg per day and maintained for six months, with no recurrence of the disease after treatment cessation [51]. It’s important to note that in some patients, discontinuation of thalidomide at a daily dose of 100 mg within 5 months can lead to the occurrence of peripheral neuropathy [52]. Thalidomide’s potential efficacy should be weighed against its side effects, and its use should be carefully monitored.
Cyclosporine: Cyclosporine is commonly employed for the rapid reduction of symptoms in HHD resistant patients. However, it’s noted that patients may experience recovery after discontinuing this medication [53]. Despite its effectiveness, long-term use of cyclosporine is associated with adverse events, including nephrotoxicity and hypertension. Nanda et al. reported that one patient successfully used cyclosporine at a low dose of 2.5 mg/kg/day for three weeks, resulting in the complete absence of lesions. The medication was gradually reduced over six months, leading to a two-year disease remission. Topical tacrolimus was employed to manage minor side effects [54]. The potential benefits of cyclosporine must be weighed against its side effects, and its use should be carefully monitored.
Azathioprine: Azathioprine is utilized in the management of HHD, particularly in patients with inguinal and axillary lesions. Malan M et al. reported a case where the patient initially received oral cetirizine and injectable glycyrrhizin acid for anti-allergic treatment, along with a topical antibiotic for three days. Azathioprine was then added to the regimen. After 5 days of azathioprine treatment, there was a significant improvement, and after 3 months, there was a partial remission [55]. This suggests that azathioprine may be a valuable component in the management of HHD, although further research is needed to establish its broader efficacy and safety in this context.
Tumor Necrosis Factorα (TNF-α) Inhibitors: TNF-α antagonist Etanercept has been explored in the treatment of HHD. Norman R et al. reported a case where the starting dose of etanercept therapy was 25 mg per week given subcutaneously for one month. To enhance the patient’s response, the dosage was then increased to 50 mg once a week for six months and further raised to 75 mg once a week. The conclusion drawn from this case study was that Etanercept helped to relieve HHD symptoms [56]. While these findings are promising, additional research is necessary to establish the broader efficacy and safety of Etanercept in treating HHD.
Ablative/Procedural Therapy
Includes lasers and light therapy (Photodynamic Therapy, carbon dioxide Laser, Diode Laser,
Erbium-doped yttrium aluminium garnet (Erbium-YAG) Laser, Pulsed Dye Laser), Botulinum Toxin Type A (BTA), Radiofrequency Surgery, Narrowband Ultraviolet B Phototherapy (NbUVB), mechanical dermabrasion.
Photodynamic therapy (PDT): It is a targeted treatment approach that utilizes a specific wavelength of light along with a photosensitizing medication to target lesions. Ruiz-Rodriguez R et al. demonstrated positive results in two patients with persistent, resistant HHD using topical 20% 5-aminolevulinic acid (ALA) in conjunction with incoherent light irradiation at a wavelength of 590 to 700 nm. After 2 weeks of therapy and the second treatment, the lesions disappeared, and at the 22-month follow-up, the patients were still in remission [57]. On the other hand, Fernandez Guarino et al. evaluated the outcomes of treating three patients with HHD using a single session of PDT involving the topical application of methyl ALA under occlusion for 3 hours, followed by red light exposure. Unfortunately, the results were not favorable, with all patients experiencing discomfort in the 3–4 weeks post-PDT, and only one showing clinical improvement. None of the patients expressed interest in undergoing the treatment again [58]. Despite mixed outcomes, PDT remains an area of exploration for HHD treatment.
CO2 Laser: CO2 laser ablation has shown significant efficacy in treating HHD, leading to prolonged or permanent remission in 13 patients, as highlighted by Hochwalt PC et al. The outcomes of this treatment have resulted in high patient satisfaction and substantial improvement in their quality of life (QoL). CO2 laser ablation is considered a promising and gratifying treatment option for HHD [59]. Similarly, Gabdrafike Z et al. reported successful fractional ablative CO2 laser treatment of recalcitrant HHD in a 35-year-old female. The laser, operating at a wavelength of 10600 nm with a power range of 7-10 W and 2-3 passes, yielded positive outcomes. The patient, with a 7-year history of therapy-resistant HHD, exhibited substantial resolution of lesions during the 2-month follow-up. Mild erythema and post-inflammatory hyperpigmentation were noted in the treated areas, indicating the effectiveness of fractional ablative CO2 laser in addressing this challenging condition [60]. These findings highlight the potential of CO2 laser therapy as a valuable intervention for HHD.
Diode Laser: There is a single documented case in the literature detailing the use of a 1450-nm diode laser in the treatment of HHD. The procedure consisted of three treatment sessions utilizing a fluence of 14 J/cm², a 6-mm spot size, and a dynamic cooling device with a 50 ms duration. Topical anesthesia was applied during the sessions; however, the patient experienced significant pain during the procedure. Fortunately, there were no adverse effects observed, except for a reduction in sweating and malodor of the lesions. Unfortunately, there was no improvement in the underlying condition [61]. This underscores the necessity for additional clinical studies to substantiate the effectiveness of the diode laser in the treatment of HHD. The limited evidence available highlights the need for further research to determine the potential benefits and limitations of diode laser therapy for HHD.
Erbium:YAG Laser: The erbium-YAG laser, classified as a solid-state crystal laser, operates at a wavelength of 2,940 nm. Its unique characteristic lies in its strong absorption by water, which restricts the laser from cutting through the skin extensively, minimizing the potential for extensive scarring [62]. Erbium:YAG laser ablation is considered an exceptionally effective and safe therapeutic approach for treating HHD. Its advantages include swift ablation of large areas and the precision of point-by-point removal for keratotic papules, along with the capability for localized deeper ablation when required. This versatility and efficacy contribute to the superiority of erbium:YAG laser ablation over abrasive methods or carbon dioxide laser vaporization in the treatment of these dermatological conditions [63].
Pulsed Dye Laser: Hunt KM et al investigated that seven instances of severe HHD treated through sessions of 595-nm pulsed dye laser every 2–4 weeks, averaging a total of five treatments per patient. Within a span of 2–8 weeks, six patients witnessed varying degrees of improvement. However, only two patients achieved a sustained and prolonged remission [64].
BTA: BTA has emerged as a prominent alternative therapy for HHD. By impeding the release of acetylcholine from nerve terminals, BTA facilitates the inhibition of sudorific nerves, leading to a reduction in sweat production by the eccrine glands [33]. In a study conducted by Dousset et al., eight HHD patients received a median dose of 300 IU per site of BTA, resulting in a favorable clinical response and complete remission in 80% of the injected areas [65]. Another study involving six patients demonstrated a significant improvement following BTA treatment [66]. Charlton et al. recently documented a case of a patient with refractory disease unresponsive to topical corticosteroids (TCS) and CO2 laser therapy, achieving complete remission with BTA [67]. Some reports even advocate for BTA as a first-line treatment for HHD at a dose of 50 IU per site [68].
Radiofrequency Surgery: Radiofrequency surgery, akin to other surgical methods, results in the ablation of the affected skin, facilitating re-epithelialization with normal keratinocytes produced from skin appendices. Nandina A et al documented case of HHD treated with radiofrequency surgery showed complete clearance without recurrence, observed 16 weeks post-therapy [69].
NbUVB: The impact of ultraviolet (UV) light exposure varies among patients. While some individuals may exhibit flares, even in unaffected skin, after sun exposure, others may experience clinical improvement. In laboratory studies, UVB light has been shown to suppress ATP2C1 mRNA, but as a standalone cellular stress factor, it is insufficient for inducing HHD lesions [70]. Several case reports have suggested the effectiveness of NbUVB therapy, either as a standalone treatment or in synergy with retinoids, for HHD [71]. For carefully selected patients, NbUVB phototherapy could be considered as a promising treatment option for managing HHD.
Mechanical dermabrasion: Dermabrasion, utilizing a wire brush or a diamond wheel with rough edges, leads to the removal of the epidermis and superficial dermis while preserving adnexal structures, allowing subsequent re-epithelialization. This procedure is typically performed under local or general anesthesia and necessitates subsequent wound care [8]. In a study conducted by Hamm et al., dermabrasion was applied to ten patients with HHD across a total of 46 regions. Remarkably, remission was observed for up to 79 months in 38 of the treated areas [72].
Conclusion
Due to the persistent and challenging nature of HHD, this comprehensive review is discussed regarding different treatment therapies, including case reports and case series. Among the therapies, topical corticosteroids are considered as the first-line therapy. However, there is an argument that BTA could also be considered first-line if it proves to be cost-effective given its demonstrated efficacy. Furthermore, Combination therapy with both first- and second-line treatments may show better results than single therapy.
Systemic agents, oral antibiotics, particularly Tetracyclines, are beneficial when used in conjunction with topical agents. Continuous CO2 laser therapy is considered an alternative procedural therapy for patients failing to respond to first-line therapies whereas Fractional CO2 laser therapy is benefit in patients with less side effects. Additionally, dermabrasion is a comparatively low-cost, non-invasive treatment and considered as a second-line therapy. In the case of HHD emergency, naltrexone is used as a first-line treatment drug due to its low cost and lack of serious adverse events. Finally, there is a need for larger research studies to evaluate and compare the effectiveness of different treatments.
References
- Adusumilli NC, Friedman AJ. Benign Familial Pemphigus. JAMA Dermatol. 2022;158(3):315. doi:10.1001/jamadermatol.2021.5177 PubMed | Crossref | Google Scholar
- de Aquino Paulo Filho T, deFreitas YK, da Nóbrega MT, et al. Hailey-Hailey disease associated with herpetic eczema the value of the Tzanck smear test. Dermatol Pract Concept. 2014;4(4):29-31. doi:10.5826/dpc.0404a04 PubMed | Crossref | Google Scholar
- Deng H, Xiao H. The role of the ATP2C1 gene in Hailey-Hailey disease. Cell Mol Life Sci. 2017;74(20):3687-3696. doi:10.1007/s00018-017-2544-7 PubMed | Crossref | Google Scholar
- Buteică E, Burada F, Stoicescu I, Stănoiu B, Georgescu CV. Darier disease and Hailey-Hailey disease. Rom J Morphol Embryol. 2007;48(4):423-426. Darier disease and Hailey-Hailey disease
- Orseth ML, Cropley TG. What’s in a name? Hailey-Hailey disease. JAMA Dermatol. 2013;149(10):1220. doi:10.1001/jamadermatol.2013.544 PubMed | Crossref | Google Scholar
- Ikeda S, Shigihara T, Mayuzumi N, Yu X, Ogawa H. Mutations of ATP2C1 in Japanese patients with Hailey-Hailey disease: intrafamilial and interfamilial phenotype variations and lack of correlation with mutation patterns. J Invest Dermatol. 2001;117(6):1654-1656. doi:10.1046/j.0022-202x.2001.01552.x. Crossref | Google Scholar
- Rao AG. Hailey-Hailey disease on sun-exposed areas. Indian J Dermatol. 2013;58(5):412. doi:10.4103/0019-5154.117371 PubMed | Crossref | Google Scholar
- Farahnik B, Blattner CM, Mortazie MB, Perry BM, Lear W, Elston DM. Interventional treatments for Hailey-Hailey disease. J Am Acad Dermatol. 2017;76(3):551-558.e3. doi:10.1016/j.jaad.2016.08.058 Crossref | Google Scholar
- Alamon-Reig F, Serra-García L, Bosch-Amate X, Riquelme-McLoughlin C, Mascaró JM Jr. Dupilumab in Hailey-Hailey disease: a case series. J Eur Acad Dermatol Venereol. 2022;36(10):e776-e779. doi:10.1111/jdv.18442. Crossref | Google Scholar
- Sousa Gomes M, Araújo Pereira J, Trocado V, Prata JP, Teixeira V, Pinheiro P. Vulvar Hailey-Hailey disease treated with low-dose naltrexone: case report and literature review. Arch Gynecol Obstet. 2020;302(5):1081-1086. doi:10.1007/s00404-020-5705-0 PubMed | Crossref | Google Scholar
- Hailey Hailey Disease. Wikipedia. 2025 Hailey Hailey Disease
- Li Y, Jiang Y, Sun J. Improvement of Hailey-Hailey disease with abrocitinib. Clin Exp Dermatol. 2023;48(5):532-533. doi:10.1093/ced/llad019 Crossref | Google Scholar
- Thompson LD. Hailey-Hailey disease. Ear Nose Throat J. 2016;95(9):370. doi:10.1177/014556131609500916 Crossref | Google Scholar
- Burge SM. Hailey-Hailey disease: the clinical features, response to treatment and prognosis. Br J Dermatol. 1992;126(3):275-282. doi:10.1111/j.1365-2133.1992.tb00658.x PubMed | Crossref | Google Scholar
- Chun I, Whang KC, Su WP. Squamous cell carcinoma arising in Hailey-Hailey disease. J Cutan Pathol. 1988;15(4):234-237. doi:10.1111/j.1600-0560.1988.tb00551.x PubMed | Crossref | Google Scholar
- Deng H, Xiao H. The role of the ATP2C1 gene in Hailey-Hailey disease. Cell Mol Life Sci. 2017;74(20):3687-3696. doi:10.1007/s00018-017-2544-7 PubMed | Crossref | Google Scholar
- Mauro TM. Yeast researchers consider Hailey-Hailey disease. J Invest Dermatol. 2004;123(6):xxii-xxiii. doi:10.1111/j.0022-202X.2004.23534.x. Crossref
- Aronchik I, Behne MJ, Leypoldt L, et al. Actin reorganization is abnormal and cellular ATP is decreased in Hailey-Hailey keratinocytes. J Invest Dermatol. 2003;121(3):681-687. doi:10.1046/j.1523-1747.2003.12483.x Crossref | Google Scholar
- Manca S, Magrelli A, Cialfi S, et al. Oxidative stress activation of miR-125b is part of the molecular switch for Hailey-Hailey disease manifestation. Exp Dermatol. 2011;20(11):932-937. doi:10.1111/j.1600-0625.2011.01353.x Crossref | Google Scholar
- Burge SM, Millard PR, Wojnarowska F. Hailey-Hailey disease: a widespread abnormality of cell adhesion. Br J Dermatol. 1991;124(4):329-332. doi:10.1111/j.1365-2133.1991.tb04947.x Crossref | Google Scholar
- Rogner DF, Lammer J, Zink A, Hamm H. Darier and Hailey-Hailey disease: update 2021. J Dtsch Dermatol Ges. 2021;19(10):1478-1501. doi:10.1111/ddg.14619. PubMed | Crossref | Google Scholar
- Reddy VB. Skin and adnexal structures. In: Gattuso P, Reddy VB, David O, Spitz DJ, Haber MH, eds. Differential Diagnosis in Surgical Pathology. 2nd ed. Philadelphia, PA: Elsevier; 2010:39-120. doi:10.1016/B978-1-4160-4580-9.00002-2 Crossref | Google Scholar
- Groves JB, Freeman AM. Erythrasma. In: StatPearls. Treasure Island, FL: StatPearls 2018. Erythrasma
- Ruocco V, Ruocco E, Caccavale S, Gambardella A, Lo Schiavo A. Pemphigus vegetans of the folds (intertriginous areas). Clin Dermatol. 2015;33(4):471-476. doi:10.1016/j.clindermatol.2015.04.006 Crossref | Google Scholar
- Syed ZU, Khachemoune A. Inverse psoriasis: case presentation and review. Am J Clin Dermatol. 2011;12(2):143-146. doi:10.2165/11539470-000000000-00000 PubMed | Crossref | Google Scholar
- Gomes J, Labareda J, Viana I. Galli-Galli disease: a rare acantholytic variant of Dowling-Degos disease. Case Rep Med. 2011;2011:703257. doi:10.1155/2011/703257 PubMed | Crossref | Google Scholar
- Scheinfeld NS, Mones J. Granular parakeratosis: pathologic and clinical correlation of 18 cases of granular parakeratosis. J Am Acad Dermatol. 2005;52(5):863-867. doi:10.1016/j.jaad.2004.12.034 Crossref | Google Scholar
- Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: a comprehensive review. Indian Dermatol Online J. 2016;7(2):77-86. doi:10.4103/2229-5178.178099 PubMed | Crossref | Google Scholar
- Tomaszewska KA, Gerlicz-Kowalczuk Z, Kręgiel M, et al. The coexistence of Darier’s disease and Hailey-Hailey disease symptoms. Postepy Dermatol Alergol. 2017;34(2):180-183. doi:10.5114/ada.2017.67077 Crossref | Google Scholar
- Burge SM. Hailey-Hailey disease: the clinical features, response to treatment and prognosis. Br J Dermatol. 1992;126(3):275-282. doi:10.1111/j.1365-2133.1992.tb07829.x Crossref | Google Scholar
- Ikeda S, Suga Y, Ogawa H. Successful management of Hailey-Hailey disease with potent topical steroid ointment. J Dermatol Sci. 1993;5(3):205-211. doi:10.1016/0923-1811(93)90768-k PubMed | Crossref | Google Scholar
- Dattola A, Silvestri M, Bennardo L, et al. Update of calcineurin inhibitors to treat inverse psoriasis: a systematic review. Dermatol Ther. 2018;31(6):e12728. doi:10.1111/dth.12728. PubMed | Crossref | Google Scholar
- Arora H, Bray F, Cervantes J, Falto-Aizpurua L. Management of familial benign chronic pemphigus. Clin Cosmet Investig Dermatol. 2016;9:281-290. doi:10.2147/CCID.S89483 PubMed | Crossref | Google Scholar
- Umar S, Bhattacharjee P, Brodell R. Treatment of Hailey-Hailey disease with tacrolimus ointment and clobetasol propionate foam. J Drugs Dermatol. 2004;3(2):200-203. Umar S, Bhattacharjee P, Brodell R. Treatment of Hailey-Hailey disease with tacrolimus ointment and clobetasol propionate foam
- Jitsukawa K, Ring J, Weyer U, Kimmig W, Radlof H. Topical cyclosporine in chronic benign familial pemphigus (Hailey-Hailey disease). J Am Acad Dermatol. 1992;27(4):625-626. doi:10.1016/s0190-9622(08)80201-0 PubMed | Crossref | Google Scholar
- Dammak A, Camus M, Anyfantakis V, Guillet G. Successful treatment of Hailey-Hailey disease with topical 5-fluorouracil. Br J Dermatol. 2009;161(4):967-968. doi:10.1111/j.1365-2133.2009.09302.x Crossref | Google Scholar
- Rajpara S, King C. Hailey-Hailey disease responsive to topical calcitriol. Br J Dermatol. 2005;152(4):816-817. doi:10.1111/j.1365-2133.2005.06483.x Crossref | Google Scholar
- Aoki T, Hashimoto H, Koseki S, Hozumi Y, Kondo S. 1α,24-Dihydroxyvitamin D3 (tacalcitol) is effective against Hailey-Hailey disease both in vivo and in vitro. Br J Dermatol. 1998;139(5):897-901. doi:10.1046/j.1365-2133.1998.02509.x Crossref | Google Scholar
- Tang MBY, Tan EST. Hailey-Hailey disease: effective treatment with topical cadexomer iodine. J Dermatolog Treat. 2011;22(5):304-305. doi:10.3109/09546631003762670 PubMed | Crossref | Google Scholar
- Pasmooij AMG. Topical gentamicin for the treatment of genetic skin disease. 2018;138(4):731-734. Topical gentamicin for the treatment of genetic skin disease
- Kellermayer R, Szigeti R, Keeling KM, Bedekovics T, Bedwell DM. Aminoglycosides as potential pharmacogenetic agents in the treatment of Hailey-Hailey disease. J Invest Dermatol. 2006;126(1):229-231. doi:10.1038/sj.jid.5700031 PubMed | Crossref | Google Scholar
- Persić-Vojinović S, Milavec-Puretić V, Dobrić I, Radoš J, Špoljar S. Disseminated Hailey-Hailey disease treated with topical tacrolimus and oral erythromycin: case report and review of the literature. Acta Dermatovenerol Croat. 2006;14(4):253-257. Disseminated Hailey-Hailey disease treated with topical tacrolimus and oral erythromycin: case report and review of the literature
- Le Saché-de Peufeilhoux L, Raynaud E, Bouchardeau A, Fraitag S, Bodemer C. Familial benign chronic pemphigus and doxycycline: a review of 6 cases. J Eur Acad Dermatol Venereol. 2014;28(3):370-373. doi:10.1111/jdv.12107 Crossref | Google Scholar
- Elfatoif FZ, Fanian F, Chiheb S, Humbert P. Hailey-Hailey disease: efficiency of combined doxycycline and vitamin PP. Ann Dermatol Venereol. 2017;144(3):216-217. doi:10.1016/j.annder.2016.12.007 Crossref | Google Scholar
- Sire DJ, Johnson BL. Benign familial chronic pemphigus treated with dapsone. Arch Dermatol. 1971;103(3):262-265. Benign familial chronic pemphigus treated with dapsone
- Lew T, Kaur M, Hague J. Oral terbinafine as an alternative treatment for Hailey-Hailey disease. Clin Exp Dermatol. 2013;38(8):929-930. doi:10.1111/ced.12185 Crossref | Google Scholar
- Hunt MJ, Salisbury ELC, Painter DM, Lee S. Vesiculobullous Hailey-Hailey disease: successful treatment with oral retinoids. Australas J Dermatol. 1996;37(4):196-198. doi:10.1111/j.1440-0960.1996.tb01052.x Crossref | Google Scholar
- Sárdy M, Ruzicka T. Successful therapy of refractory Hailey-Hailey disease with oral alitretinoin. Br J Dermatol. 2014;170(1):209-211. doi:10.1111/bjd.12654 Crossref | Google Scholar
- Benmously-Mlika R, Bchetnia M, Deghais S, et al. Hailey-Hailey disease in Tunisia. Int J Dermatol. 2010;49(4):396-401. doi:10.1111/j.1365-4632.2010.04403.x PubMed | Crossref | Google Scholar
- D’Errico A, Bonciani D, Bonciolini V, et al. Hailey-Hailey disease treated with methotrexate. J Dermatol Case Rep. 2012;6(2):49-51. doi:10.3315/jdcr.2012.1103 Crossref | Google Scholar
- Nanda KB, Saldanha CS, Jacintha M, Kamath G. Hailey-Hailey disease responding to thalidomide. Ind J Dermatol. 2014;59(2):190-192. doi:10.4103/0019-5154.127684 PubMed | Crossref | Google Scholar
- Dadban A, Guillot B. Oesophageal involvement in familial benign chronic pemphigus. Acta Derm Venereol. 2006;86(3):252-253. doi:10.2340/00015555-0069 Crossref | Google Scholar
- Berth-Jones J, Smith SG, Graham-Brown RAC. Benign familial chronic pemphigus (Hailey-Hailey disease) responds to cyclosporin. Clin Exp Dermatol. 1995;20(1):70-72. doi:10.1111/j.1365-2230.1995.tb01204.x PubMed | Crossref | Google Scholar
- Nanda A, Khawaja F, Harbi R, et al. Benign familial pemphigus (Hailey-Hailey disease) responsive to low dose cyclosporine. Ind J Dermatol Venereol Leprol. 2010;76(4):422-424. doi:10.4103/0378-6323.66595 Crossref | Google Scholar
- Malan M, Xuejingzi W, Si J, Quan SJ. Hailey-Hailey disease: the role of azathioprine an immunomodulator. Pan Afr Med J. 2019;32:65. doi:10.11604/pamj.2019.32.65.17877 PubMed | Crossref | Google Scholar
- Norman R, Greenberg RG, Jackson JM. Case reports of etanercept in inflammatory dermatoses. J Am Acad Dermatol. 2006;54(1):139-142. doi:10.1016/j.jaad.2005.09.015. Crossref | Google Scholar
- Ruiz-Rodriguez R, Alvarez JG, Jaén P, et al. Photodynamic therapy with 5-aminolevulinic acid for recalcitrant familial benign pemphigus (Hailey-Hailey disease). J Am Acad Dermatol. 2002;47(5):740-742. doi:10.1067/mjd.2002.124605 Crossref | Google Scholar
- Fernández Guarino M, Ryan AM, Harto A, et al. Experience with photodynamic therapy in Hailey-Hailey disease. J Dermatolog. 2008;19(5):288-290. doi:10.1080/09546630801958220 PubMed | Crossref | Google Scholar
- Hochwalt PC, Christensen KN, Cantwell SR, et al. Carbon dioxide laser treatment for Hailey-Hailey disease: a retrospective chart review with patient-reported outcomes. Int J Dermatol. 2015;54(11):1309-1314 doi:10.1111/ijd.12991 PubMed | Crossref | Google Scholar
- Gabdrafike, Z., Blazeviciute, M., Janonyte, U., Makstiene, J., & Valiukeviciene, S. A case report of Hailey-Hailey disease treated with fractional carbon dioxide laser. Dermatologic Reports 2023;15(3): 9658. doi:10.4081/dr.2023.9658 PubMed | Crossref | Google Scholar
- Wollina U. Erbium-YAG laser therapy: analysis of more than 1,200 treatments. Glob Dermatol. 2016;3(2):268-272. doi:10.15761/GOD.1000171 Crossref | Google Scholar
- Beier C, Kaufmann R. Efficacy of erbium:YAG laser ablation in Darier disease and Hailey-Hailey disease. Arch Dermatol. 1999;135(4):423-427. doi:10.1001/archderm.135.4.423 PubMed| Crossref | Google Scholar
- Downs A. Smoothbeam laser treatment may help improve hidradenitis suppurativa but not Hailey-Hailey disease. J Cosmet Laser Ther. 2004;6(3):163-164. doi:10.1080/14764170410003057 Crossref | Google Scholar
- Hunt KM, Jensen JD, Walsh SB, et al. Successful treatment of refractory Hailey-Hailey disease with a 595-nm pulsed dye laser: a series of 7 cases. J Am Acad Dermatol. 2015;72(4):735-737. doi:10.1016/j.jaad.2015.01.011 Crossref | Google Scholar
- L. Dousset, A. Pham-Ledard, M.S. Doutre. Treatment of Hailey–Hailey disease with botulinic toxin: a retrospective study of 8 cases. Annals of Dermatology and Venereology. 2017;144(10):599-606. Treatment of Hailey–Hailey disease with botulinic toxin: a retrospective study of 8 cases
- Koeyers WJ, van der Geer S, Krekels G. Botulinum toxin type A as an adjuvant treatment modality for extensive Hailey-Hailey disease. J Dermatolog Treat. 2008;19(4):251-254. doi:10.1080/09546630801955135 PubMed| Crossref | Google Scholar
- Charlton OA, Stewart TJ, Rosen RH. Treatment of Hailey-Hailey disease with botulinum toxin. Australas J Dermatol. 2018;59(3):229-231. do.:10.1111/ajd.12682 Crossref | Google Scholar
- Kothapalli A, Caccetta T. Botulinum toxin type A for the first-line treatment of Hailey-Hailey disease. Australas J Dermatol. 2019;60(1):73-74. doi:10.1111/ajd.12869 Crossref | Google Scholar
- Nandini A, Mysore V. Hailey-Hailey disease: a novel method of management by radiofrequency surgery. J Cutan Aesthet Surg. 2009;1(2):92-93. doi:10.4103/0974-2077.44163 Crossref | Google Scholar
- Bernards M, Korge BP. Desmosome assembly and keratin network formation after Ca2+/serum induction and UVB radiation in Hailey-Hailey keratinocytes. J Invest Dermatol. 2000;114(5):1058-1061. doi:10.1046/j.1523-1747.2000.00974.x Crossref | Google Scholar
- Vanderbeck KA, Giroux L, Murugan NJ, Karbowski LM. Combined therapeutic use of oral alitretinoin and narrowband ultraviolet-B therapy in the treatment of Hailey-Hailey disease. Dermatol Rep. 2014;6(1):5604. doi:10.4081/dr.2014.5604 PubMed| Crossref | Google Scholar
- Hamm H, Metze D, Bröcker EB. Hailey-Hailey disease: eradication by dermabrasion. Arch Dermatol. 1994;130(9):1143-1149. Hailey-Hailey disease: eradication by dermabrasion
Acknowledgments
Not applicable
Funding
Not applicable
Author Information
Raziya Begum Sheikh
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Email: raziya.pharma@gmail.com
Author Contribution
The author contributed to the conceptualization, investigation, and data curation by acquiring and critically reviewing the selected articles and was involved in the writing – original draft preparation and writing – review & editing to refine the manuscript.
Informed Consent
Not applicable
Conflict of Interest Statement
Not applicable.
Guarantor
None
DOI
Cite this Article
Begum RS. A literature review on Hailey-Hailey disease. medtigo J Med. 2023;1(3):e3062132. doi:10.63096/medtigo3062132 Crossref









