Author Affiliations
Abstract
Herpes zoster, commonly known as shingles, is a localized ailment caused by the reactivation of the varicella-zoster virus (VZV). The virus initially infiltrates cutaneous nerve endings during a previous episode of chickenpox, after which it travels to the dorsal root ganglia and assumes a latent form. This condition is characterized by the appearance of multiple painful vesicles and ulceration on one side of the body, typically following a specific dermatome that is innervated by a single dorsal root or cranial sensory ganglion. In this case report, we present a 60-year-old male with an erythematous rash on his and neck region that started as mild pain. He has had a history of type 2 diabetes mellitus for 15 years and is currently undergoing twice-daily therapy with gliclazide and metformin. This case underscores the importance of considering herpes zoster infection in individuals with predisposing conditions, such as diabetes mellitus. It emphasizes the need for a comprehensive clinical evaluation to guide appropriate management.
Keywords
Herpes zoster virus, Infection, Diabetes mellitus, Blisters, Lesions, Physical examination
Introduction
Herpes zoster is characterized by a blistering skin rash, typically triggered by the reawakening of the dormant VZV, which initially causes chickenpox, from neurons located in the dorsal root ganglia. Herpes zoster commonly manifests on dermatomes in the thoracic or cranial regions. The overall lifetime likelihood of developing shingles is approximated to be 10-20%, yet this risk increases by 50% among individuals aged 80 years or older [1]. The reactivation can be prompted by various factors like trauma, aging, or immunodeficiency [1,2]. It remains uncertain whether additional factors such as radiation, physical injury, specific medications, infections, and stress may also serve as triggers for the onset of shingles [2,3]. The reasons for the inability of circulating varicella antibodies and cell-mediated immune mechanisms to prevent the recurrence of evident disease, unlike in most other viral infections, remain somewhat unclear. The incidence of zoster appears to be inversely correlated with the host’s ability to enhance the cellular immune response. However, a significant number of individuals with zoster still display normal immune function [3–5]. The hypothesis in such cases suggests that zoster occurs when both the antibody titers against the varicella-zoster virus (VZV) and cellular immunity decrease to a level where they no longer offer complete protection against viral intrusion.
Herpes zoster typically appears in one or two adjacent dermatomes, with a higher incidence in the thoracic region
(50-60%), followed by the cervical (10-20%) and trigeminal (10-20%) dermatomes. In contrast, the lumbar (5-10%) and sacral (5%) dermatomes are less frequently affected. In individuals with a well-functioning immune system, the occurrence of herpes zoster in non-contiguous dermatomes is absent, although there is evidence of overlap in 20% of cases involving adjacent dermatomes. The rarity of cases where the maxillary and mandibular branches are affected without involvement of the ocular branch accounts for approximately 1.7% to 2% of herpes zoster cases [6,7].
This case intends to underscore the diverse clinical manifestations of herpes zoster infection observed in this patient with distinct underlying medical conditions.
Case Presentation
In this case presentation, a 60-year-old male sought medical attention in an emergency room due to a painful rash that initially began as mild discomfort in the face and neck region. Alongside the rash, the individual reported experiencing fever and chills over the past two days. The patient had a history of type-2 diabetes mellitus for the last 15 years, which is controlled with gliclazide and metformin. No significant medical, dental, or family history details were provided. The patient’s vital signs were within normal limits.
Upon physical examination, prominent findings included fluid-filled blisters spread on the right side of his face. These initially presented on the lower lip and extended to the chin, cheek, and forehead, progressing gradually to involve the eye, and subsequently affecting the earlobe on the same side (Figure 1). Several fluid-filled sacs merged to create blisters on the right side of the chin. The skin around the sacs appeared erythematous. The examination inside the mouth revealed ruptured vesicles, which had merged to create sizable ulcers varying in diameter from 1 to 2 cm. These ulcers were distributed extensively across the left side of the lower lip and buccal mucosa. Characterized by irregular shapes, the ulcers exhibited a pseudomembranous slough at the base, surrounded by an erythematous halo. Palpation elicited tenderness. Examination beyond the mouth and face revealed numerous fluid-filled lesions on the anterior mandible region below the lower lip. Vesicles also showed signs of involvement in the preauricular, auricular, and temple scalp regions, accompanied by the presence of crusts. The adjacent skin appeared red and was notably tender upon Palpation.
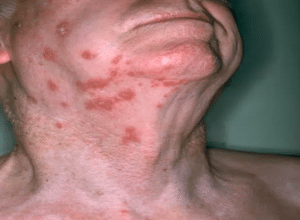
Figure 1: Erythematous rash on his face
Case Management
The patients received antiviral and supportive therapy for one week. Valacyclovir (1 g) was administered to manage the active viral phase. Pain and fever were alleviated with Tramadol hydrochloride (100 mg) and acetaminophen (325 mg). For facial and intraoral lesions, acyclovir cream (5%) was topically applied three times a day. Lignocaine gel (2%) was recommended for local application before meals, and a multivitamin preparation was included as part of the supportive therapy.
Discussion
Herpes zoster, commonly known as shingles, gets its name from the Latin word “cingulum,” which means ‘girdle.’ This nomenclature is fitting because of the usual presentation of a unilateral rash that encircles the waist or torso, resembling a girdle. The term “zoster” has its roots in classical Greek, specifically referring to a belt-like binding (referred to as a zoster) used by warriors to secure their armor [8]. In 1988, Von Bokay put forward the initial hypothesis that both chickenpox and herpes zoster were induced by the same infectious agent. [9]. VZV, belonging to the family of eight herpes viruses affecting humans, exhibits a structure featuring an icosahedral nucleocapsid enveloped by lipids. The virus has a diameter of approximately 150–200 nm and a molecular weight of around 80 million [10].
The incidence of herpes zoster is significantly higher, up to 15 times more, in individuals with HIV, and it occurs in 25% of those diagnosed with Hodgkin’s lymphoma. In the broader population, the reported incidence of herpes zoster infection is 5.4% [11]. Typically, this infection affects individuals over the age of 45, with the highest frequency observed in those aged between 68 and 90 years [11]. In the case we are discussing, the patient is 60 years old, but with no history of HIV or lymphoma.
Herpes zoster progresses through three distinct stages: prodromal, active, and chronic [12]. Typically, it initiates with a prodrome characterized by mild-to-moderate burning or tingling or, in some instances, numbness in the skin of a specific dermatome [8]. This phase is often accompanied by symptoms such as fever, headache, general malaise, and, at times, stomach upset. The case we are discussing exhibited similar findings during the prodromal stage, although stomach upset was not specifically reported. Within approximately 48–72 hours from the onset of prodromal symptoms, a unilateral rash develops, characterized by erythematous maculopapular features along the dermatome. Over time, this rash evolves into vesicular lesions, marking the transition into the active stage [8].
Facial and intraoral lesions manifest unilaterally, serving as distinctive features of herpes zoster. The presented case illustrated the involvement of all three branches of the trigeminal nerve, featuring vesicular eruptions across the face and neck, accompanied by intraoral lesions. Additionally, the case showed engagement of the maxillary and mandibular divisions, with lesions exclusively on one side of the chin, malar area, and temporal region, including the preauricular area and ear. Additionally, intraoral lesions were observed on the buccal mucosa. The pain associated with shingles varies in severity, ranging from mild to intense, with even minor stimuli capable of triggering excruciating spasms. In this particular case, the pain was of severe intensity [13].
The lesions typically begin to dry, and crusts start forming within 3–5 days. The overall duration of the disease is typically period of 7 to 10 days. Though the restoration of hypopigmented skin to its normal state may take several weeks. In some instances, the vesicular rash may not manifest (referred to as zoster sine herpete), posing a challenge for diagnosis [14]. Interestingly, it is noted that pain tends to reduce when the rash is more active, but it resurfaces until the rash clears during the crusting and scaling phase [15]. Regardless of this, herpes zoster is not as infectious as the primary varicella infection. Though individuals with reactivated infections can transfer the VZV to non-immune contacts, with household transmission rates assessed as nearly 15%. [16]. The prolonged stage, characterized as postherpetic neuralgia (PHN), involves pain persisting for 1–3 months after the resolution of skin lesions but can extend for years or even decades [17]. Notably, 10–20% of individuals experience PHN following the acute phase of herpes zoster infection are predominantly elderly patients, accounting for over 20% of cases [18]. The pain associated with PHN is described as intermittent, sharp, or shocking allodynia, coupled with a continuous, typically deep pain, contributing significantly to morbidity.
Additional common complication of herpes zoster is the occurrence of secondary bacterial infections. In recent times, infections caused by group A β-haemolytic streptococci, including conditions like cellulitis and necrotizing fasciitis, have increasingly complicated the progression of varicella. Moreover, life-threatening complications such as skin scarring, keratitis, retinal necrosis resulting in blindness, keratouveitis, cranial and peripheral nerve palsies, cerebral ataxia, and pneumonia have occurred among patients [18]. Furthermore, herpes zoster infection has been associated with periapical lesions, root resorption, tooth exfoliation, and alveolar osteonecrosis [19,20]. In our case, this patient was recommended for topical soframycin to help prevent secondary bacterial infection and also advised to visit follow-ups for 4 weeks to monitor any other complications.
When the geniculate ganglion is affected, it can lead to James Ramsay Hunt’s syndrome, characterized by facial paralysis and the incidence of painful vesicular eruptions in the external auditory meatus and pinna of the ear [18]. But, in this case, there were no indications of facial paralysis regardless of the prevailing contribution of the condition. It’s a difficult challenge to culture VZV due to its labile nature and obtaining a sufficient sample from vesicular fluid. Other methods, such as direct immunofluorescence assay and PCR were used; among these, the direct immunofluorescence assay is faster, costly, and sensitive [18].
While it is a condition that tends to resolve on its own, the early initiation of both antiviral and symptomatic therapy significantly decreases morbidity. Commencing antiviral treatment within 72 hours of the onset of cutaneous eruption is crucial, as most virus replication ceases within this timeframe after the rash begins. Effective antiviral options include guanosine analogues such as acyclovir (800mg, 5 times/day for 7–10 days), famciclovir (500mg, 3 times/day for 7 days), and valacyclovir (1000mg, 3 times/day for 7 days). These antivirals are selectively monophosphorylated by the viral thymidine kinase and undergo further phosphorylation by cellular kinases, thereby inhibiting viral DNA. [21].
The administration of acyclovir (800mg, 5 times/day for 7–10 days) results in a reduction in the duration of viral shedding, impedes the occurrence of new lesions, hastens the healing process, and reduces the intensity of acute pain. Variable outcomes have been documented regarding the decrease in the frequency and duration of PHN [22]. In this case, the patient’s symptoms were relieved with acyclovir, and the patient was advised to follow up.
Valacyclovir, it is a prodrug of acyclovir, results in a serum acyclovir level 3–5 times higher than what is achieved through oral acyclovir therapy. The use of corticosteroids in the treatment of herpes zoster has been a subject of consideration. While their possible benefits include a moderate but significant acceleration in the rate of cutaneous healing and the relief of acute pain when used in combination with acyclovir, concerns about diminished immune response have overshadowed their advantages. Consequently, corticosteroids are contraindicated in immunocompromised patients. Conversely, no study has proved any effect of corticosteroids on the incidence or duration of PHN. It was recommended that patients should take corticosteroids along with antiviral therapy [22].
Tricyclic antidepressants can indeed serve as an alternative treatment option for managing certain aspects of herpes zoster, particularly in addressing neuropathic pain associated with the condition. Medications such as amitriptyline or nortriptyline may be considered for their analgesic effects in addition to antiviral therapy. However, the choice of treatment should be tailored to the individual’s specific symptoms and medical history, and consultation with a healthcare professional is crucial for appropriate management. Capsaicin is the sole topical preparation sanctioned for providing temporary relief from the pain linked to herpes zoster infections; however, its application is not recommended until the skin lesions have fully healed. Different clinical approaches must be followed to fasten the recovery process, such as maintaining cleanliness and dryness of cutaneous lesions, using sterile non-adherent dressing for lesion protection, applying topical applications such as Calamine lotion, employing sympathetic nerve obstructions, and prescribing analgesics like aspirin [18].
Recent medications for herpes zoster include Hexadecyloxypropyl-cidofovir (CMX 001), Nucleoside analogue H2G (Valamaciclovir), Helicase primase inhibitor (ASP2151), and two bicyclic nucleoside analogues (FV100) [23].
Conclusion
Herpes Zoster denotes the resurgence of the VZV in the host. Individuals experiencing Herpes Zoster infection may exhibit diverse symptoms, as observed in this case. Acyclovir, owing to its selective activation in infected cells and targeted inhibition of viral DNA polymerase, boasts a remarkably favorable therapeutic index. Administering Acyclovir at a dosage of 800 mg/5 times a day for 7 days proves to be an effective treatment for herpes zoster.
References
- Marra F, Parhar K, Huang B, Vadlamudi N. Risk factors for herpes zoster infection: a meta-analysis. Open Forum Infect Dis. 2020;7(1):ofaa005. doi:10.1093/ofid/ofaa005 PubMed | Crossref | Google Scholar
- Patil A, Goldust M, Wollina U. Herpes zoster: a review of clinical manifestations and management. Viruses. 2022;14(2):192. doi:10.3390/v14020192 PubMed | Crossref | Google Scholar
- Muchtar E, Koehler AB, Johnson MJ, et al. Humoral and cellular immune responses to recombinant herpes zoster vaccine in patients with chronic lymphocytic leukemia and monoclonal B cell lymphocytosis. Am J Hematol. 2022;97(1):90-98. doi:10.1002/ajh.26388 PubMed | Crossref | Google Scholar
- Freer G, Pistello M. Varicella-zoster virus infection: natural history, clinical manifestations, immunity, and current and future vaccination strategies. New Microbiol. 2018;41(2):95-105. Varicella-zoster virus infection: natural history, clinical manifestations, immunity and current and future vaccination strategies – PubMed
- Schmader K. Herpes zoster. Ann Intern Med. 2018;169(3):ITC19-ITC31. doi:10.7326/L18-0558 PubMed | Crossref | Google Scholar
- Raj SS, Verma P, Mahajan P, Puri A. Herpes zoster infection of the face: a case report with review of literature. J Indian Acad Oral Med Radiol. 2017;29(2):159-161. doi:10.4103/jiaomr.JIAOMR_91_16 Crossref | Google Scholar
- Soni A. Herpes zoster: a case series with varied clinical presentations. Indian J Case Rep. 2018;4(4):253-257. doi:10.32677/IJCR.2018.v04.i04.001 Crossref | Google Scholar
- Roxas M. Herpes zoster and postherpetic neuralgia: diagnosis and therapeutic considerations. Altern Med Rev. 2006;11(2):102-113. Herpes zoster and postherpetic neuralgia: diagnosis and therapeutic considerations – PubMed
- Hambleton S, Gershon AA. Preventing varicella-zoster disease. Clin Microbiol Rev. 2005;18(1):70-80. doi:10.1128/CMR.18.1.70-80.2005 PubMed | Crossref | Google Scholar
- Whitley RJ. Varicella-zoster virus infections. In: Fauci AS, Braunwald E, Isselbacher KJ, et al, eds. Harrison’s Principles of Internal Medicine. 14th ed. McGraw Hill; 1998:1086-1089. Varicella-Zoster Virus Infections | Harrison’s Principles of Internal Medicine, 21e | AccessMedicine | McGraw Hill Medical
- Vineet DA, Mithra R, Baskaran P, et al. Oro-facial herpes zoster: a case report with a detailed review of literature. Oral Maxillofac Pathol J. 2013;4:346-354. 02d054f332250213245db8c97f2218fe-Vineet.pdf
- Neville BW, Damm DD, Allen CM, et al. Viral infections. In: Neville BW, Damm DD, Allen CM, et al, eds. Oral and Maxillofacial Pathology. 3rd ed. Reed Elsevier India Pvt Ltd; 2005:222-223. Oral and Maxillofacial Pathology – 3rd Edition | Elsevier Shop
- Katz J, Cooper EM, Walther RR, et al. Acute pain in herpes zoster and its impact on health-related quality of life. Clin Infect Dis. 2004;39:342-348. doi:10.1086/421942 PubMed | Crossref | Google Scholar
- Tidwell E, Hutson B, Burkhart N, et al. Herpes zoster of the trigeminal nerve third branch: a case report and review of the literature. Int Endod J. 1999;32:61-66. doi:10.1046/j.1365-2591.1999.00187.x PubMed | Crossref | Google Scholar
- Strommen GL, Pucino F, Tight RR, et al. Human infection with herpes zoster: etiology, pathophysiology, diagnosis, clinical course and treatment. Pharmacotherapy. 1988;8:52-68. doi:10.1002/j.1875-9114.1988.tb04066.x PubMed | Crossref | Google Scholar
- Stankus SJ, Dlugopolski M, Packer D. Management of herpes zoster (shingles) and postherpetic neuralgia. Am Fam Physician. 2000;61:2437-2444. Management of Herpes Zoster (Shingles) and Postherpetic Neuralgia | AAFP
- Blasberg B, Greenberg MS. Orofacial pain. In: Greenberg MS, Glick M, eds. Burket’s Oral Medicine. 10th ed. BC Decker; 2003:330. Burket’s Oral Medicine Diagnosis & Treatment
- Mehta DN, Thakkar B, Asrani M. Herpes zoster of orofacial region-a review. Nat J Int Res Med. 2013;4:112-116 (PDF) Herpes Zoster of orofacial region- A review – | Semantic Scholar Google Scholar
- Ramchandani PL, Mellor TK. Herpes zoster associated with tooth resorption and periapical lesions. Br J Oral Maxillofac Surg. 2007;45:71-73. doi:10.1016/j.bjoms.2005.05.008 PubMed | Crossref | Google Scholar
- Schwartz O, Kvorning SA. Tooth exfoliation, osteonecrosis of the jaw, and neuralgia following herpes zoster of the trigeminal nerve. Int J Oral Surg. 1982;11:364-371. doi:10.1016/S0300-9785(82)80060-4 Crossref | Google Scholar
- Cohen JI, Brunell PA, Straus SE, et al. Recent advances in varicella-zoster virus infection. Ann Intern Med. 1999;130:922-932. doi:10.7326/0003-4819-130-11-199906010-00017 PubMed | Crossref | Google Scholar
- Gnann JW Jr, Whitley RJ. Clinical practice. Herpes zoster. N Engl J Med. 2002;347:340-346. doi:10.1056/NEJMcp013211 PubMed | Crossref | Google Scholar
- Janniger CK. Herpes zoster. Medscape. Updated July 21, 2021. Accessed March 6, 2025. Herpes Zoster
Acknowledgments
Not applicable
Funding
Not applicable
Author Information
Corresponding Author:
Bhandhavi V.
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Email: bvasireddy@medtigo.com
Co-Author:
T Sindhoori
Independent Researcher, Department of Content
medtigo India Pvt Ltd, Pune, India
Authors Contributions
All patient-related data were collected by Dr. Sindhoori. Dr. Bhandhavi V contributed to the writing of the manuscript, including the original draft preparation and subsequent review and editing to refine the final version.
Informed Consent
Not applicable
Conflict of Interest Statement
This case report is based on a clinical case published in the “Cases” section of medtigo.com. The authors declare no conflicts of interest related to this publication.
Guarantor
Not applicable
DOI
Cite this Article
Bhandhavi V, Sindhoori T. A 60-Year-Old Adult with an Erythematous Rash on the Face. medtigo J Med. 2023;1(1):e3062111. doi: 10.63096/medtigo3062111 Crossref









